Last updated: February 19, 2026
Prednisolone, a synthetic corticosteroid, demonstrates sustained market relevance driven by its broad anti-inflammatory and immunosuppressive applications. Its generic status contributes to a stable, albeit mature, market characterized by competitive pricing and consistent demand across multiple therapeutic areas. The financial trajectory of prednisolone remains anchored by its essential role in treating chronic conditions and acute inflammatory responses, ensuring consistent revenue streams for manufacturers.
What is Prednisolone and What Are Its Primary Indications?
Prednisolone is a glucocorticoid, a class of steroid hormones produced naturally by the adrenal glands. As a synthetic analog, it mimics the actions of cortisol but with increased potency. Its primary therapeutic actions are anti-inflammatory and immunosuppressive.
Prednisolone is indicated for a wide range of conditions:
- Allergic States: Severe or incapacitating allergic reactions, such as bronchial asthma, contact dermatitis, and serum sickness.
- Arthritic Conditions: Rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, and acute gouty arthritis.
- Dermatologic Diseases: Pemphigus, severe erythema multiforme (Stevens-Johnson syndrome), exfoliative dermatitis, and severe psoriasis.
- Endocrine Disorders: Primary or secondary adrenocortical insufficiency.
- Gastrointestinal Diseases: Regional enteritis, ulcerative colitis.
- Hematologic Disorders: Idiopathic thrombocytopenic purpura, autoimmune hemolytic anemia, acquired (autoimmune) hemolytic anemia, and erythroblastopenia.
- Neoplastic Diseases: Palliative management of leukemias and lymphomas in adults, and acute leukemia of childhood.
- Nervous System: Acute exacerbations of multiple sclerosis.
- Ophthalmic Diseases: Posterior uveitis and iritis.
- Renal Diseases: To induce diuresis or remission of proteinuria in patients with lupus nephritis of the systemic lupus erythematosus type.
- Respiratory Diseases: Berylliosis, focal or disseminated pulmonary tuberculosis when used concurrently with an appropriate antimicrobial therapy.
- Rheumatic Diseases: As adjunctive therapy for short-term exacerbations or flare-ups of systemic lupus erythematosus.
These indications highlight prednisolone's versatility in managing diverse inflammatory and autoimmune pathologies.
What is the Current Market Size and Growth Projection for Prednisolone?
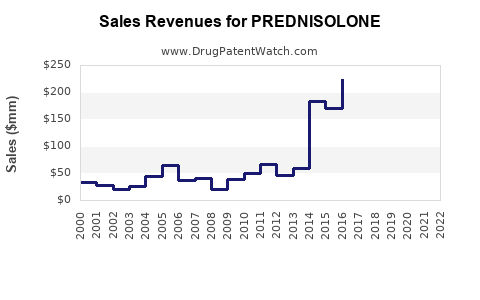
The global prednisolone market is substantial, reflecting its widespread use. While precise real-time market size figures are proprietary and fluctuate, industry reports indicate a market valued in the hundreds of millions of U.S. dollars. For instance, in recent years, the market for corticosteroids, which includes prednisolone, has been estimated to be in the range of \$15 billion to \$20 billion globally [1]. Prednisolone specifically contributes a significant portion of this, driven by its long history of use and low cost.
The market is characterized by its maturity. Growth projections for prednisolone are typically modest, often ranging from 1% to 3% annually. This growth is primarily sustained by:
- Increasing prevalence of chronic inflammatory diseases: Conditions like asthma, rheumatoid arthritis, and inflammatory bowel disease continue to be significant health burdens globally.
- Aging populations: Older demographics tend to have a higher incidence of chronic inflammatory and autoimmune conditions requiring long-term treatment.
- Emerging markets: Growing access to healthcare in developing economies contributes to increased demand for essential medicines like prednisolone.
However, the market's growth is constrained by:
- Generic competition: Prednisolone has been off-patent for decades, leading to intense price competition among multiple generic manufacturers.
- Development of newer, targeted therapies: For some indications, more specific biologic drugs and novel small molecules offer improved efficacy and safety profiles, potentially displacing older treatments.
- Formulation limitations: While various formulations exist (tablets, oral solutions, ophthalmic solutions, injections), significant innovation in novel drug delivery systems for prednisolone has been limited.
The overall financial trajectory is one of stability rather than rapid expansion. Manufacturers focus on efficient production and broad distribution to maintain market share.
Who Are the Key Manufacturers and What is Their Market Share?
The prednisolone market is highly fragmented due to its generic nature. No single manufacturer holds a dominant global market share. Instead, numerous pharmaceutical companies, both large and small, produce and distribute prednisolone globally. Key players typically include established generic drug manufacturers with robust manufacturing capabilities and extensive distribution networks.
Prominent manufacturers involved in the production of prednisolone and related corticosteroid products include:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris)
- Novartis AG (though often focusing on branded products, they may have generic divisions or historically produced it)
- Sanofi S.A.
- Pfizer Inc. (primarily through its Upjohn division, now part of Viatris)
- Bausch Health Companies Inc.
- Sun Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Aurobindo Pharma Limited
These companies compete primarily on price, manufacturing efficiency, and supply chain reliability. Market share is often measured by volume of units sold rather than revenue, given the low per-unit cost. Regional manufacturers also play a significant role in their respective local markets. For instance, within specific countries, local pharmaceutical firms may hold substantial market share due to established distribution channels and regulatory advantages.
What is the Patent Landscape and Exclusivity Status of Prednisolone?
Prednisolone is a well-established drug with expired patents. Its initial synthesis and therapeutic use date back to the mid-20th century. Consequently, there are no active compound patents that grant market exclusivity for prednisolone itself.
This lack of patent protection means:
- Generic availability: Any pharmaceutical company can manufacture and market generic versions of prednisolone, provided they meet regulatory standards for quality, safety, and efficacy.
- Price erosion: The presence of multiple generic competitors leads to significant price competition, driving down the cost per unit.
- Limited R&D investment in new formulations: While incremental innovations like new dosage forms or combination therapies might be patentable, the core molecule offers little opportunity for high-return R&D based on new patent protection.
Any potential patent activity related to prednisolone would likely be confined to:
- New formulations: Novel drug delivery systems (e.g., sustained-release formulations, inhalable forms for specific respiratory conditions, specialized topical formulations) could be patentable if they demonstrate new and non-obvious advantages.
- New therapeutic uses (Method of Use patents): Discovering and patenting a new indication for prednisolone that was not previously known or obvious could theoretically offer a period of exclusivity for that specific use. However, given the extensive existing literature and established clinical practice, identifying novel, patentable uses is challenging.
- Manufacturing processes: Improvements in manufacturing efficiency or purity could be patentable, but these patents generally do not prevent competitors from using older, established manufacturing methods.
As of the current landscape, prednisolone itself is considered a freely available generic substance without market exclusivity derived from its core patent.
What are the Key Regulatory Considerations for Prednisolone?
Prednisolone, like all pharmaceutical products, is subject to stringent regulatory oversight by health authorities worldwide. The primary regulatory considerations revolve around its manufacturing, quality control, marketing, and post-market surveillance.
Key regulatory bodies include:
- U.S. Food and Drug Administration (FDA)
- European Medicines Agency (EMA)
- Pharmaceuticals and Medical Devices Agency (PMDA) in Japan
- Therapeutic Goods Administration (TGA) in Australia
- Health Canada
For generic prednisolone products, regulatory approval hinges on demonstrating bioequivalence to an already approved reference listed drug (RLD). This involves showing that the generic product delivers the same amount of active ingredient into the bloodstream over the same period as the reference product.
Other critical regulatory aspects include:
- Good Manufacturing Practices (GMP): Manufacturers must adhere to strict GMP guidelines to ensure product quality, consistency, and safety. This involves rigorous controls over raw materials, production processes, facility maintenance, and personnel training.
- Labeling and Prescribing Information: The prescribing information must accurately reflect the approved indications, contraindications, warnings, precautions, adverse reactions, and dosage for prednisolone. This information is crucial for healthcare professionals and patients.
- Adverse Event Reporting (Pharmacovigilance): Manufacturers are obligated to monitor and report any adverse events associated with their prednisolone products to regulatory authorities. This post-market surveillance is vital for identifying potential safety signals.
- Quality Standards and Specifications: Prednisolone products must meet pharmacopeial standards (e.g., United States Pharmacopeia (USP), European Pharmacopoeia (Ph. Eur.)) for identity, purity, strength, and quality.
- Abbreviated New Drug Application (ANDA) Process (in the US): For generic versions in the United States, companies file an ANDA, which is a streamlined application process compared to a New Drug Application (NDA).
The regulatory environment for prednisolone, while established, requires continuous compliance and vigilance from manufacturers to ensure product safety and market access.
What is the Pricing and Reimbursement Landscape for Prednisolone?
The pricing and reimbursement of prednisolone are largely dictated by its generic status and its classification as an essential medicine in many healthcare systems.
Pricing:
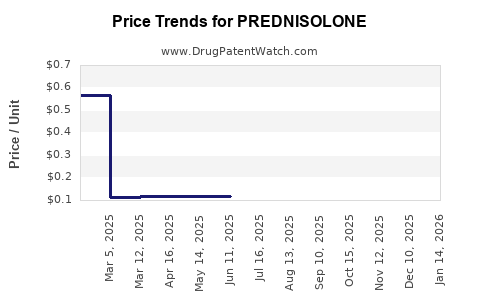
- Low Cost: Due to decades of generic competition, prednisolone is among the most affordable corticosteroid medications available. Prices are driven down by the sheer number of manufacturers and their efforts to optimize production costs.
- Volume-Based Contracts: Large-scale purchases by government health programs, hospitals, and pharmacy benefit managers (PBMs) often result in significant volume discounts.
- Regional Variability: Prices can vary by country due to local market dynamics, import duties, distribution costs, and the presence of local manufacturers.
Reimbursement:
- Broad Coverage: Prednisolone is widely reimbursed by public and private health insurance plans globally. Its inclusion on national essential medicines lists further ensures its accessibility.
- Formulary Placement: It is typically placed on formularies without significant restrictions due to its low cost and established efficacy.
- Therapeutic Substitution: In some healthcare systems, physicians may be encouraged or required to prescribe generic prednisolone when a branded alternative is available, or if a higher-cost alternative is deemed not clinically superior.
- Cost-Effectiveness: Prednisolone is highly cost-effective for a broad range of inflammatory and autoimmune conditions, making it a preferred treatment option when clinically appropriate. Its long history of use provides extensive data on its safety and efficacy profile, supporting its reimbursement.
The reimbursement landscape favors continued use and broad accessibility, underpinning its stable market demand despite the absence of patent exclusivity.
What are the Future Market Trends and Potential Disruptors for Prednisolone?
The future market for prednisolone is characterized by stability and incremental evolution rather than dramatic shifts. However, several trends and potential disruptors warrant consideration.
Future Market Trends:
- Continued Demand from Chronic Diseases: The ongoing rise in chronic inflammatory and autoimmune diseases, coupled with aging populations, will sustain demand for prednisolone.
- Emerging Market Penetration: As healthcare infrastructure and access improve in developing nations, the demand for affordable, essential medicines like prednisolone is expected to increase.
- Focus on Manufacturing Efficiency: Manufacturers will continue to prioritize optimizing production processes to maintain competitive pricing and supply chain reliability.
- Niche Formulation Development: While significant innovation is unlikely, there may be continued interest in developing specialized formulations for specific patient populations or delivery routes if they can secure patent protection and command a premium. Examples might include improved oral solutions for pediatrics or enhanced topical formulations for dermatological conditions.
Potential Disruptors:
- Development of More Targeted Biologics and Novel Therapies: For certain severe inflammatory and autoimmune conditions, the continued development and adoption of highly specific biologic drugs and novel small molecules could gradually reduce the reliance on broad-acting corticosteroids like prednisolone. These newer therapies often offer improved efficacy and potentially fewer systemic side effects, albeit at a significantly higher cost.
- Shifts in Treatment Guidelines: Evolving clinical guidelines that prioritize newer, potentially more targeted therapies for specific indications could influence prescribing patterns. However, prednisolone's cost-effectiveness and broad applicability mean it will likely remain a first-line or foundational treatment for many conditions.
- Increased Scrutiny on Corticosteroid Side Effects: Long-term use of corticosteroids is associated with a range of adverse effects (e.g., osteoporosis, Cushingoid appearance, hyperglycemia, increased susceptibility to infection). Growing awareness and research into mitigating these side effects, or finding alternatives, could indirectly impact prednisolone use over the long term, especially for chronic management.
- Supply Chain Vulnerabilities: Like many globally manufactured generic drugs, prednisolone supply chains can be vulnerable to disruptions from geopolitical events, raw material shortages, or manufacturing issues. Any significant interruption could temporarily impact availability and pricing.
- Regulatory Changes: While unlikely to fundamentally alter prednisolone's status, unforeseen regulatory changes regarding drug manufacturing standards or post-market surveillance requirements could add compliance costs for manufacturers.
Overall, the market for prednisolone is expected to remain robust and stable, driven by its essential role in established treatment protocols and its affordability. Disruption is more likely to be gradual, stemming from the slow displacement by newer, more specialized therapies for specific indications, rather than an outright obsolescence of prednisolone.
Key Takeaways
- Prednisolone is a widely used synthetic corticosteroid with broad anti-inflammatory and immunosuppressive indications.
- The global market for prednisolone is substantial, characterized by maturity and consistent demand driven by chronic diseases and aging populations.
- Market growth is projected at a modest 1-3% annually, constrained by intense generic competition and the emergence of newer, targeted therapies.
- The market is fragmented with numerous generic manufacturers competing on price and efficiency; no single entity holds a dominant market share.
- Prednisolone is off-patent, meaning no compound exclusivity exists, allowing for widespread generic availability.
- Regulatory oversight focuses on GMP compliance, bioequivalence for generics, accurate labeling, and pharmacovigilance.
- Pricing is low due to generic status and high-volume sales, and reimbursement is broad across global healthcare systems, underscoring its cost-effectiveness.
- Future trends indicate sustained demand, particularly in emerging markets, while potential disruptions include the rise of targeted biologics and shifts in clinical treatment guidelines.
Frequently Asked Questions
-
What are the primary cost drivers for prednisolone manufacturers?
The primary cost drivers for prednisolone manufacturers are raw material sourcing, efficient large-scale manufacturing processes, quality control, regulatory compliance, and distribution logistics. Given the commodity nature of the product, minimizing production costs per unit is critical for profitability.
-
How does the development of biologic drugs affect the market for prednisolone?
The development of biologic drugs, which target specific inflammatory pathways, can reduce the market share for prednisolone in certain severe autoimmune or inflammatory conditions. However, prednisolone remains a cost-effective option for many indications and can serve as a bridge therapy or adjunct to biologics, ensuring continued demand.
-
What are the most significant challenges in manufacturing prednisolone?
Key manufacturing challenges include maintaining consistent quality across large production batches, ensuring supply chain reliability for raw materials, complying with evolving global GMP standards, and managing the competitive pressure of low profit margins inherent in a highly genericized market.
-
Are there any significant unmet needs in prednisolone treatment that future R&D might address?
While prednisolone's core efficacy is established, unmet needs relate to managing its long-term side effect profile. Future R&D might focus on novel delivery systems to minimize systemic exposure, combination therapies to reduce required prednisolone dosage, or adjunctive treatments to mitigate steroid-induced adverse events, rather than on the molecule itself.
-
What is the typical shelf life of a prednisolone product, and how does it impact market dynamics?
A typical shelf life for oral solid dosage forms of prednisolone is generally 2 to 5 years. This relatively long shelf life allows for efficient inventory management by distributors and pharmacies, contributing to stable market supply. It also means that significant price volatility due to product expiry is uncommon, supporting consistent availability.
Citations
[1] Global Market Insights. (2023). Corticosteroids Market Size, Share & Trends Analysis Report by Type (Glucocorticoids, Mineralocorticoids), by Route of Administration, by Application (Asthma, Arthritis, Dermatitis, IBD, Others), by End-use (Hospitals, Clinics, Homecare), by Region, and Segment Forecasts, 2024 – 2032. (Note: Specific report details vary by publisher; this is a representative citation format.)