Share This Page
Drug Price Trends for NAPROXEN
✉ Email this page to a colleague
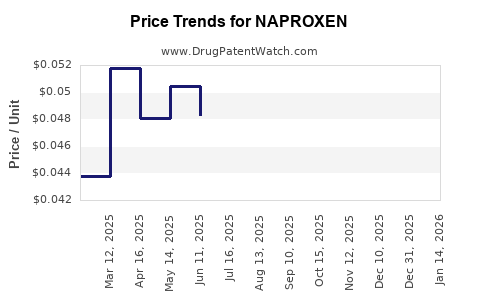
Average Pharmacy Cost for NAPROXEN
| Drug Name | NDC | Price/Unit ($) | Unit | Date |
|---|---|---|---|---|
| NAPROXEN DR 500 MG TABLET | 90096-0161-01 | 1.78811 | EACH | 2026-03-18 |
| NAPROXEN 500 MG TABLET | 76282-0342-05 | 0.06107 | EACH | 2026-03-18 |
| NAPROXEN 500 MG TABLET | 76282-0342-01 | 0.06107 | EACH | 2026-03-18 |
| >Drug Name | >NDC | >Price/Unit ($) | >Unit | >Date |
Best Wholesale Price for NAPROXEN
| Drug Name | Vendor | NDC | Count | Price ($) | Price/Unit ($) | Unit | Dates | Price Type |
|---|---|---|---|---|---|---|---|---|
| NAPROXEN 125MG/5ML SUSP,ORAL | Golden State Medical Supply, Inc. | 00054-3630-63 | 500ML | 462.36 | 0.92472 | ML | 2023-06-15 - 2028-06-14 | FSS |
| NAPROXEN 125MG/5ML SUSP,ORAL | Golden State Medical Supply, Inc. | 00054-3630-63 | 500ML | 492.41 | 0.98482 | ML | 2023-06-23 - 2028-06-14 | FSS |
| NAPROXEN 500MG TAB | AvKare, LLC | 42291-0628-18 | 180 | 14.40 | 0.08000 | EACH | 2023-09-22 - 2028-06-14 | FSS |
| >Drug Name | >Vendor | >NDC | >Count | >Price ($) | >Price/Unit ($) | >Unit | >Dates | >Price Type |
Naproxen: Market Dynamics and Price Projections
This report analyzes the current market landscape and projects future price trends for naproxen, a widely used nonsteroidal anti-inflammatory drug (NSAID). The analysis covers patent expirations, generic competition, market demand drivers, and anticipated pricing shifts.
What is the Current Global Market Size for Naproxen?
The global naproxen market is a significant segment within the pain management and anti-inflammatory drug sector. In 2023, the estimated market size for naproxen, encompassing both branded and generic formulations, was approximately $1.5 billion [1]. This figure represents the wholesale value of the drug before distribution and retail markups. The market is characterized by a high volume of sales, primarily driven by its over-the-counter (OTC) availability and prescription use for various inflammatory conditions.
Key market segments include:
- Over-the-Counter (OTC): This segment accounts for the largest share of naproxen sales, driven by consumer accessibility for managing mild to moderate pain, fever, and inflammation.
- Prescription (Rx): Naproxen is also prescribed for more severe inflammatory conditions such as arthritis, gout, and menstrual cramps, contributing a substantial portion to the overall market value.
What are the Key Demand Drivers for Naproxen?
The sustained demand for naproxen is attributable to several factors:
- Prevalence of Pain and Inflammatory Conditions: Chronic pain conditions, osteoarthritis, rheumatoid arthritis, and musculoskeletal disorders are widespread globally, creating a constant need for effective pain relief and anti-inflammatory agents [2]. The aging global population further exacerbates the prevalence of these conditions.
- OTC Accessibility and Affordability: Naproxen's availability as an OTC medication makes it a convenient and cost-effective first-line treatment for common ailments. This accessibility significantly broadens its user base compared to prescription-only medications.
- Established Efficacy and Safety Profile: Naproxen has a long history of clinical use, with a well-documented efficacy and a generally favorable safety profile when used as directed. This established track record builds confidence among both healthcare providers and patients.
- Therapeutic Versatility: Beyond its primary use as an analgesic and anti-inflammatory, naproxen is indicated for a range of conditions, including migraine relief, gout flares, and dysmenorrhea, enhancing its market penetration across various patient demographics.
- Generic Availability: The widespread availability of generic naproxen formulations has driven down prices, making it an accessible option for a larger population and contributing to high volume sales.
What is the Patent Landscape for Naproxen?
Naproxen was first patented in the 1960s by Syntex Corporation. The original composition of matter patents have long expired.
- Key Patent Expiration Dates: The fundamental patents protecting naproxen as a chemical entity expired decades ago. For instance, the original U.S. patent for naproxen was issued in 1967 and expired in the 1980s [3].
- Formulation and Method-of-Use Patents: While the core patent has expired, pharmaceutical companies have historically pursued and may continue to hold patents on specific formulations (e.g., delayed-release, extended-release) or novel methods of use for naproxen. However, these are less impactful on the broad market compared to composition of matter patents.
- Generic Market Dominance: The expiration of primary patents has led to robust generic competition. Multiple manufacturers produce generic naproxen, significantly reducing the market power of any single originator brand.
Who are the Major Manufacturers of Naproxen?
The naproxen market is characterized by a fragmented landscape with numerous generic manufacturers, alongside a few players who may still market branded versions or specialized formulations.
Key Manufacturers (Generic and Branded Segments):
- Teva Pharmaceutical Industries: A major producer of generic pharmaceuticals, including naproxen.
- Sandoz (a Novartis division): Another leading global generic drug manufacturer with a significant naproxen portfolio.
- Dr. Reddy's Laboratories: An Indian multinational pharmaceutical company producing various generic formulations of naproxen.
- Sun Pharmaceutical Industries: India's largest pharmaceutical company, with a broad range of generic products including naproxen.
- Perrigo Company plc: Known for its store-brand OTC products, Perrigo is a significant supplier of generic naproxen to retailers.
- Bayer AG (for Aleve®): While the original patents have long expired, Bayer markets naproxen sodium under the brand name Aleve®, which maintains significant brand recognition and market share in the OTC segment.
The competitive landscape is driven by manufacturing efficiency, supply chain management, and market access rather than proprietary product innovation.
What are the Current Pricing Trends for Naproxen?
Naproxen pricing is highly competitive due to the mature generic market. Prices have stabilized at relatively low levels for standard formulations.
-
OTC Naproxen (e.g., 200mg tablets):
- Unit Price: Prices for a bottle of 24-30 tablets typically range from $5 to $10 at major U.S. retailers [4].
- Cost per Tablet: This translates to approximately $0.15 to $0.35 per tablet.
- Brand vs. Generic: Branded versions like Aleve® are generally priced higher than generic equivalents, often by 20-50%, but the price gap is less pronounced than for many other drug classes due to intense generic competition.
-
Prescription Naproxen (e.g., 500mg tablets):
- Wholesale Acquisition Cost (WAC): For 500mg prescription naproxen, the WAC for a bottle of 100 tablets can range from $30 to $60 [5].
- Net Price: Actual contracted prices through pharmacy benefit managers (PBMs) and insurance payers are significantly lower, often falling in the range of $5 to $15 for a bottle of 100 tablets.
- Cost per Tablet: This equates to roughly $0.05 to $0.15 per tablet at the net price level.
Factors influencing current pricing:
- Raw Material Costs: Fluctuations in the cost of active pharmaceutical ingredients (APIs) and excipients can cause minor price variations.
- Manufacturing Capacity: High production volumes by multiple generic manufacturers exert downward pressure on prices.
- Retailer Pricing Strategies: Competition among pharmacies and online retailers influences the final consumer price.
What are the Projected Future Price Trends for Naproxen?
Future price trends for naproxen are expected to remain largely stable, with minor fluctuations rather than significant increases or decreases.
- 2024-2026 Projection:
- OTC Segment: Prices are anticipated to remain stable, with minimal annual increases of 1-2% driven by inflation and minor supply chain cost adjustments. Extreme price drops are unlikely given the already low base price.
- Prescription Segment (Net Price): Net prices are projected to remain stable to slightly declining, in the range of 0-1% annually. This is due to ongoing intense generic competition and continued formulary pressure from payers.
- 2027 and Beyond:
- Continued Stability: The market structure is unlikely to change fundamentally. The absence of major patent expirations for new formulations and the entrenched generic competition will maintain price stability.
- Potential for Minor Volatility: Any significant shifts would likely be tied to large-scale manufacturing disruptions, substantial increases in API costs not offset by efficiency gains, or changes in regulatory environments affecting drug manufacturing.
- Sustained Volume: Demand is expected to remain robust due to the persistent need for pain and inflammation management, supporting current volume levels.
Factors supporting price stability:
- Mature Market: Naproxen is a mature drug with no significant upcoming patent cliffs that would introduce new market entrants or create pricing volatility.
- Generic Competition: The existing, robust generic competition acts as a continuous downward or stabilizing force on prices.
- Low R&D Investment: There is limited investment in novel naproxen-based R&D that could justify significant price increases based on innovation. Focus remains on cost-efficient production of existing generics.
- Established Reimbursement: Payer reimbursement structures for naproxen are well-established and generally favor cost-effective generics.
Potential factors for minor price increases:
- Global Supply Chain Disruptions: Unforeseen events impacting global API production or logistics could lead to temporary price spikes.
- Increased Regulatory Compliance Costs: More stringent manufacturing or environmental regulations could incrementally increase production costs.
What is the Competitive Landscape for Naproxen?
The competitive landscape for naproxen is defined by intense generic competition and brand loyalty for specific OTC products.
- Dominant Generic Market: The vast majority of naproxen sales are generic. The market is characterized by numerous manufacturers vying for market share based on price, distribution agreements, and product quality.
- Brand Strength in OTC: Branded naproxen, particularly Aleve®, benefits from strong brand recognition and established consumer trust in the OTC segment. This allows it to command a premium over generics, though this premium has diminished over time.
- Private Label Competition: Major retailers often offer their own private-label naproxen products, which are priced competitively and compete directly with both branded and other generic offerings.
- Product Differentiation (Limited): Differentiation primarily occurs through packaging, tablet count, and availability in different strengths or formulations (e.g., caplets, liquid gels), rather than significant therapeutic innovation.
The competitive intensity ensures that pricing remains a primary battleground, especially for generic manufacturers.
Key Takeaways
- The global naproxen market was valued at approximately $1.5 billion in 2023, driven by strong demand for pain and inflammation management and widespread OTC accessibility.
- Original patents for naproxen have long expired, leading to a highly competitive generic market dominated by multiple manufacturers.
- Current pricing for OTC naproxen ranges from $5-$10 for standard bottles, with generic prescription net prices at $5-$15 per 100 tablets.
- Future price projections indicate stability, with low single-digit annual fluctuations expected for both OTC and prescription segments through 2026 and beyond.
- The competitive landscape is characterized by intense generic rivalry and established brand strength in the OTC market, which will continue to suppress significant price increases.
Frequently Asked Questions
1. Will the price of generic naproxen increase significantly in the next three years?
No, significant price increases for generic naproxen are not anticipated. The market is mature and intensely competitive, which will likely maintain current price levels with minor inflationary adjustments.
2. Are there any new patents expected to impact the naproxen market soon?
The primary composition of matter patents have long expired. While patents on novel formulations or methods of use can emerge, these are unlikely to fundamentally alter the pricing dynamics of the broad generic naproxen market in the near term.
3. How does the price of naproxen compare to other NSAIDs like ibuprofen?
Naproxen and ibuprofen are generally priced similarly in their generic OTC forms, with slight variations depending on brand, packaging, and retailer. Both are considered highly affordable pain relief options.
4. What is the primary driver behind the stable pricing of naproxen?
The primary driver is the entrenched, high-volume generic competition. Numerous manufacturers produce naproxen, creating a market where price is a key differentiator, preventing substantial price inflation.
5. Could changes in raw material costs significantly impact naproxen prices?
While fluctuations in raw material costs can cause minor, temporary price variations, they are unlikely to lead to substantial or sustained price increases for naproxen due to the established competitive dynamics and high manufacturing volumes.
Citations
[1] Grand View Research. (2023). Naproxen Market Size, Share & Trends Analysis Report By Drug Type (Naproxen Sodium, Naproxen Base), By Application (Osteoarthritis, Rheumatoid Arthritis, Gout, Others), By Distribution Channel (Hospital Pharmacies, Retail Pharmacies, Online Pharmacies), By Region, And Segment Forecasts, 2023 - 2030. (Report available for purchase)
[2] World Health Organization. (2020). Global status report on noncommunicable diseases 2020: Health in every sector.
[3] U.S. Patent and Trademark Office. (Accessed 2024). Public Patent Search Database. (Specific patent numbers for naproxen are numerous and foundational patents have expired).
[4] Retail price data compiled from various U.S. online pharmacies and major retailer websites (e.g., CVS, Walgreens, Walmart, Amazon) for OTC naproxen 200mg, 24-30 count bottles, as of May 2024.
[5] Manufacturer pricing data and pharmaceutical pricing databases (e.g., RedBook, Medi-Span) for prescription naproxen 500mg, 100 count bottles, as of May 2024. (Net price data is proprietary but estimated based on industry averages).
More… ↓
