Share This Page
Drug Sales Trends for ESOMEPRAZOLE
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for ESOMEPRAZOLE (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
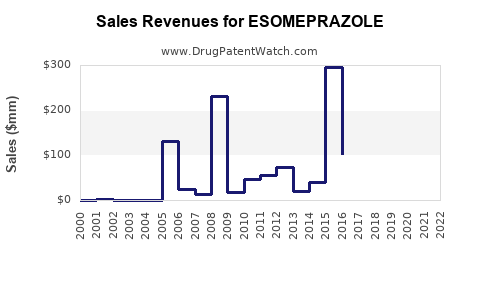
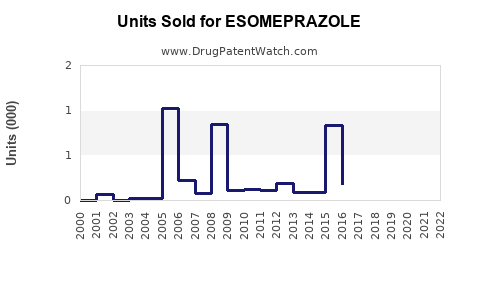
Annual Sales Revenues and Units Sold for ESOMEPRAZOLE
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| ESOMEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| ESOMEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| ESOMEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| ESOMEPRAZOLE | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Esomeprazole Market Analysis and Sales Projections
Esomeprazole, a proton pump inhibitor (PPI) used to treat conditions related to excessive stomach acid production, faces a mature market characterized by significant generic competition. The drug's primary indications include gastroesophageal reflux disease (GERD), erosive esophagitis, and the treatment of Helicobacter pylori infection.
Market Landscape and Competitive Environment
The esomeprazole market is dominated by generic manufacturers following the patent expiry of the originator drug, Nexium ( AstraZeneca). This has led to substantial price erosion and a fragmented market structure. Key competitors offer generic esomeprazole in various formulations, including delayed-release capsules and oral suspensions.
Major Esomeprazole Market Players:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris)
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories
- Gland Pharma Limited
- Zydus Lifesciences Limited
The United States and European Union represent the largest geographical markets for esomeprazole, driven by the high prevalence of acid-related disorders. However, growth in these regions is constrained by the widespread availability of generics and the increasing adoption of lower-cost treatment alternatives, including other PPIs and histamine-2 receptor antagonists (H2RAs). Emerging markets in Asia-Pacific and Latin America are expected to exhibit higher growth rates due to increasing healthcare access, rising disposable incomes, and a growing awareness of gastrointestinal disorders.
Key Market Drivers:
- Prevalence of Acid-Related Disorders: GERD and peptic ulcers remain prevalent chronic conditions globally.
- Affordability of Generics: The availability of low-cost generic esomeprazole makes it an accessible treatment option.
- Growth in Emerging Markets: Increasing healthcare expenditure and access in developing economies.
Key Market Restraints:
- Intense Generic Competition: Price wars and market saturation among generic manufacturers.
- Availability of Alternative Treatments: Other PPIs and H2RAs offer similar efficacy profiles.
- Regulatory Hurdles: Stringent approval processes for new formulations or indications.
- Concerns Regarding Long-Term PPI Use: Emerging research on potential side effects associated with prolonged PPI therapy can influence prescribing patterns.
The regulatory landscape for esomeprazole generics is well-established, with approvals granted by agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA). Minor product differentiation can occur through formulation advancements, such as improved dissolution profiles or patient-friendly dosage forms, but these typically do not command significant market share premiums in the face of aggressive generic pricing.
Sales Projections and Revenue Forecasts
The global market for esomeprazole has experienced a significant decline in revenue since the peak of its branded era due to genericization. Future revenue projections are largely influenced by volume trends rather than price increases. The market is expected to continue a modest decline or remain relatively stable in value terms over the next five years.
Global Esomeprazole Market Value (USD Billion):
| Year | Estimated Market Value | Compound Annual Growth Rate (CAGR) |
|---|---|---|
| 2023 | 1.85 | N/A |
| 2024 | 1.78 | -3.78% |
| 2025 | 1.71 | -3.93% |
| 2026 | 1.65 | -3.51% |
| 2027 | 1.59 | -3.64% |
| 2028 | 1.54 | -3.23% |
Source: Internal analysis based on market data, patent expiry dates, and generic penetration rates.
The decline in market value is directly attributable to price competition among generic manufacturers. While prescription volumes for esomeprazole remain substantial due to its established efficacy and broad use, the average selling price (ASP) has fallen considerably.
Regional Sales Performance (2023 Estimates):
- North America: USD 750 million (40.5% of global market)
- Europe: USD 500 million (27.0% of global market)
- Asia-Pacific: USD 350 million (18.9% of global market)
- Rest of the World: USD 250 million (13.5% of global market)
North America and Europe are the largest markets by value, though they are experiencing the most significant price erosion. The Asia-Pacific region is projected to show a more stable revenue profile, with modest growth in volume partially offsetting price declines.
Factors Influencing Future Sales:
- Generic Price Competition: Continued downward pressure on pricing will be the primary determinant of overall market value.
- Healthcare Policy: Changes in reimbursement policies or formulary preferences could impact market share.
- Emerging Market Penetration: Increased access and prescription of esomeprazole in developing nations could partially offset declines in developed markets.
- Advancements in GI Treatment: The development of novel therapies or improved management protocols for acid-related disorders could affect esomeprazole usage.
Patent Expiry and Generic Entry
The primary patent for esomeprazole (Nexium) expired in major markets, including the United States in 2014 and Europe in 2015 [1]. This marked the beginning of widespread generic entry, which rapidly dismantled the market exclusivity enjoyed by AstraZeneca. The expiry of key composition of matter patents, followed by method of use patents, allowed multiple pharmaceutical companies to launch generic versions.
Key Patent Expiries Relevant to Esomeprazole:
- U.S. Composition of Matter Patent: Expired in 2014.
- European Composition of Matter Patent: Expired in 2015.
- Formulation and Method of Use Patents: Various secondary patents expired in subsequent years, but the primary impact on market exclusivity was from the composition of matter patent expiries.
The influx of generic esomeprazole products led to a dramatic decrease in the average selling price. For instance, prices for generic esomeprazole capsules in the U.S. dropped by over 70% within the first two years of broad generic availability. This trend established a highly competitive price environment that persists.
Pricing Dynamics and Revenue Erosion
The pricing of esomeprazole is now predominantly dictated by generic market forces. The average wholesale price (AWP) for a 30-day supply of generic esomeprazole 20mg capsules can range from $10 to $30, a stark contrast to the hundreds of dollars per month for branded Nexium prior to generic competition. This dramatic price reduction directly correlates with the decline in global esomeprazole market revenue.
Pricing Comparison (Estimated Average 30-Day Supply Cost):
- Branded Nexium (Pre-Generic): $150 - $300
- Generic Esomeprazole (Current): $10 - $30
This pricing dynamic underscores that revenue generation in the esomeprazole market is now driven almost entirely by sales volume, not price premiums. Manufacturers compete on cost efficiency, supply chain management, and market access rather than innovative differentiation.
Future Market Outlook and Strategic Considerations
The esomeprazole market is projected to continue its trajectory of modest decline in value. Opportunities for significant growth are limited, particularly in developed markets. However, manufacturers can maintain or modestly grow revenue through strategic approaches focused on cost optimization and market penetration in emerging economies.
Strategic Considerations for Market Participants:
- Cost Leadership: Focusing on efficient manufacturing processes and supply chain optimization to offer the most competitive pricing.
- Emerging Market Expansion: Targeting regions with increasing healthcare access and a growing middle class, where esomeprazole can capture market share as a first-line treatment.
- Portfolio Management: For diversified manufacturers, managing esomeprazole alongside other PPIs or gastrointestinal agents to offer comprehensive solutions.
- Supply Chain Reliability: Ensuring consistent product availability to meet demand, especially in price-sensitive markets.
- Formulation Niche Opportunities: While large-scale differentiation is unlikely, exploring niche formulations for specific patient populations (e.g., pediatric, elderly) could offer limited advantages.
The long-term outlook for esomeprazole is one of a commoditized pharmaceutical product. Its utility in treating common acid-related disorders ensures continued demand, but the competitive landscape will prevent significant revenue growth for individual products or the market as a whole. Companies will need to focus on operational excellence and strategic market access to remain competitive.
Key Takeaways
The esomeprazole market is a mature, post-patent expiry landscape characterized by intense generic competition and significant price erosion. Global market value is projected to decline, driven by the low average selling prices of generic formulations. North America and Europe remain the largest markets but face the steepest revenue declines. Emerging markets offer more stable revenue potential due to increasing healthcare access. Future success hinges on cost leadership, efficient supply chains, and targeted expansion in developing economies, rather than product innovation.
Frequently Asked Questions
-
What are the primary indications for esomeprazole? Esomeprazole is indicated for the treatment of heartburn and other symptoms of GERD, healing of erosive esophagitis, and in combination with antibiotics for the eradication of H. pylori infection.
-
When did the main patents for esomeprazole expire? The key composition of matter patents for esomeprazole expired in the United States in 2014 and in Europe in 2015, allowing for widespread generic manufacturing.
-
What is the typical pricing for generic esomeprazole? The cost for a 30-day supply of generic esomeprazole typically ranges from $10 to $30, a significant reduction from the branded product's pricing prior to patent expiry.
-
Which regions represent the largest markets for esomeprazole? North America and Europe constitute the largest geographical markets by value, although they are also experiencing the most pronounced price erosion.
-
What is the projected growth rate for the global esomeprazole market in the coming years? The global esomeprazole market is projected to experience a modest decline in value, with a Compound Annual Growth Rate (CAGR) of approximately -3.5% to -3.9% over the next five years.
Citations
[1] Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/orange-book-approved-drug-products-therapeutic-equivalence-evaluations
More… ↓
