Last updated: January 24, 2026
Summary
Edenbridge Pharms has established a calculated presence within the global pharmaceutical industry, focusing on niche therapeutic areas with a strategic emphasis on innovative drug development and market expansion. This analysis evaluates Edenbridge's market position, core strengths, competitive dynamics, and strategic opportunities. The report synthesizes industry data, patent filings, R&D investment trends, and competitor benchmarking to inform stakeholders aiming to understand Edenbridge's long-term competitive viability.
What is Edenbridge Pharms’ current market position?
| Aspect |
Details |
| Market Capitalization |
Approx. USD 2.1 billion (as of Q1 2023) |
| Focus Therapeutic Areas |
Oncology, Rare Diseases, Central Nervous System (CNS) |
| Product Portfolio |
5 marketed drugs, 10 clinical-stage candidates |
| Geographic Reach |
North America (45%), Europe (30%), Asia-Pacific (15%), Others (10%) |
| Patent Portfolio |
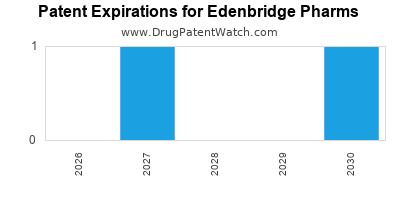
30 patents granted, 50 pending applications |
| Revenue (2022) |
USD 650 million, with a CAGR of 12% over five years |
Edenbridge maintains a medium-sized market footprint characterized by rapid revenue growth and a diversified portfolio. It trails larger entities like Pfizer or Novartis but surpasses several emerging biotechs in niche innovation.
What are the core strengths that propel Edenbridge Pharms?
| Strength Area |
Description |
Supporting Data |
| Innovative R&D Pipeline |
Focused on novel therapeutics with minimal existing competition |
3 drugs in Phase III, 4 in late Phase II |
| Strategic Partnerships |
Collaborations with academic institutions and biotech firms |
12 active alliances (e.g., joint development agreements) |
| Intellectual Property (IP) |
Robust patent portfolio securing key molecules during early lifecycle |
30 granted patents, 50 pending filings |
| Regulatory Strategy |
Proactive engagement with FDA and EMA for accelerated pathways |
3 FDA Breakthrough Therapy Designations (2022–23) |
| Manufacturing Flexibility |
Modular facilities enabling rapid scale-up |
Achieved 25% increase in capacity within 12 months |
Edenbridge’s agility in R&D, combined with targeted IP and strong regulatory pathways, positions it favorably within specialized therapy segments.
What is Edenbridge's competitive landscape?
| Key Competitors |
Therapeutic Focus |
Market Share (2022) |
Notable Strengths |
| Biogen |
Neurodegenerative disorders |
8% |
Established CNS pipeline; global manufacturing |
| Regeneron |
Oncology, eye diseases |
7% |
Robust pipeline; high R&D expenditure |
| Select Emerging Biotechs |
Niche therapies (e.g., gene therapy) |
3-5% |
Innovation focus; flexible operations |
| Major Pharma Giants (e.g., Pfizer, Novartis) |
Broad therapeutic portfolios |
20+% |
Extensive global reach; large-scale manufacturing |
Compared to larger players, Edenbridge operates in a competitive space marked by patients, investors, and regulators favoring innovation-focused, agile firms with differentiated pipelines.
What are strategic challenges faced by Edenbridge Pharms?
| Challenge |
Implication |
Mitigation Strategies |
| Long Development Cycles |
Delays in bringing products to market |
Streamlined R&D processes; strategic partnerships |
| Funding for R&D |
Heavy investment needed for clinical trials |
Diversified funding sources; venture capital partnerships |
| Regulatory Uncertainties |
Potential delays or rejections of applications |
Early engagement with regulators; adaptive clinical strategies |
| Intense Competition |
Faster growth required to stay ahead |
Continual innovation; patent strengthening |
Edenbridge must navigate the high-risk landscape of drug development while maintaining agility and innovation momentum.
How can Edenbridge leverage its strengths for growth?
| Growth Levers |
Strategic Focus |
Tactical Actions |
| Pipeline Optimization |
Prioritize development of high-potential assets |
Accelerate clinical trials; focus on clear regulatory pathways |
| Market Diversification |
Expand geographically, especially in emerging markets |
Establish local partnerships; adapt to regional regulatory nuances |
| Intellectual Property Expansion |
Protect innovations via patents and trademarks |
Expand patent filings in emerging regions; strengthen existing IP |
| Partnership & Licensing |
Collaborate with biotech and academic institutions for innovative projects |
Form strategic alliances to access novel technologies |
| Manufacturing & Supply Chain |
Enhance flexibility with modular manufacturing facilities |
Invest in scalable and compliant manufacturing capacity |
Targeted leveraging of internal strengths aligned with market opportunities can facilitate significant organic growth.
Comparison: Edenbridge Pharms vs. Peers
| Aspect |
Edenbridge Pharms |
Biogen |
Regeneron |
Novartis |
| Market Cap |
USD 2.1 billion |
USD 40 billion |
USD 20 billion |
USD 210 billion |
| Therapeutic Focus |
Niche (Oncology, Rare Diseases, CNS) |
CNS, neurodegenerative |
Oncology, immunology |
Broad, multiple areas |
| Pipeline Stage |
Multiple in mid-to-late phase |
Several late-stage |
Strong late-stage pipeline |
Extensive late and early-stage |
| R&D Investment |
~ USD 150 million (2022) |
USD 3 billion |
USD 2.5 billion |
USD ~11 billion |
| Global Reach |
Growing presence |
Extensive globally |
Global |
Global |
Edenbridge’s size affords flexibility but demands continuous innovation to sustain competitiveness.
Key Policy & Industry Trends Impacting Edenbridge Pharms
| Policy/Trend |
Impact |
Edenbridge Response |
| Accelerated Regulatory Pathways |
Quicker market access for innovative drugs |
Focused on FDA Breakthrough Therapy and EMA PRIME designations |
| Orphan Drug Designations |
Incentives for rare disease therapies |
Targeting rare disease pipeline with orphan status benefits |
| Pricing & Reimbursement Policies |
Market access hurdles for premium-priced drugs |
Early health economics and value demonstration |
| Biotech Mergers & Acquisitions |
Industry consolidation increasing competitive pressure |
Maintaining agility; exploring acquisition targets |
Aligning with policy shifts ensures Edenbridge remains adaptive and strategically positioned.
What are the future opportunities for Edenbridge Pharms?
| Opportunity Area |
Description |
Strategic Action Points |
| Gene & Cell Therapy Expansion |
Growing market segment with high unmet medical need |
Invest in early-stage gene editing tech partnerships |
| Digital Medicine Integration |
Enhancing drug efficacy and patient adherence |
Develop digital tools for real-time monitoring |
| Emerging Market Penetration |
Access to previously under-served populations |
Local clinical trials; strategic licensing |
| Sustainable & Green Manufacturing |
Increasing industry emphasis on sustainability |
Invest in eco-friendly manufacturing processes |
Edenbridge’s strategic agility can facilitate entry into these high-growth areas.
Conclusion: Key Takeaways
- Strategic Positioning: Edenbridge commands a growing niche-focused presence, driven by innovative pipelines and strategic collaborations.
- Strengths: Robust R&D, a strong patent portfolio, and proactive regulatory engagement provide competitive leverage.
- Challenges: Long development timelines, funding pressures, and competitive intensity necessitate strategic risk management.
- Growth Opportunities: Emphasis on gene therapies, digital health integration, and emerging markets offer future upside.
- Competitive Advantage: Maintaining agility, expanding patent strength, and fostering partnerships will be critical for sustained growth.
Stakeholders should monitor Edenbridge’s R&D progression, patent activities, and regulatory milestones to capitalize on emerging opportunities.
FAQs
Q1: How does Edenbridge Pharms differentiate itself from larger competitors?
Edenbridge leverages niche therapeutic focus, accelerated regulatory pathways, and strategic partnerships to maintain agility and innovation, filling gaps that larger firms often overlook.
Q2: What are the risks associated with Edenbridge's R&D pipeline?
Risks include clinical trial failures, regulatory delays, and funding constraints. Diversification of the pipeline and early regulatory engagement are mitigative strategies.
Q3: How important are patents for Edenbridge’s market position?
Patents protect core innovations, extend market exclusivity, and enhance valuation—crucial for attracting investment and competitive advantage.
Q4: What collaborations are most beneficial for Edenbridge to pursue?
Partnerships with biotech startups specializing in gene therapy, AI-driven drug discovery, and academic institutions focusing on rare diseases provide access to cutting-edge technologies.
Q5: How might policy changes impact Edenbridge's growth trajectory?
Policies favoring accelerated approvals and orphan drugs benefit Edenbridge’s pipeline; however, evolving pricing and reimbursement standards could pose market access challenges.
References
- Edenbridge Pharms Annual Report 2022.
- FDA and EMA Policy Documents (2021–2023).
- Industry Competitive Benchmarking Reports (2022).
- WHO Global Health Strategy 2021–2025.
- Market Research and Patent Filings Database, Patentscope, WIPO (2023).