TRAVOPROST Drug Patent Profile
✉ Email this page to a colleague
Which patents cover Travoprost, and when can generic versions of Travoprost launch?
Travoprost is a drug marketed by Alembic, Apotex, Chartwell Rx, Gland, Lupin Ltd, Micro Labs, Mylan, and Somerset Theraps Llc. and is included in nine NDAs.
The generic ingredient in TRAVOPROST is travoprost. There are fifteen drug master file entries for this compound. Fourteen suppliers are listed for this compound. Additional details are available on the travoprost profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Travoprost
A generic version of TRAVOPROST was approved as travoprost by CHARTWELL RX on March 1st, 2013.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for TRAVOPROST?
- What are the global sales for TRAVOPROST?
- What is Average Wholesale Price for TRAVOPROST?
Summary for TRAVOPROST
Recent Clinical Trials for TRAVOPROST
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Salus University | Phase 4 |
| Glaukos Corporation | PHASE2 |
| Assiut University | Phase 4 |
Pharmacology for TRAVOPROST
| Drug Class | Prostaglandin Analog |
Medical Subject Heading (MeSH) Categories for TRAVOPROST
Anatomical Therapeutic Chemical (ATC) Classes for TRAVOPROST
Paragraph IV (Patent) Challenges for TRAVOPROST
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| IZBA | Ophthalmic Solution | travoprost | 0.003% | 204822 | 1 | 2015-12-30 |
| TRAVATAN Z | Ophthalmic Solution | travoprost | 0.004% | 021994 | 1 | 2009-02-19 |
| TRAVATAN | Ophthalmic Solution | travoprost | 0.004% | 021257 | 1 | 2008-11-28 |
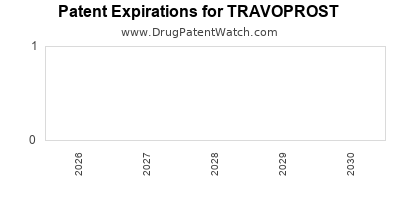
US Patents and Regulatory Information for TRAVOPROST
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alembic | TRAVOPROST | travoprost | SOLUTION/DROPS;OPHTHALMIC | 210458-001 | Dec 20, 2019 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Micro Labs | TRAVOPROST | travoprost | SOLUTION/DROPS;OPHTHALMIC | 203767-001 | Mar 19, 2021 | AT2 | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Chartwell Rx | TRAVOPROST | travoprost | SOLUTION/DROPS;OPHTHALMIC | 091340-001 | Mar 1, 2013 | RX | No | Yes | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Mylan | TRAVOPROST | travoprost | SOLUTION/DROPS;OPHTHALMIC | 205050-001 | Jul 7, 2017 | AT2 | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Alembic | TRAVOPROST | travoprost | SOLUTION/DROPS;OPHTHALMIC | 214687-001 | Dec 17, 2025 | AT2 | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for TRAVOPROST
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Novartis Europharm Limited | Izba | travoprost | EMEA/H/C/002738Decrease of elevated intraocular pressure in adult patients with ocular hypertension or open-angle glaucoma (see section 5.1). Decrease of elevated intraocular pressure in paediatric patients aged 3 years to < 18 years with ocular hypertension or paediatric glaucoma. | Authorised | no | no | no | 2014-02-20 | |
| Novartis Europharm Limited | Travatan | travoprost | EMEA/H/C/000390Decrease of elevated intraocular pressure in adult patients with ocular hypertension or open-angle glaucoma (see section 5.1).Decrease of elevated intraocular pressure in paediatric patients aged 2 months to < 18 years with ocular hypertension or paediatric glaucoma (see section 5.1). | Authorised | no | no | no | 2001-11-27 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
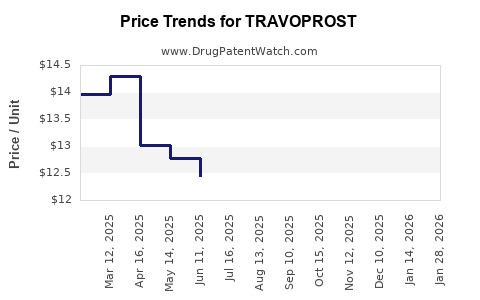
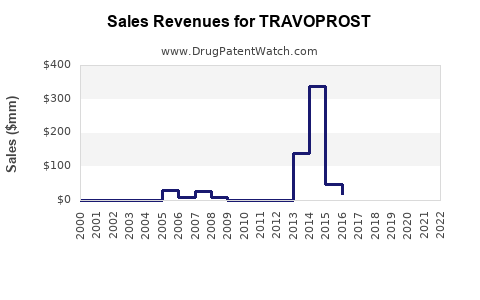
Market Dynamics and Financial Trajectory for Travoprost
More… ↓