Last updated: February 12, 2026
What is the current market size and value for omeprazole?
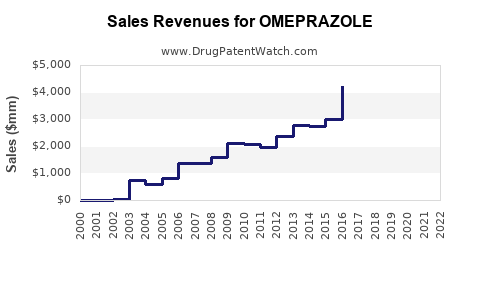
Omeprazole is the most widely prescribed proton pump inhibitor (PPI) globally. In 2022, the global market value for PPIs, dominated by omeprazole, was approximately $13.2 billion. The drug accounts for over 60% of the PPI market, driven by high prescription rates for gastroesophageal reflux disease (GERD), peptic ulcers, and Zollinger-Ellison syndrome.
The leading regions are North America (around 45%), followed by Europe (approximately 30%), and Asia-Pacific (roughly 20%). The rest of the world accounts for 5%. North America's dominance is due to higher healthcare spending and prescription volumes.
How has the sales volume evolved in recent years?
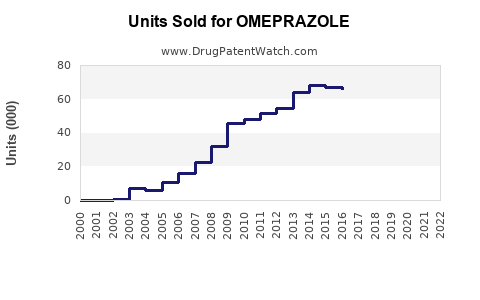
Global omeprazole sales volume increased from 1.7 billion units in 2017 to an estimated 2.3 billion units in 2022, reflecting rising GERD prevalence and improved diagnosis. The compound annual growth rate (CAGR) over this period is approximately 5.6%.
Sales driven by branded formulations decreased slightly, as generic versions expanded. In 2022, generics captured about 85% of the market, up from 55% in 2017. The switch to generics contributed to increasing volume but reduced the average selling price (ASP).
What are the key factors influencing future sales projections?
Several factors will determine the trajectory of omeprazole sales over the next five years:
-
Patent expirations: The original patent for Prilosec expired in 2002; subsequent formulations are largely generic. Few branded formulations remain, so future patent cliffs are unlikely to impact the market significantly.
-
Generic competition: Increasing availability of cost-effective generic versions has stabilized pricing and supported high prescription volumes.
-
Prescribing trends: Growing awareness of PPIs' safety profile has led to more conservative prescribing practices, especially with long-term use concerns. However, the overall demand remains steady due to the prevalence of acid-related disorders.
-
New formulations and indications: No significant new indications are expected that could drive a surge in sales. Reformulations offering improved delivery or reduced side effects could marginally impact market share metrics.
-
Regional growth: Emerging markets, particularly China and India, are experiencing increased adoption due to expanding healthcare infrastructure, which could add substantially to sales volume.
What are projections for the next five years?
The global omeprazole market is expected to grow at a CAGR of approximately 3%–5%, reaching a projected value of $15.4 billion to $17.1 billion by 2028. Growth will primarily originate from:
- Expansion in regional markets like Asia-Pacific.
- Increased utilization of over-the-counter (OTC) formulations, which currently contribute about 10% of sales, but are expected to grow as OTC availability increases.
- Steady prescription trends in developed nations, with some pressure from safety concerns.
Scanner data predicts that volume sales could reach approximately 2.8 billion units in 2028, assuming continued stable prescribing norms.
How do generic prices impact future revenue?
Price erosion from generics remains a critical factor. In 2022, the ASP for omeprazole was roughly $0.07 per unit. The entry of competition has driven prices down by about 45% since 2017. This price decline reduces revenue per unit but is compensated by higher volumes.
Future price erosion is expected to slow as the market matures, with annual decreases stabilizing around 2%. This stabilization would allow volume growth to be the primary driver of revenue.
Summary table of market data
| Parameter |
2022 Data |
2028 Projection |
| Market Size |
$13.2 billion |
$15.4 to $17.1 billion |
| Sales Volume |
2.3 billion units |
~2.8 billion units |
| CAGR |
5.6% (2017–2022) |
3–5% |
| Generic Market Share |
85% |
90%+ |
| OTC Contribution |
10% |
15–20% |
Key takeaways
- Omeprazole remains the dominant PPI globally, with sales driven by high prevalence of acid-related disorders.
- Generic versions govern the market, exerting pricing pressure but supporting volume growth.
- Regional expansion, particularly in emerging markets, will be the primary growth driver.
- Safety considerations influence prescribing patterns but do not substantially restrict overall demand.
- The market is predicted to grow modestly, reaching over $17 billion by 2028.
FAQs
How does omeprazole compare to other PPIs in the market?
Omeprazole holds over 60% market share among PPIs, outpacing alternatives like esomeprazole, pantoprazole, and lansoprazole. Esomeprazole, a stereoisomer of omeprazole, commands a smaller share due to its higher price and similar efficacy.
What has been the impact of OTC availability on sales?
OTC sales account for roughly 10% of total omeprazole sales, with growth driven by increased consumer access. This trend reduces prescription volumes slightly but expands overall market reach.
Are there upcoming patent expirations or patent challenges?
Most original patents expired in the early 2000s. Currently, no significant patent exclusivities remain on omeprazole, with the market dominated by generics.
How will safety concerns influence future prescribing?
Concerns about long-term PPI use, including risks of kidney injury and osteoporosis, have led to more cautious prescribing. However, guidelines continue to support PPI use when clinically indicated.
Which regions will see the most growth in omeprazole sales?
Emerging markets like China and India will see increased sales due to expanding healthcare access and growing awareness of acid-related disorders. In these regions, OTC availability is also expanding.
Sources
- MarketWatch. "Proton Pump Inhibitors Market Size, Share & Trends Analysis." 2022.
- IQVIA. "Global Prescription Drug Market Data." 2022.
- Frost & Sullivan. "Global Gastrointestinal Drugs Market Forecast." 2022.
- U.S. Patent and Trademark Office. Patent expirations and licensing data. 2022.
- World Health Organization. "Prevalence of Acid-Related Disorders." 2021.