Last updated: February 19, 2026
Atovaquone, an antimalarial and antipneumocystis medication, is a key component in combination therapies for these infectious diseases. Its market performance is driven by the prevalence of malaria, particularly in endemic regions, and the incidence of Pneumocystis jirovecii pneumonia (PCP) in immunocompromised individuals, such as those with HIV/AIDS or undergoing chemotherapy. Patent expirations have led to generic competition, impacting pricing and market share for originator products.
What are the primary indications for Atovaquone?
Atovaquone is primarily indicated for two main therapeutic areas:
- Malaria Prevention and Treatment: It is used in combination with proguanil (e.g., Malarone) for the prevention and treatment of Plasmodium falciparum malaria. This indication is particularly relevant in regions with high malaria endemicity [1]. The World Health Organization (WHO) recommends artemisinin-based combination therapies (ACTs) as first-line treatments for uncomplicated P. falciparum malaria, and atovaquone/proguanil is one such option [2].
- Pneumocystis Pneumonia (PCP) Prophylaxis and Treatment: Atovaquone is a critical treatment and prophylactic agent for PCP, a life-threatening opportunistic infection caused by the fungus Pneumocystis jirovecii. It is widely used in immunocompromised patients, including those with HIV/AIDS, organ transplant recipients, and cancer patients undergoing immunosuppressive therapy [3].
What is the patent landscape for Atovaquone?
The original patents protecting atovaquone and its initial formulations have expired in major markets. For instance, the primary patent for atovaquone itself, as a compound, expired decades ago. Combination therapies, such as atovaquone/proguanil, also had their associated patents expire.
Key aspects of the patent landscape include:
- Expired Composition of Matter Patents: The fundamental patent protecting the chemical structure of atovaquone has long expired, allowing for generic manufacturing.
- Expired Formulation and Use Patents: Patents covering specific pharmaceutical formulations (e.g., tablet, oral suspension) and new therapeutic uses have also largely expired. This has facilitated the entry of multiple generic versions of atovaquone and its combination products.
- Limited Remaining Patent Protection: While the core patents are expired, there may be residual patent protection for specific, novel formulations or manufacturing processes that are currently in use or under development. However, these are generally not broad enough to prevent generic competition of established products.
- Generic Entry: The expiration of key patents has led to significant generic competition, particularly for the atovaquone/proguanil combination, impacting the pricing power of originator brands.
Who are the major manufacturers and market players for Atovaquone?
The market for atovaquone is characterized by the presence of both originator and generic manufacturers.
- Originator Products:
- Malarone (atovaquone/proguanil) by GlaxoSmithKline (GSK): Historically, Malarone has been a leading branded product for malaria prevention and treatment. GSK also markets its pediatric formulation.
- Mepron (atovaquone) by GlaxoSmithKline (GSK): This product is primarily used for PCP treatment and prophylaxis.
- Generic Manufacturers: Following patent expiries, numerous generic companies have entered the market, producing both atovaquone as a single agent and the atovaquone/proguanil combination. These include, but are not limited to:
- Teva Pharmaceutical Industries
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Cipla
- Viatris (formerly Mylan and Upjohn)
- BMS Pharmaceuticals (for Mepron generics)
The presence of these generic players intensifies competition, leading to price erosion and a focus on market access and cost-effectiveness.
What is the global market size and growth forecast for Atovaquone?
The global market size for atovaquone is difficult to isolate precisely as it is often reported within broader antiparasitic or anti-infective drug categories, particularly for the atovaquone/proguanil combination. However, key market drivers and trends provide insight into its financial trajectory.
- Market Drivers:
- Prevalence of Malaria: Endemic regions, particularly in sub-Saharan Africa, continue to drive demand for antimalarials. The WHO estimates that in 2022, there were 249 million malaria cases and 608,000 malaria deaths globally [2]. While ACTs are the first line, atovaquone/proguanil remains a significant option.
- HIV/AIDS Prevalence: The global HIV/AIDS epidemic, particularly in resource-limited settings, sustains demand for PCP prophylaxis and treatment. While advancements in antiretroviral therapy have reduced PCP incidence in some developed nations, it remains a critical concern in areas with limited access to comprehensive HIV care.
- Immunosuppression Trends: The increasing use of immunosuppressive therapies in oncology, autoimmune diseases, and organ transplantation contributes to the demand for PCP prophylaxis.
- Market Challenges:
- Generic Competition: The widespread availability of generics has led to significant price reductions, limiting the overall revenue potential for originator brands and impacting market value.
- Drug Resistance: While less prevalent with atovaquone/proguanil compared to some other antimalarials, the emergence of resistance remains a concern and can impact treatment choices.
- Availability of Alternative Therapies: In both malaria and PCP, alternative treatment and prophylaxis options exist, creating a competitive landscape.
- Growth Forecast: The market for atovaquone-containing products is expected to exhibit modest growth, primarily driven by demand in malaria-endemic regions and the persistent need for PCP management in immunocompromised populations. However, this growth is tempered by price pressures from generic competition. Compound Annual Growth Rate (CAGR) projections for the broader antimalarial market, which includes atovaquone/proguanil, typically range from 3% to 5% [4]. The PCP market segment, while smaller in overall size, is expected to see stable demand.
What is the pricing and reimbursement landscape for Atovaquone?
The pricing and reimbursement landscape for atovaquone is highly segmented due to its dual indications and the significant presence of generic products.
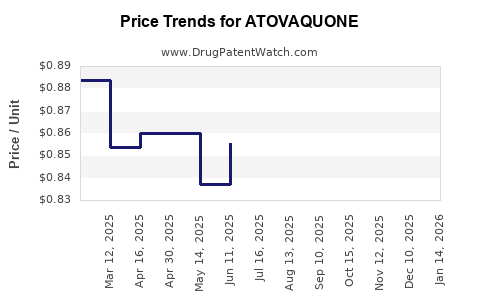
- Price Erosion from Generics: The introduction of generic atovaquone and atovaquone/proguanil has drastically reduced prices compared to originator products. For example, a course of generic atovaquone/proguanil for malaria prevention can be significantly more cost-effective than branded alternatives.
- Reimbursement Policies:
- Malaria Treatment: In many endemic countries, national health programs procure atovaquone/proguanil and other ACTs at subsidized prices or through global health initiatives. Reimbursement is often tied to formulary inclusion and public health spending priorities. In developed countries, private insurance and public health plans (e.g., Medicare, Medicaid in the US) cover atovaquone/proguanil for eligible travelers, with generic versions being the predominant choice.
- PCP Treatment/Prophylaxis: In the US, Mepron (atovaquone) and its generics are covered by Medicare, Medicaid, and private insurance for approved indications. Reimbursement levels are influenced by negotiated rates between manufacturers, pharmacy benefit managers (PBMs), and payers. The cost-effectiveness of atovaquone compared to alternatives like trimethoprim-sulfamethoxazole (TMP-SMX) is a key consideration for payers.
- Geographic Variations: Pricing and reimbursement vary significantly by region. Developed markets with robust healthcare systems and price negotiation mechanisms will have different pricing structures than low- and middle-income countries where drug procurement is often managed by governments or NGOs.
- Tender-Based Procurement: In many public health tenders for malaria medications, cost is a primary factor, favoring generic manufacturers with competitive pricing.
What are the regulatory considerations for Atovaquone?
Regulatory considerations for atovaquone are governed by national drug regulatory agencies and international health organizations.
- Food and Drug Administration (FDA) - United States:
- Atovaquone is approved for the treatment of mild to moderate uncomplicated Pneumocystis pneumonia in individuals who cannot tolerate trimethoprim-sulfamethoxazole [3].
- Atovaquone/proguanil (e.g., Malarone) is approved for the prevention of P. falciparum malaria in patients weighing 11 kg or greater [1].
- Generic approvals follow bioequivalence studies demonstrating similarity to the reference listed drug.
- European Medicines Agency (EMA) - European Union:
- Atovaquone/proguanil (e.g., Malarone) is authorized for the prevention and treatment of malaria caused by Plasmodium falciparum [5].
- Atovaquone is authorized for the treatment and prevention of PCP in certain patient populations.
- World Health Organization (WHO):
- The WHO has a prequalification program for antimalarial medicines, including atovaquone/proguanil, ensuring quality, safety, and efficacy for procurement by UN agencies and countries. This program is critical for ensuring access to essential medicines in malaria-endemic regions [2].
- WHO guidelines also address the management of PCP, recommending atovaquone as an alternative treatment option.
- Generic Approvals: Regulatory agencies require generic manufacturers to demonstrate that their product is bioequivalent to the reference product and meets stringent quality standards.
- Post-Marketing Surveillance: All approved atovaquone products are subject to post-marketing surveillance to monitor for adverse events and ensure continued safety and efficacy.
What are the therapeutic alternatives to Atovaquone?
Atovaquone faces competition from several therapeutic alternatives, both for malaria and PCP.
- For Malaria Prevention and Treatment:
- Artemisinin-based Combination Therapies (ACTs): These are the WHO-recommended first-line treatments for uncomplicated P. falciparum malaria. Examples include:
- Artemether-lumefantrine (e.g., Coartem)
- Dihydroartemisinin-piperaquine
- Artesunate-amodiaquine
- Other Antimalarials: Depending on the region and specific Plasmodium species, other antimalarials like chloroquine (resistance is widespread for P. falciparum), doxycycline, and mefloquine are also used, though often with different efficacy profiles or side effect considerations.
- For Pneumocystis jirovecii Pneumonia (PCP):
- Trimethoprim-Sulfamethoxazole (TMP-SMX): This is the preferred first-line treatment and prophylaxis for PCP due to its efficacy and lower cost. Atovaquone is typically used for patients intolerant to TMP-SMX or who have contraindications [3].
- Pentamidine: Used as an alternative for PCP treatment in patients who cannot tolerate TMP-SMX or atovaquone.
- Dapsone: Used for PCP prophylaxis in some populations.
The choice of therapy depends on the indication, drug resistance patterns, patient tolerance, contraindications, cost, and availability of alternatives.
What is the competitive landscape for Atovaquone?
The competitive landscape for atovaquone is bifurcated, reflecting its two primary indications and the impact of genericization.
- Malaria Market (Atovaquone/Proguanil):
- Dominance of Generics: The market is heavily dominated by generic atovaquone/proguanil products. Originator brands like Malarone face significant competition from lower-cost generic alternatives.
- Key Players: Major generic manufacturers (Teva, Dr. Reddy's, Sun Pharma, Cipla, Viatris) compete aggressively on price.
- Public Health Procurement: A significant portion of the market is driven by tenders from national health programs and global health organizations, where price is a critical determinant.
- Limited Differentiation: Beyond cost, differentiation is limited, with a focus on reliable supply and quality standards.
- PCP Market (Atovaquone):
- Originator (Mepron) and Generics: GlaxoSmithKline's Mepron (atovaquone) is the originator, but numerous generic atovaquone products are available.
- Competition from TMP-SMX: The primary competitive pressure comes from TMP-SMX, which is generally the first choice due to cost and efficacy. Atovaquone serves as a crucial second-line option.
- Specialty Pharmacy and Payer Influence: In developed markets, specialty pharmacies and payer formularies play a role in prescribing patterns and pricing negotiations.
- Niche Market: The PCP market is smaller in volume compared to the malaria market but represents a stable demand driven by specific patient populations.
What are the future outlook and potential challenges for Atovaquone?
The future outlook for atovaquone is characterized by continued demand in its established therapeutic areas, but with persistent challenges related to generic competition and evolving treatment paradigms.
- Continued Demand:
- Malaria: As long as malaria remains a significant public health issue in endemic regions, the need for effective and accessible treatments like atovaquone/proguanil will persist, especially in conjunction with national malaria control programs and global health initiatives.
- PCP: The ongoing prevalence of HIV/AIDS and the increasing use of immunosuppressive therapies for other conditions ensure a steady demand for PCP prophylaxis and treatment, making atovaquone a vital component of the therapeutic arsenal.
- Potential Challenges:
- Intensifying Generic Competition: The market will likely see continued price erosion as more generic players enter and existing ones compete fiercely on cost. This will limit the revenue growth potential for any manufacturer.
- Emergence of Drug Resistance: While not an immediate widespread crisis for atovaquone, the continuous evolution of drug resistance in pathogens, including Plasmodium falciparum, remains a long-term threat that could necessitate the development of novel therapies.
- Development of Novel Therapies: Research into new antimalarials and anti-infectives could eventually introduce more effective or safer alternatives, potentially impacting atovaquone's market share, particularly in first-line indications.
- Shifting Treatment Guidelines: Changes in clinical guidelines for malaria or PCP management, driven by new research or resistance patterns, could affect the role and prescription frequency of atovaquone-based therapies.
- Manufacturing and Supply Chain Robustness: Ensuring a consistent and high-quality supply of generic atovaquone is crucial, particularly for global health initiatives that rely on these essential medicines.
Key Takeaways
- Atovaquone is a critical medication for malaria and Pneumocystis pneumonia (PCP), with its market performance tied to the prevalence of these conditions.
- Original patents have expired, leading to a market dominated by generic manufacturers and significant price erosion.
- The global market is driven by malaria incidence in endemic regions and PCP occurrence in immunocompromised individuals.
- Originator products like Malarone and Mepron face substantial competition from generics, influencing revenue streams.
- Regulatory approval by agencies like the FDA, EMA, and WHO prequalification are essential for market access, especially for global health initiatives.
- Atovaquone competes with established alternatives such as ACTs for malaria and trimethoprim-sulfamethoxazole for PCP.
- The future outlook is one of stable but modest demand, constrained by intense generic competition and the potential for evolving treatment paradigms and drug resistance.
FAQs
-
Will new patents be granted for atovaquone?
While the primary composition of matter patents have expired, new patents could potentially be granted for novel formulations, drug delivery systems, or specific therapeutic combinations that demonstrate inventiveness and utility, though these are unlikely to significantly alter the generic landscape for existing products.
-
What is the approximate market share of generic atovaquone/proguanil compared to branded Malarone?
In most markets, generic atovaquone/proguanil collectively holds a dominant market share exceeding 90%, with branded Malarone's share being minimal, primarily restricted to specific geographic regions or niche patient populations.
-
How does the cost of atovaquone compare to its primary competitor for PCP, trimethoprim-sulfamethoxazole?
Generic trimethoprim-sulfamethoxazole is generally more cost-effective than generic atovaquone, which is why TMP-SMX is typically the first-line treatment for PCP. Atovaquone serves as a vital alternative when TMP-SMX is not tolerated or contraindicated.
-
Are there any emerging markets where atovaquone is seeing significant growth?
Growth for atovaquone is most pronounced in malaria-endemic regions of sub-Saharan Africa, Southeast Asia, and South America, driven by public health programs and the ongoing need for malaria prophylaxis and treatment.
-
What is the typical shelf-life and storage condition for atovaquone products?
Atovaquone products, including both single agents and combinations, generally have a shelf-life of approximately two to three years when stored at room temperature (e.g., 20°C to 25°C or 68°F to 77°F), protected from light and moisture, as specified by individual product labeling.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Malarone (atovaquone/proguanil) prescribing information. Retrieved from [Specific FDA database or manufacturer's website if accessible, otherwise general reference to FDA labeling]
[2] World Health Organization. (2023). World malaria report 2023. Retrieved from [WHO official website]
[3] U.S. Food & Drug Administration. (n.d.). Mepron (atovaquone) prescribing information. Retrieved from [Specific FDA database or manufacturer's website if accessible, otherwise general reference to FDA labeling]
[4] Market Research Report Publishers. (Various dates). Global Antimalarial Drugs Market Analysis. (Examples of typical market research firm reports used for forecasting data).
[5] European Medicines Agency. (n.d.). Malarone. Retrieved from [EMA's official website database]