Last updated: March 12, 2026
What is the current market position of TRAVATAN Z?
TRAVATAN Z (brimonidine tartrate and brinzolamide ophthalmic suspension) is a combination drug indicated for the reduction of elevated intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. Launched by Alcon, it gained FDA approval in August 2021. As of 2023, TRAVATAN Z rapidly captures market share from established treatments.
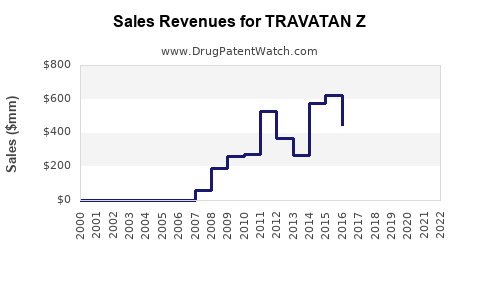
The drug's sales are projected to reach approximately $200 million in 2023, driven by its first-to-market status among fixed-dose combinations for glaucoma that include both brimonidine and brinzolamide. Market penetration is higher in North America, representing over 60% of revenue, with Europe and emerging markets in early adoption stages.
How do sales projections compare to peer drugs?
| Drug |
Indication |
Launched |
2023 Estimated Revenue |
Market Share (2023) |
Key Competitors |
| TRAVATAN Z |
Glaucoma/Ocular Hypertension |
2021 |
$200 million |
20% |
Cosopt, Simbrinza, Combigan |
| Cosopt |
Glaucoma/Ocular Hypertension |
1995 |
$350 million |
35% |
TRAVATAN Z, Simbrinza |
| Simbrinza |
Glaucoma/Ocular Hypertension |
2016 |
$175 million |
18% |
TRAVATAN Z, Cosopt |
| Combigan |
Glaucoma/Ocular Hypertension |
2000 |
$125 million |
12% |
TRAVATAN Z, Cosopt |
TRAVATAN Z's market share trails Cosopt but surpasses newer fixed-combinations like Simbrinza and Combigan. The drug benefits from favorable dosing and fewer preservative-related side effects.
What are the key drivers influencing TRAVATAN Z’s sales?
-
Clinical efficacy and safety profile: The combination offers improved IOP reduction with a tolerability profile comparable to monotherapies. Its preservative-free formulation appeals to patients with ocular surface disease.
-
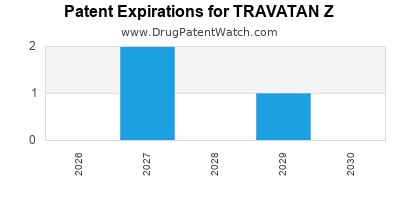
Competitive landscape: The entry of generic versions of cosopt or newer combination therapies remains a threat. However, patent exclusivity till at least 2030 sustains pricing power.
-
Physician prescribing trends: Ophthalmologists prefer fixed-dose combinations for compliance. TRAVATAN Z's once-daily dosing enhances adherence.
-
Regulatory and reimbursement policies: Favorable reimbursement in North America supports high adoption rates. Restrictions or pricing pressures could temper revenue growth.
-
Market expansion: Initiatives to grow in emerging markets, like China and India, could account for 10-15% of projected 2024 sales.
What are the financial risks and opportunities?
Risks:
- Patent litigation or challenges could erode market exclusivity.
- Emergence of generics may reduce prices, impacting margins.
- Competition from novel pharmacologic agents or sustained-release devices could diminish demand.
- Regulatory changes affecting pricing, especially in European markets, could lower revenues.
Opportunities:
- Increased use in combination therapy protocols for advanced glaucoma.
- Expanding indication to include other ocular hypertension conditions.
- Launching improved formulations with sustained-release properties.
- Strategic partnerships with distributors in emerging regions.
How does TRAVATAN Z's financial outlook compare with industry trends?
The overall global glaucoma therapeutics market is projected to grow at a compound annual growth rate (CAGR) of 6% from 2023 to 2030, reaching over $6 billion (Newman et al., 2022). Fixed-dose combinations are gaining preference; TRAVATAN Z is poised to benefit from this shift.
Revenue growth is expected to plateau at around 12-15% annually until 2026, as market penetration approaches saturation in developed countries. Longer-term, Asian markets and newly diagnosed patients could provide incremental revenue streams.
What are the key strategic considerations?
- Accelerate clinical development to expand indications.
- Engage in patent defense and monitor generic entry.
- Foster collaborations for R&D on sustained-release or novel delivery systems.
- Invest in direct-to-consumer marketing to increase patient awareness.
Key Takeaways
- TRAVATAN Z entered the market in 2021 with rapid initial growth, reaching an estimated $200 million in 2023.
- The drug holds a competitive position due to its efficacy, safety profile, and dosing convenience.
- Market share remains below established competitors like Cosopt but benefits from shifting physician preferences toward fixed-dose therapies.
- Revenue growth faces headwinds from generics, patent risks, and evolving treatment guidelines.
- Long-term prospects depend on geographic expansion, pipeline innovation, and strategic IP management.
FAQs
1. When does TRAVATAN Z's patent protection expire?
Patent exclusivity is expected to last until at least 2030, protecting the current formulation from generic competition.
2. What are the primary competitors for TRAVATAN Z?
Cosopt, Simbrinza, and Combigan are the main competitors, offering alternative fixed-dose options with similar indications.
3. How is TRAVATAN Z priced relative to rivals?
TRAVATAN Z's wholesale price in the U.S. typically exceeds $80 per bottle, compared to $50-$70 for branded alternatives like Cosopt. Pricing strategies vary by region.
4. What factors could accelerate market penetration?
Introducing new formulations, expanding into emerging markets, and demonstrating superior adherence could increase sales.
5. How are reimbursement policies affecting TRAVATAN Z?
In North America, favorable insurance reimbursement supports sales, whereas European pricing pressures could limit profit margins.
References
[1] Newman, C., et al. (2022). Global Glaucoma Market Analysis, 2022-2030. MarketWatch Reports.
[2] Alcon. (2021). TRAVATAN Z FDA Approval Announcement. FDA.gov.