OMEPRAZOLE Drug Patent Profile
✉ Email this page to a colleague
When do Omeprazole patents expire, and when can generic versions of Omeprazole launch?
Omeprazole is a drug marketed by Actavis Labs Fl Inc, Apotex, Aurobindo Pharma, Breckenridge, Dr Reddys Labs Ltd, Glenmark Pharms Ltd, Hetero Labs Ltd Iii, Impax Labs, Lannett Co Inc, Lupin Ltd, Mylan, Sandoz, Strides Pharma, Teva Pharms Usa, Xiromed, Zydus Pharms Usa Inc, Dexcel Pharma, Dr Reddys, Sun Pharm, Dexcel, Cumberland, Ajanta Pharma Ltd, Anda Repository, Aurolife Pharma Llc, Chartwell Rx, Perrigo R And D, Sciegen Pharms Inc, Zydus, Zydus Pharms, Aurobindo Pharma Ltd, L Perrigo Co, Spil, and P And L. and is included in forty-six NDAs. There are three patents protecting this drug and one Paragraph IV challenge.
This drug has thirteen patent family members in eight countries.
The generic ingredient in OMEPRAZOLE is omeprazole magnesium. There are one hundred and thirty-one drug master file entries for this compound. Forty-nine suppliers are listed for this compound. Additional details are available on the omeprazole magnesium profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Omeprazole
A generic version of OMEPRAZOLE was approved as omeprazole magnesium by DR REDDYS LABS LTD on June 5th, 2009.
Summary for OMEPRAZOLE
| International Patents: | 13 |
| US Patents: | 3 |
| Applicants: | 33 |
| NDAs: | 46 |
| Finished Product Suppliers / Packagers: | 94 |
| Raw Ingredient (Bulk) Api Vendors: | 225 |
| Clinical Trials: | 433 |
| Patent Applications: | 3,825 |
| Formulation / Manufacturing: | see details |
| Drug Prices: | Drug price information for OMEPRAZOLE |
| Drug Sales Revenues: | Drug sales revenues for OMEPRAZOLE |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for OMEPRAZOLE |
| What excipients (inactive ingredients) are in OMEPRAZOLE? | OMEPRAZOLE excipients list |
| DailyMed Link: | OMEPRAZOLE at DailyMed |
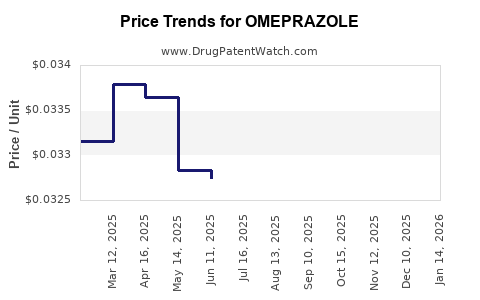
See drug prices for OMEPRAZOLE
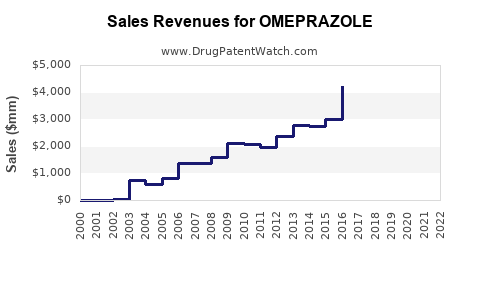
Recent Clinical Trials for OMEPRAZOLE
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Amgen | Early Phase 1 |
| Boehringer Ingelheim | N/A |
| Aya Mohamed Sadek Elsaid | Phase 3 |
Pharmacology for OMEPRAZOLE
| Drug Class | Proton Pump Inhibitor |
| Mechanism of Action | Cytochrome P450 2C19 Inhibitors Proton Pump Inhibitors |
Medical Subject Heading (MeSH) Categories for OMEPRAZOLE
Anatomical Therapeutic Chemical (ATC) Classes for OMEPRAZOLE
Paragraph IV (Patent) Challenges for OMEPRAZOLE
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| OMEPRAZOLE | Delayed-release Tablets | omeprazole | 20 mg | 022032 | 1 | 2015-06-03 |
US Patents and Regulatory Information for OMEPRAZOLE
OMEPRAZOLE is protected by three US patents.
Patents protecting OMEPRAZOLE
Stable orally disintegrating pharmaceutical compositions
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Stable orally disintegrating pharmaceutical compositions
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Stable benzimidazole formulation
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Zydus Pharms | OMEPRAZOLE AND SODIUM BICARBONATE | omeprazole; sodium bicarbonate | CAPSULE;ORAL | 203290-002 | May 25, 2018 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Teva Pharms Usa | OMEPRAZOLE | omeprazole | CAPSULE, DELAYED REL PELLETS;ORAL | 204661-002 | Jun 13, 2017 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Mylan | OMEPRAZOLE | omeprazole | CAPSULE, DELAYED REL PELLETS;ORAL | 205070-002 | Jun 29, 2018 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Dr Reddys | OMEPRAZOLE | omeprazole | TABLET, DELAYED RELEASE;ORAL | 207740-001 | Nov 5, 2018 | OTC | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Aurolife Pharma Llc | OMEPRAZOLE AND SODIUM BICARBONATE | omeprazole; sodium bicarbonate | CAPSULE;ORAL | 204922-002 | Aug 19, 2016 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Zydus Pharms Usa Inc | OMEPRAZOLE | omeprazole | CAPSULE, DELAYED REL PELLETS;ORAL | 091352-002 | Nov 19, 2012 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Sun Pharm | OMEPRAZOLE | omeprazole | TABLET, DELAYED RELEASE;ORAL | 207891-001 | Oct 12, 2018 | OTC | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
International Patents for OMEPRAZOLE
See the table below for patents covering OMEPRAZOLE around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| World Intellectual Property Organization (WIPO) | 2017216789 | ⤷ Try a Trial | |
| European Patent Office | 1187599 | FORMULATION DE BENZIMIDAZOLE STABLE (STABLE BENZIMIDAZOLE FORMULATION) | ⤷ Try a Trial |
| Israel | 147232 | STABLE BENZIMIDAZOLE FORMULATION | ⤷ Try a Trial |
| European Patent Office | 3471708 | COMPOSITIONS PHARMACEUTIQUES STABLES À DÉSINTÉGRATION ORALE (STABLE ORALLY DISINTEGRATING PHARMACEUTICAL COMPOSITIONS) | ⤷ Try a Trial |
| Australia | 5423200 | ⤷ Try a Trial | |
| World Intellectual Property Organization (WIPO) | 0078284 | ⤷ Try a Trial | |
| Canada | 2377605 | FORMULATION DE BENZIMIDAZOLE STABLE (STABLE BENZIMIDAZOLE FORMULATION) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for OMEPRAZOLE
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1411900 | 2011C/016 | Belgium | ⤷ Try a Trial | PRODUCT NAME: NAPROXENE ET ESOMEPRAZOLE (SOUS LA FORME D'ESOMEPRAZOLE MAGNESIUM TRIHYDRATE); AUTHORISATION NUMBER AND DATE: BE382505 20101214 |
| 0984957 | SPC/GB11/013 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: NAPROXEN AND ESOMEPRAZOLE; REGISTERED: UK PL 17901/0263-0001 20101105 |
| 1411900 | 2011/016 | Ireland | ⤷ Try a Trial | PRODUCT NAME: NAPROXEN AND ESOMEPRAZOLE MODIFIED-RELEASE TABLETS; NAT REGISTRATION NO/DATE: PA0970/060/001 20101221; FIRST REGISTRATION NO/DATE: PL17901/0263-0001 20101105 |
| 1411900 | SPC/GB11/015 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: NAPROXEN AND ESOMEPRAZOLE; REGISTERED: UK PL 17901/0263-0001 20101105 |
| 0984957 | 2012/048 | Ireland | ⤷ Try a Trial | PRODUCT NAME: A COMBINATION PRODUCT COMPRISING ASPIRIN AND ESOMEPRAZOLE MAGNESIUM TRIHYDRATE; NAT REGISTRATION NO/DATE: PA 970/063/001 20120831; FIRST REGISTRATION NO/DATE: 5402359; 5402367 5402375 20110812 |
| 0124495 | SPC/GB01/006 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: ESOMEPRAZOLE AS MAGNESIUM TRIHYDRATE; REGISTERED: SE 15945 20000310; SE 15946 20000310; UK PL 17901/0068-0069 20000727 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


