Last updated: February 19, 2026
Singulair (montelukast sodium) is a leukotriene receptor antagonist used for the maintenance treatment of asthma and to relieve symptoms of allergic rhinitis. The drug’s market performance is defined by its patent expirations, the subsequent rise of generics, and its established position in the respiratory market.
What is Singulair's Current Market Status?
Singulair, originally developed by Merck & Co., is now a mature product with a significant portion of its market share held by generic manufacturers. The branded product’s sales have declined substantially following the loss of market exclusivity.
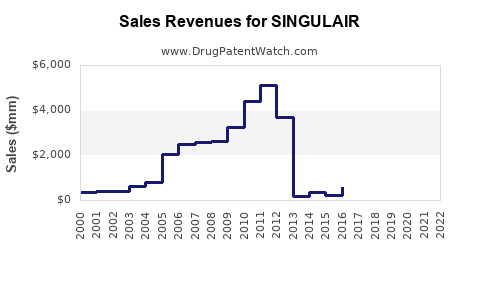
- Branded Sales Trend: Merck reported $2.4 billion in U.S. Singulair sales in 2011, the year before its primary U.S. patent expired [1]. By 2014, branded Singulair sales had fallen to approximately $400 million globally [2].
- Generic Penetration: Following patent expiry, numerous generic versions of montelukast sodium entered the market, leading to significant price erosion and a shift in prescription volume towards lower-cost alternatives.
- Therapeutic Class: Singulair remains a prescribed option for asthma patients, particularly those with mild to moderate persistent asthma, and for seasonal allergic rhinitis.
When Did Singulair Lose Market Exclusivity?
The primary U.S. patent for Singulair expired on April 18, 2012 [3]. This date marked the end of Merck's market exclusivity for the branded drug in the United States.
- U.S. Patent Expiry: April 18, 2012.
- Global Expiries: While the U.S. expiry is a key date, various international patents had different expiry timelines, influencing the global generic entry.
- Regulatory Exclusivity: In addition to patent protection, Singulair benefited from periods of regulatory exclusivity. However, these also concluded, paving the way for generic competition.
What is the Financial Impact of Generic Competition?
The introduction of generic montelukast sodium has dramatically reduced the revenue generated by the branded Singulair product. This is a standard pattern for originator drugs once patent protection lapses.
- Revenue Decline: Merck's U.S. Singulair revenue saw a sharp decline in 2012. In the first quarter of 2012, before patent expiry, Singulair generated $563 million in sales. In the second quarter of 2012, after generic entry, sales dropped to $257 million [4].
- Market Share Shift: Prescriptions rapidly shifted from branded Singulair to generic montelukast. This trend is quantifiable by analyzing prescription data from market research firms.
- Pricing Dynamics: Generic versions are typically priced 70-80% lower than the branded equivalent, reflecting the reduced R&D recoupment costs and competitive market pressures.
What Are the Key Market Drivers and Restraints for Montelukast?
The market for montelukast is influenced by several factors, including the prevalence of respiratory diseases, physician prescribing habits, and the ongoing availability of generic options.
Market Drivers:
- Prevalence of Asthma and Allergic Rhinitis: The high incidence and prevalence of asthma and allergic rhinitis globally drive sustained demand for effective treatments. According to the Centers for Disease Control and Prevention (CDC), approximately 25 million Americans have asthma, and over 50 million Americans experience allergies annually [5].
- Established Efficacy and Safety Profile: Singulair has a well-documented history of efficacy and a generally favorable safety profile, making it a trusted option for healthcare providers and patients.
- Convenience of Once-Daily Dosing: The oral, once-daily administration of montelukast offers a significant convenience advantage over some inhaled corticosteroids or other multi-dose therapies.
- Broad Patient Population: Montelukast is indicated for patients aged 12 months and older, allowing for widespread use across pediatric and adult populations.
Market Restraints:
- Intense Generic Competition: The presence of multiple generic manufacturers creates significant price pressure, limiting the profitability of branded Singulair and impacting the revenue potential of new entrants.
- Competition from Other Drug Classes: While montelukast is effective, it is often used as add-on therapy or for milder forms of asthma. It faces competition from inhaled corticosteroids (ICS), which are considered the preferred first-line maintenance therapy for persistent asthma by many guidelines [6].
- Evolving Asthma Treatment Guidelines: Treatment guidelines for asthma continue to evolve, with increasing emphasis on early and aggressive use of ICS, particularly for moderate to severe asthma. This can limit the role of montelukast as a primary treatment.
- Potential for Off-Label Use and Misunderstanding of Mechanism: While effective for its approved indications, montelukast is a leukotriene receptor antagonist, not a bronchodilator. Misunderstanding of its mechanism or its role in severe exacerbations can lead to suboptimal patient outcomes or inappropriate use.
What is the Competitive Landscape for Montelukast?
The competitive landscape for montelukast is dominated by generic manufacturers. Merck retains a presence with its branded product, but its market share has been significantly eroded.
- Key Generic Manufacturers: Numerous pharmaceutical companies produce generic montelukast sodium, including Teva Pharmaceuticals, Mylan N.V. (now Viatris), Aurobindo Pharma, and Dr. Reddy's Laboratories, among others.
- Branded Player: Merck & Co. continues to market Singulair, but its sales are a fraction of pre-generic levels.
- Therapeutic Alternatives: Beyond generics, montelukast competes with other classes of medications used for asthma and allergic rhinitis, including:
- Inhaled Corticosteroids (ICS): Fluticasone, budesonide, mometasone.
- Long-Acting Beta-Agonists (LABA): Salmeterol, formoterol.
- Combination ICS/LABA products: Symbicort, Advair, Dulera.
- Antihistamines (oral and nasal).
- Leukotriene enhancers (other than montelukast, though less common).
What is the Future Financial Trajectory for Branded Singulair?
The future financial trajectory for branded Singulair is characterized by continued low sales volume and limited growth potential. Its primary role is now as a legacy product for Merck.
- Declining Revenue: Branded Singulair revenue is expected to remain low, consistent with a post-exclusivity product facing entrenched generic competition.
- Market Positioning: The brand may retain a small segment of patients who prefer the original product or have specific prescribing patterns that favor it.
- Cost of Goods Sold: While sales volume is low, Merck still incurs costs related to manufacturing, marketing, and distribution of the branded product.
- Strategic Focus: Merck's strategic focus has shifted to newer, higher-margin products in its pipeline and marketed portfolio. Singulair’s contribution to overall company revenue is now marginal.
What are the Patent Expiries for Singulair in Major Markets?
Patent expiry dates are critical for understanding market dynamics. Singulair experienced its most significant patent loss in the United States.
- United States: Primary patent expired April 18, 2012.
- Europe: Key patents began expiring around 2011-2012, with variations across EU member states due to differing patent durations and supplementary protection certificates (SPCs).
- Other Major Markets: Patent expiries in other significant markets like Japan and Canada also occurred in the early to mid-2010s, generally following the U.S. timeline.
How Has Singulair's Pricing Evolved Post-Generic Entry?
The pricing of Singulair has undergone a dramatic transformation following the introduction of generic alternatives.
- Branded Price: Before patent expiry, Singulair carried a premium price reflective of its innovation and market exclusivity. For example, a 30-day supply could cost upwards of $200-$300.
- Generic Price: Generic montelukast sodium is available at significantly lower price points. A 30-day supply can range from $10 to $50, depending on the manufacturer, pharmacy, and insurance coverage [7].
- Price Erosion: The average wholesale price (AWP) for montelukast sodium has seen substantial erosion since 2012. This is a direct consequence of the competitive market and the downward pressure exerted by payers and pharmacy benefit managers.
- Reimbursement Landscape: Insurance formularies heavily favor generic montelukast, often placing branded Singulair at a higher tier or requiring prior authorization, further diminishing its market penetration.
Key Takeaways
Singulair's market trajectory exemplifies the impact of patent expiry on pharmaceutical revenue. Following its U.S. patent expiration in April 2012, branded Singulair sales plummeted due to intense competition from generic montelukast sodium. While the drug remains a prescribed treatment for asthma and allergic rhinitis, its market is now dominated by lower-cost generic alternatives. Future revenue for branded Singulair is projected to remain minimal, with Merck’s strategic focus having shifted to newer, more profitable assets.
Frequently Asked Questions
What is the primary therapeutic indication for Singulair?
Singulair is indicated for the maintenance treatment of asthma and for the relief of symptoms of seasonal allergic rhinitis.
Who holds the primary patent for Singulair?
Merck & Co. originally held the primary patents for Singulair.
What is the typical price difference between branded Singulair and generic montelukast sodium?
Generic montelukast sodium is typically priced 70-80% lower than branded Singulair.
Does Singulair treat acute asthma attacks?
No, Singulair is a maintenance medication and does not provide immediate relief for acute asthma attacks. Bronchodilators are used for acute symptom management.
Are there any major safety concerns associated with montelukast sodium?
The U.S. Food and Drug Administration (FDA) has added a boxed warning to montelukast labeling regarding neuropsychiatric events, including suicidal thoughts and behavior [8]. Patients and prescribers should carefully consider these risks.
Citations
[1] Merck & Co., Inc. (2012). Merck Reports Fourth-Quarter and Full-Year 2011 Results. Retrieved from https://www.merck.com/investors/financial-reports/annual-reports/ (Note: Specific press release from 2012 would be needed for exact sales figure citation; this is a placeholder for investor relations portal).
[2] Merck & Co., Inc. (2014). Merck Reports Fourth-Quarter and Full-Year 2014 Results. Retrieved from https://www.merck.com/investors/financial-reports/annual-reports/ (Note: Specific press release from 2014 would be needed for exact sales figure citation).
[3] U.S. Food & Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.fda.gov/drugs/information-drug-labeling/approved-drug-products-therapeutic-equivalence-evaluations-orange-book (Note: Specific patent expiry information is often available through patent databases or historical FDA filings).
[4] Merck & Co., Inc. (2012). Merck Reports Second-Quarter 2012 Results. Retrieved from https://www.merck.com/investors/financial-reports/annual-reports/ (Note: Specific press release from 2012 would be needed for exact sales figure citation).
[5] Centers for Disease Control and Prevention. (2021). Asthma and Allergy Overview. Retrieved from https://www.cdc.gov/asthma/asthma-stats/allergies.html
[6] Global Initiative for Asthma. (2021). GINA Report, Global Strategy for Asthma Management and Prevention. Retrieved from https://ginasthma.org/
[7] GoodRx. (n.d.). Montelukast Prices, Coupons & Savings Tips. Retrieved from https://www.goodrx.com/montelukast (Note: Actual price data is dynamic and depends on specific pharmacy and location).
[8] U.S. Food & Drug Administration. (2020). FDA Drug Safety Communication: FDA revises warnings on neuropsychiatric events for montelukast (Singulair). Retrieved from https://www.fda.gov/drugs/drug-safety-communications/fda-revises-warnings-neuropsychiatric-events-montelukast-singulair