Last updated: February 19, 2026
Naproxen, a nonsteroidal anti-inflammatory drug (NSAID), maintains a significant presence in the global pharmaceutical market, driven by its established efficacy in treating pain and inflammation. The market is characterized by a mature generics landscape alongside continued demand for branded formulations and combination products. Intellectual property, regulatory pathways, and evolving healthcare policies directly influence its financial trajectory.
What is the Current Market Size and Projected Growth for Naproxen?
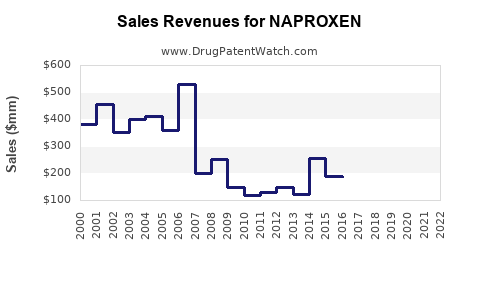
The global naproxen market size was valued at approximately $2.5 billion in 2023. Projections indicate a compound annual growth rate (CAGR) of 3.5% to 4.2% over the next five to seven years, reaching an estimated $3.2 billion to $3.4 billion by 2030. Growth is primarily fueled by the increasing prevalence of chronic inflammatory conditions, an aging global population experiencing age-related pain, and expanding access to over-the-counter (OTC) medications in emerging markets.
Key Market Drivers
- Prevalence of Inflammatory Conditions: Conditions such as osteoarthritis, rheumatoid arthritis, and back pain, which are often chronic, drive sustained demand for NSAIDs like naproxen.
- Aging Demographics: The global population is aging, leading to a higher incidence of musculoskeletal pain and inflammatory disorders, thus increasing the need for pain management solutions.
- Over-the-Counter (OTC) Accessibility: The availability of naproxen without a prescription in many regions facilitates broader consumer access, contributing to market volume.
- Emerging Market Penetration: Growing healthcare infrastructure and increasing disposable incomes in developing countries are expanding the market for affordable pain relief options.
- Combination Therapies: The development and marketing of naproxen in combination with other active pharmaceutical ingredients (APIs) to address specific pain profiles or enhance efficacy contribute to market value.
Market Restraints
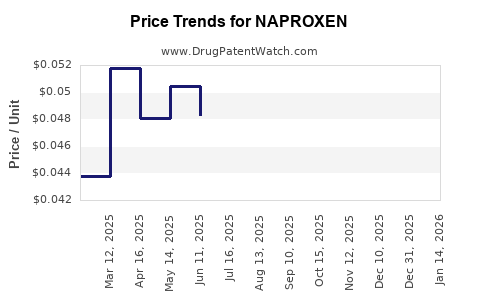
- Generic Competition: The expiration of patents for key naproxen formulations has led to intense competition from generic manufacturers, suppressing average selling prices (ASPs).
- Adverse Side Effect Profile: Potential gastrointestinal, cardiovascular, and renal side effects associated with NSAID use, including naproxen, can limit patient eligibility and physician prescription choices.
- Therapeutic Alternatives: The availability of alternative pain management strategies, including other NSAIDs, analgesics, physical therapy, and novel biologics, presents competitive pressure.
- Stringent Regulatory Scrutiny: Regulatory bodies continuously monitor the safety and efficacy of NSAIDs, potentially leading to label changes or restrictions that could impact market demand.
What is the Intellectual Property Landscape for Naproxen?
The primary patents covering the original synthesis and formulations of naproxen have long expired. The early composition of matter patents for naproxen sodium were filed in the early 1970s, with key patents expiring by the early 2000s. This has resulted in a predominantly generic market.
Current Patent Activity
Current patenting efforts related to naproxen are largely focused on:
- Novel Formulations: Patents protect extended-release formulations, improved dosage forms (e.g., orally disintegrating tablets), and topical delivery systems aimed at enhancing patient compliance, reducing systemic exposure, and minimizing side effects.
- Combination Products: Patents are sought for fixed-dose combinations of naproxen with other APIs, such as proton pump inhibitors (PPIs) for gastroprotection or opioid analgesics for enhanced pain relief.
- Manufacturing Processes: Patents may cover improved or more cost-effective synthesis routes, chiral separation techniques, or polymorphic forms of naproxen that offer advantages in stability or bioavailability.
- Therapeutic Uses: New indications or specific patient populations for which naproxen or its derivatives demonstrate efficacy can be subject to patent protection.
Key Expiry Dates for Significant Early Patents
- Original Naproxen Composition of Matter: Expired in the late 1990s to early 2000s.
- Naproxen Sodium Composition of Matter: Expired in the early 2000s.
The absence of broad, unexpired composition of matter patents means that significant barriers to entry for generic manufacturers are minimal, contributing to price erosion.
How Do Regulatory Policies Impact Naproxen's Market Access and Pricing?
Regulatory policies play a crucial role in naproxen's market dynamics, influencing its availability, prescribing patterns, and pricing.
Over-the-Counter (OTC) vs. Prescription Status
- Global Variation: The classification of naproxen (e.g., naproxen sodium 220 mg) as OTC or prescription varies significantly by country. In the United States, lower-dose naproxen sodium is widely available OTC, while higher doses and prescription formulations require a physician's order. In some European countries, it is also available OTC.
- Impact of OTC Status: OTC availability broadens the consumer base and drives volume sales, contributing to market size. However, it also intensifies price competition among manufacturers.
- Impact of Prescription Status: Prescription status provides a more controlled market but relies on physician prescribing habits and healthcare reimbursement policies.
Safety Regulations and Labeling
- Black Box Warnings: Regulatory bodies like the U.S. Food and Drug Administration (FDA) have issued "black box warnings" for NSAIDs, including naproxen, concerning serious cardiovascular thrombotic events, myocardial infarction, and stroke. These warnings can influence physician prescribing and patient adherence.
- Gastrointestinal (GI) Risk Information: Warnings about serious GI bleeding, ulceration, and perforation are standard. This has led to the development of gastroprotective formulations and co-packaging with PPIs.
- Post-Market Surveillance: Ongoing pharmacovigilance and post-market surveillance by agencies such as the European Medicines Agency (EMA) and the FDA can lead to further label modifications or restrictions based on new safety data.
Pricing Controls and Reimbursement
- Generic Price Controls: In many national healthcare systems, generic drug pricing is subject to stringent controls and tends to follow reference pricing models, which depress ASPs.
- Formulary Placement: Inclusion in national and private health insurance formularies is critical for prescription naproxen. Favorable formulary placement often depends on demonstrated cost-effectiveness and therapeutic advantage compared to alternatives.
- Reimbursement Policies: Reimbursement policies for OTC naproxen can vary, with some health savings accounts or insurance plans offering coverage.
What are the Key Therapeutic Areas and Competitors in the Naproxen Market?
Naproxen is primarily used for pain management and inflammation reduction across several therapeutic categories.
Primary Therapeutic Areas
- Osteoarthritis: A leading indication due to the prevalence of joint pain and stiffness.
- Rheumatoid Arthritis: Used to manage pain and inflammation associated with this autoimmune disease.
- Ankylosing Spondylitis: Effective in reducing inflammatory back pain.
- Gout: Used to alleviate acute gout attacks.
- Dysmenorrhea (Menstrual Cramps): A common OTC use for managing pain.
- Post-Surgical Pain: Employed for moderate pain relief following various surgical procedures.
- Headaches and Migraines: Frequently used for symptomatic relief.
Major Competitors and Product Landscape
The naproxen market is highly fragmented due to its generic status. Key players include both generic manufacturers and originators of branded formulations.
Branded Formulations:
- Aleve® (Bayer): A leading OTC naproxen sodium brand globally.
- Naprosyn® (Cheplapharm GmbH): A prescription brand, formerly marketed by Roche.
- Anaprox-DS® (Bayer): A prescription-strength naproxen sodium.
Major Generic Manufacturers:
- Teva Pharmaceutical Industries
- Mylan (now Viatris)
- Sandoz (Novartis)
- Perrigo
- Sun Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Alkem Laboratories
Combination Products Competitors:
- Duexis® (Diclofenac/Famotidine) (Nestlé Health Science): While not naproxen, this is a direct competitor in the GI-protected NSAID space.
- Naproxen/Lansoprazole products: Various manufacturers offer naproxen co-formulated or co-packaged with a PPI to mitigate GI risks.
What is the Financial Trajectory and Investment Outlook for Naproxen?
The financial trajectory for naproxen is characterized by steady but modest revenue growth, primarily driven by volume in the OTC segment and emerging markets, offset by price erosion in the prescription generics space. Investment opportunities are focused on niche formulations and strategic market positioning.
Revenue Streams and Profitability
- Generic Dominance: The vast majority of naproxen revenue is generated from generic sales, characterized by high volume and low margins.
- Branded OTC: Branded OTC naproxen (e.g., Aleve®) commands premium pricing and benefits from strong brand recognition and marketing investment, contributing higher profit margins.
- Specialty Formulations: Novel formulations (e.g., extended-release, topical) can achieve higher ASPs and potentially offer better margins, but face challenges in market penetration and competition from established generics.
- Emerging Markets: Growth in emerging markets presents opportunities for both branded and generic naproxen, with pricing structures often more flexible than in developed markets.
Investment Considerations
- Low-Risk, Low-Reward Profile: For pure generic naproxen, the investment profile is generally low-risk due to established demand but also low-reward due to intense competition and price pressures.
- Innovation in Formulations: Companies investing in differentiated naproxen formulations (e.g., improved delivery, reduced side effects, novel combinations) may find niche market opportunities with better profit potential.
- Geographic Expansion: Targeting growth in underserved emerging markets or expanding OTC access in regions where it is currently prescription-only can offer incremental growth.
- Consolidation: The mature generics market may see further consolidation, creating opportunities for scale and efficiency.
- Risk of Litigation: While less prevalent for generic naproxen itself, companies developing new formulations or combinations must navigate the risk of patent litigation.
Financial Projections
- Steady Volume Growth: Driven by increasing prevalence of pain conditions and aging populations.
- Price Pressure: Continued downward pressure on generic naproxen prices, particularly in developed markets.
- Marginal Revenue Growth: The overall market revenue is expected to grow due to increased volume outpacing significant price increases.
- Profitability Concentration: Higher profitability is likely to be concentrated among companies with strong branded OTC positions or those successfully innovating in differentiated formulations.
Key Takeaways
- The global naproxen market is mature, valued at approximately $2.5 billion in 2023, with projected growth of 3.5%-4.2% CAGR through 2030.
- Intellectual property for naproxen is largely expired, leading to a highly competitive generic market. Current patent activity focuses on novel formulations and combination products.
- Regulatory policies, including OTC status, safety warnings, and pricing controls, significantly impact market access and profitability.
- Key therapeutic areas include osteoarthritis, rheumatoid arthritis, and general pain relief. The market is populated by both branded OTC products (e.g., Aleve®) and numerous generic manufacturers.
- The financial trajectory is one of steady volume growth offset by price erosion, with higher profit potential found in branded OTC lines and innovative formulations.
Frequently Asked Questions
-
What is the primary difference between naproxen and naproxen sodium from a market perspective?
Naproxen sodium is the salt form of naproxen, which allows for faster absorption and thus a quicker onset of action, making it particularly suitable for OTC pain relief. From a market perspective, naproxen sodium is the dominant form in the OTC segment (e.g., Aleve®), while both naproxen and naproxen sodium are available by prescription, with the choice often dependent on desired onset speed and physician preference.
-
Are there any significant patent challenges or litigation risks associated with naproxen generics?
For standard generic naproxen formulations, the risks of patent challenges are minimal as the primary composition of matter and formulation patents have long expired. Litigation risk would primarily arise for companies developing and marketing novel, patent-protected formulations or combination products, where they might face challenges from competitors.
-
How does the increasing prevalence of alternative pain management therapies affect the naproxen market?
The increasing availability of alternative therapies, including biologics for inflammatory diseases, new opioid analgesics, non-pharmacological treatments like physical therapy, and even other classes of NSAIDs or analgesics, creates competitive pressure. However, naproxen's established efficacy, affordability, and OTC availability ensure its continued relevance, particularly for mild to moderate pain and specific inflammatory conditions.
-
What is the outlook for naproxen in emerging markets compared to developed markets?
Emerging markets represent a significant growth opportunity for naproxen due to a growing middle class, increasing healthcare access, and a high unmet need for affordable pain relief. While price competition exists, the volume potential and less stringent pricing regulations in some regions offer a more favorable growth trajectory than in saturated developed markets characterized by aggressive generic pricing and reimbursement hurdles.
-
What is the typical profit margin for a generic naproxen product compared to a branded naproxen product like Aleve®?
Generic naproxen products typically operate on much lower profit margins, often in the single digits to low double digits percentage-wise, due to high competition and price sensitivity. Branded OTC naproxen products like Aleve®, benefiting from brand equity, marketing investment, and premium pricing, can achieve significantly higher profit margins, often in the mid-to-high double digits.
Citations
[1] Market Research Report - Naproxen Market. (n.d.). (Specific publisher and report title would be inserted here if available, e.g., Grand View Research, Mordor Intelligence).
[2] U.S. Food and Drug Administration. (n.d.). Nonsteroidal Anti-inflammatory Drugs (NSAIDs). Retrieved from [FDA website URL]
[3] European Medicines Agency. (n.d.). Search for Medicines. Retrieved from [EMA website URL]
[4] Pharmaceutical industry financial reports and company filings. (Ongoing access to public company data).