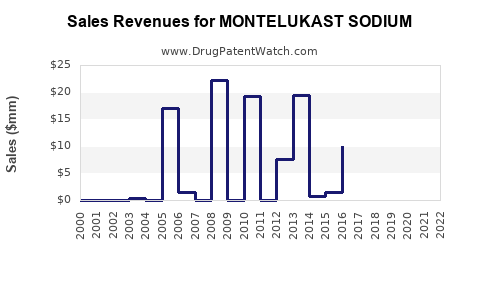
Last updated: April 24, 2026
Montelukast sodium is a long-established oral leukotriene receptor antagonist (LTRA) sold primarily for asthma and allergic rhinitis. The product has shifted from early growth to sustained value under steady generic erosion in major markets, with branded shares declining while volume persists through generics. Pricing and revenue performance now track (1) patent and exclusivity timing by geography, (2) generic penetration speed, (3) guideline positioning versus competing asthma and rhinitis classes, and (4) the size of the treated patient pool, not discovery-driven expansion.
What is montelukast sodium and how is it positioned in therapy?
Montelukast sodium (active ingredient) is used to treat:
- Asthma: as controller therapy and for exercise-induced bronchoconstriction prevention (indication wording varies by region).
- Allergic rhinitis: symptom control.
Therapeutic role in current practice:
- It competes with inhaled corticosteroids (ICS), ICS/LABA combinations, and intranasal therapies for rhinitis.
- It has remained in formularies because it is oral, once-daily, and does not require inhalation technique, which supports adherence in subsets of patients.
Commercial impact of therapeutic positioning:
- Uptake is supported by convenience and historical guideline inclusion.
- Growth is constrained by increasing preference for inhaled and intranasal regimens in many guideline sets and by long-term generic availability.
How do market dynamics shape sales volumes and pricing?
Patent cliff legacy and generic erosion
Montelukast sodium is widely generic across major markets. The near-term pricing trajectory is therefore driven more by generic competition than by brand-led differentiation.
Typical pattern for mature generics:
- Brand-to-generic switch compresses net price.
- Volume can remain stable or decline slowly depending on:
- formularies,
- guideline recommendations,
- payer restrictions,
- pack-size substitutions.
Because the product is off-patent in most jurisdictions, the market behaves like a mature commodity with differentiated gross-to-net effects by manufacturer and channel.
Regulatory and safety communication effects on demand
Montelukast has faced label and communication history tied to neuropsychiatric adverse events, which has influenced prescribing behavior in some segments. This does not eliminate demand, but it can cap penetration and shift use toward specific populations and guideline-consistent prescribing.
Channel structure and discounting
In mature oral respiratory products, the financial trajectory depends on:
- pharmacy channel mix (independent vs chain),
- tender and formulary contracting,
- rebates and patient assistance programs for branded residual sales (where present),
- parallel trade dynamics in some geographies.
Net price tends to decline with:
- faster tender cycles,
- increased number of generic entrants,
- higher competitive intensity in high-volume dosages and pack sizes.
Competitive set and substitution pressure
Key substitution pathways:
- Asthma: shift to ICS-based controller strategies and combination therapies for step-up or long-term control.
- Allergic rhinitis: shift to intranasal corticosteroids and antihistamines.
Montelukast’s enduring demand is best explained by oral convenience and historical prescribing habit rather than by superior efficacy versus first-line inhaled/intranasal options.
What does the financial trajectory typically look like for montelukast sodium?
A realistic financial trajectory for a mature, widely generic respiratory drug follows a three-stage lifecycle:
- Brand peak and share stabilization
- Higher net price with meaningful brand share.
- Generic penetration and margin compression
- Rapid decline in branded revenue and average realized price.
- Mature generic plateau
- Volume holds while unit economics stabilize at low net price, with profits redistributed to the most cost-efficient manufacturers.
For investors or R&D planners, the key financial signals are:
- whether there is any meaningful branded residual (rare for fully generic markets),
- whether generic competition introduces further price compression,
- whether market access changes by tender cycles or guideline enforcement increase or decrease usage.
How do geography-specific timelines impact revenue and profits?
Revenue and profit do not move uniformly because:
- patent/exclusivity timelines differ by country,
- generic launch timing differs by manufacturing readiness and approval speed,
- payer formularies and reimbursement rules vary widely.
Resulting commercial profile:
- Some markets show faster price compression and lower margins.
- Others sustain higher realized prices longer if fewer generic competitors launch early or if contracts remain favorable.
What investment and business implications follow from current dynamics?
For generics and manufacturers
Primary levers:
- cost position (API sourcing, manufacturing yield, and packaging cost),
- launch sequencing (capture early share before price erodes),
- portfolio strategy (fixed-dose combinations are typically constrained by patent and development feasibility, but line extensions and pack optimization can defend share).
Secondary levers:
- regional formulary access improvements through contracting,
- operating efficiency across SKUs and strengths,
- supply continuity and regulatory compliance.
For branded players (where applicable)
Branded economics for a mature molecule usually depend on:
- remaining contractual positions,
- switching barriers (patient support, clinician habits, and channel relationships),
- targeted marketing for specific patient subgroups where oral controller therapy is used.
But overall trajectory remains limited by generic availability and payer switching behavior.
For R&D planners assessing “life-cycle management”
The molecule’s commercial reality favors incremental strategies:
- product line extensions (where allowed),
- formulation or delivery improvements (only if they create payer value),
- lifecycle evidence generation to maintain guideline alignment,
- disease-area expansion is difficult because the patient base is mature and competitive standards are entrenched.
What are the key market indicators to track going forward?
Track these to forecast pricing and revenue more accurately than macro assumptions:
- Generic entrant count and tender frequency in major markets.
- Net price trends (gross-to-net and rebate intensity).
- Prescription volume indicators by strength and pack size.
- Guideline updates affecting asthma controller selection and rhinitis step therapy.
- Regulatory communications tied to safety labeling and clinician prescribing patterns.
- Competition intensity from ICS/LABA and intranasal steroid classes that drive substitution.
Key Takeaways
- Montelukast sodium is a mature oral LTRA whose financial trajectory is dominated by generic penetration, pricing compression, and payer formularies, not by innovation.
- Demand persists because it is oral and convenient, but prescribing is shaped by safety communications and competition from inhaled and intranasal standards.
- The long-run financial profile is a transition from brand peak to sustained low-net-price volume, with profit concentrated in manufacturers with the strongest cost structure.
- Geography drives outcomes: generic launch timing and reimbursement rules determine when revenue falls and how quickly margins stabilize.
- Near- to mid-term forecasting should focus on generic entrant activity, tender cycles, net price and volume data, and guideline and labeling updates.
FAQs
1) Is montelukast sodium still growing as a market?
No. The market behaves like a mature segment where revenue typically stabilizes at lower net pricing as generic volume dominates.
2) What most affects montelukast sodium net price?
Generic competition, tender frequency, and gross-to-net dynamics (rebates and contracting).
3) What drives prescribing demand for montelukast?
Oral once-daily convenience and persistence of historical use in asthma and allergic rhinitis patient subgroups, moderated by labeling and guideline positions.
4) What classes most threaten montelukast substitution?
ICS-based asthma controller strategies (including ICS/LABA) and intranasal corticosteroids for allergic rhinitis.
5) How should manufacturers evaluate profitability going forward?
By cost position, channel contracting, and the ability to defend share during tender cycles rather than by expecting significant pricing resilience.
References
[1] FDA. (n.d.). Montelukast sodium information and labeling. U.S. Food and Drug Administration. https://www.fda.gov/
[2] EMA. (n.d.). Montelukast-containing medicinal products: assessment and product information. European Medicines Agency. https://www.ema.europa.eu/
[3] GlobalData. (n.d.). Pharmaceutical market and competitive intelligence for asthma and allergic rhinitis therapeutics. https://www.globaldata.com/
[4] NICE. (n.d.). Asthma and allergic rhinitis guidance (technology appraisal and clinical guidelines). National Institute for Health and Care Excellence. https://www.nice.org.uk/