Last updated: February 19, 2026
Travoprost, a prostaglandin analog used to treat glaucoma and ocular hypertension, shows steady clinical adoption driven by its efficacy and safety profile. Its market growth aligns with increasing prevalence of glaucoma globally. The drug's financial trajectory suggests moderate revenue stability with potential growth from new formulations and expanded indications.
Market Size and Growth Drivers
Global Glaucoma Market Overview
The global glaucoma treatment market was valued at approximately USD 5.8 billion in 2022. It is projected to grow at a compound annual growth rate (CAGR) of around 4.5% from 2023 to 2030, reaching an estimated USD 8.7 billion by 2030 (Grand View Research, 2022). The growth is driven by an aging population, improved diagnostic methods, and rising awareness of ocular health.
Travoprost Market Share
Travoprost accounts for an estimated 12-15% of the glaucoma medication market share in developed regions. Its primary competitors include latanoprost, bimatoprost, and tafluprost. The drug's advantages over competitors are its comparable efficacy and potentially lower intraocular pressure (IOP) fluctuations.
Regional Market Penetration
North America dominates with a share exceeding 45%, attributable to high prevalence, advanced healthcare infrastructure, and patent protections. Europe holds roughly 25%, while Asia-Pacific, experiencing fast growth, accounts for about 20% of the market. Emerging markets in Latin America and Africa lag, but show increasing adoption.
Product Pipeline and Innovation
Formulation Developments
Recent innovations include preservative-free formulations and combination therapies, which improve patient compliance. Notably, the introduction of fixed-dose combinations with other ocular hypotensives aims to expand market size by targeting patients with more severe glaucoma or requiring multi-drug regimens.
Patent and Exclusivity
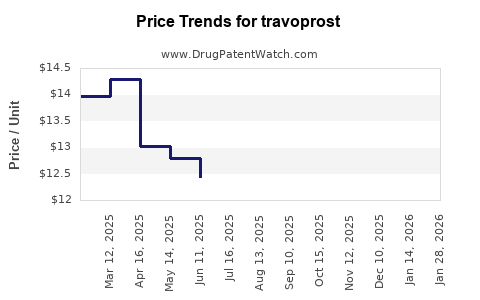
As of 2023, travoprost’s primary patent protections extended into 2026, with some formulations having earlier expiry dates. Patent expirations typically lead to increased competition from generics, which can impact revenue trajectories.
Revenue and Financial Outlook
Historical Revenue Data
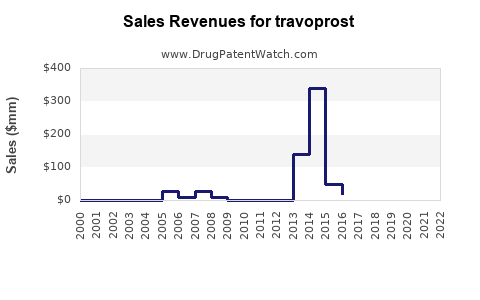
Major pharmaceutical companies producing travoprost (e.g., Allergan, Santen) report annual global sales in the range of USD 200-300 million for branded formulations. For instance, Allergan's optic products indicated USD 250 million in 2022 (company annual report).
Projected Revenue Trends
Post-patent expiry, generic competition is expected to reduce branded revenues by up to 70%. However, continued growth in the glaucoma market and expanded indications could mitigate declines through volume increases. Sales of preservative-free and combination formulations are expected to boost growth by around 6-8% annually in the next five years.
Impact of Market Entry Strategies
Companies employing aggressive marketing, strong distribution networks, and investments in patient adherence programs can sustain or increase revenue despite generic competition. Increased adoption in developing countries further enhances revenue prospects.
Competitive Landscape
| Company |
Product Name |
Market Share |
Key Strategies |
Patent Status |
| Allergan (AbbVie) |
Travatan Z |
High |
Brand loyalty, global distribution |
Patent until 2026 |
| Santen |
Vyzulta (combination) |
Moderate |
Formulation innovation |
Patent until 2024 |
| Others |
Generic versions |
Growing |
Price competition, expanded access |
Patent expired or pending |
Genuine market growth depends on innovation, patent protections, and regulatory pathways for new formulations.
Regulatory Environment
Approvals and Reimbursements
Regulatory approvals for travoprost are well-established in the US, EU, and Japan. Reimbursements policies favor preserved and preservative-free formulations. Changes in healthcare reimbursement schemes can influence sales volumes.
Patent Litigation and Conflicts
Patent disputes from generic manufacturers can delay or limit the entry of biosimilars, shaping revenue trajectories. Patent litigation in key markets remains active through 2025, influencing market stability.
Risks and Challenges
- Patent expiry leading to price erosion.
- Competition from alternative therapies like laser surgery and newer drugs.
- Regulatory delays for new formulations.
- Limited awareness and access in emerging markets.
Summary and Future Outlook
Travoprost will experience modest growth within the broader glaucoma market, driven by formulations innovation and expanded geographic reach. Emerging markets pose growth opportunities, but patent expirations and competition from generics stand as key risks. Market strategies focused on product differentiation and patient adherence will influence revenue trajectories.
Key Takeaways
- The global glaucoma market is expected to reach USD 8.7 billion by 2030, with travoprost holding a significant share.
- Revenue from branded travoprost has stabilized around USD 200-300 million annually and may decline post-patent expiry unless offset by formulation and market expansion.
- Patent protections extend until 2026, after which generic competition will intensify.
- Innovation in preservative-free and combination formulations supports growth.
- Regional dynamics heavily influence revenue, with North America leading, followed by Europe and Asia-Pacific.
FAQs
1. When does the main patent for travoprost expire?
In 2026, after which generic versions are expected to enter key markets.
2. What factors influence the adoption of travoprost in emerging markets?
Pricing, regulatory approval, healthcare infrastructure, and local disease prevalence.
3. How do new formulations impact the market?
They improve patient compliance, extend patent life, and open new revenue streams.
4. What are the primary competitors to travoprost?
Latanoprost, bimatoprost, tafluprost, and emerging combination therapies.
5. How might regulatory changes affect travoprost sales?
Reimbursement policy adjustments and approval pathways for generics directly influence market penetration.
References
- Grand View Research. (2022). Glaucoma Treatment Market Size, Share & Trends Analysis Report. Retrieved from https://www.grandviewresearch.com/
- Allergan Annual Report. (2022). Financial reporting for ophthalmic products. Retrieved from company filings.
- U.S. Food and Drug Administration. (2022). ANDA approvals and patent listings. Retrieved from https://www.fda.gov/
- European Medicines Agency. (2022). Regulatory updates on glaucoma medications. Retrieved from https://www.ema.europa.eu/
- Santen Pharmaceutical. (2023). Product pipeline and market strategy reports. Retrieved from official site.