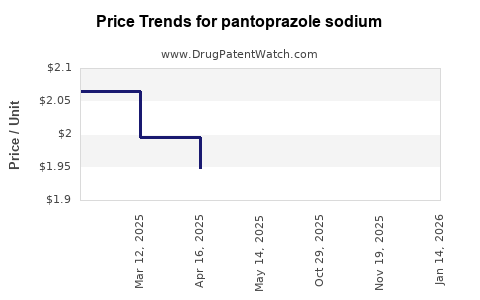
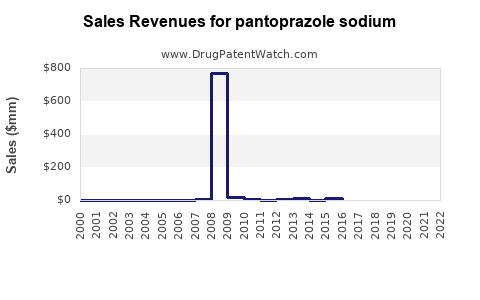
Last updated: February 19, 2026
What is the current market size and growth rate for pantoprazole sodium?
The global proton pump inhibitor (PPI) market, which includes pantoprazole sodium, was valued at approximately USD 13.8 billion in 2021. It is projected to grow at a compound annual growth rate (CAGR) of 4.2% from 2022 to 2028. Pantoprazole sodium accounts for roughly 8-10% of this market, driven by its use in GERD, Zollinger-Ellison syndrome, and erosive esophagitis.
How competitive is the pantoprazole sodium market?
Major players include:
- Pfizer Inc. ("Protonix")
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V.
- Sandoz (Novartis)
- Aurobindo Pharma
Pfizer's Protonix remains the leading formulation. Patent expirations for brand-name versions in the U.S. occurred in 2014, opening the market to generics. Generics dominate sales, representing approximately 60% of total pantoprazole sodium revenue globally.
What are the patents and regulatory barriers affecting market entry?
Pfizer's original patents on pantoprazole sodium expired in 2014 in the U.S., but certain formulations gained extended exclusivity through secondary patents and formulation-specific claims until 2020. The expiry of patents in key markets like the U.S. and Europe facilitated generics' entry.
Regulatory approval processes are generally straightforward due to established biosimilar pathways. However, patent litigations may delay generic market entry in some jurisdictions.
What trends influence the financial trajectory?
- Increasing prevalence of GERD and related disorders: Global GERD prevalence is approximately 20%, driving sustained demand.
- Patent expirations: Expected manufactured generic proliferation post-2014, matching sales declines of proprietary brands.
- Price erosion: Entry of generics has reduced average selling prices (ASPs) by 30-50%, affecting revenue.
- Rise of OTC formulations: Moving from prescription-only to OTC status in some regions expands market size, but pressures prices.
- Manufacturing changes: Companies adopting cost-effective APIs and formulations reduce production costs and improve margins.
How do supply chain and manufacturing factors impact the outlook?
Supply chain disruptions, such as raw material shortages (e.g., omeprazole intermediates), can impact production volumes. Companies investing in vertical integration mitigate some risks. API cost reductions from process innovations support competitiveness.
Financial projections: Revenue and profit prospects
| Year |
Estimated Global Sales (USD billions) |
Key Drivers |
Comments |
| 2022 |
2.5 – 2.7 |
Generics market share expansion |
Market stabilization after patent expiry |
| 2024 |
2.3 – 2.5 |
Price erosion, competitive intensification |
Margin impact from pricing pressures |
| 2026 |
2.0 – 2.2 |
Market saturation, OTC shift |
Revenue plateau unless new indications emerge |
Profitability depends heavily on manufacturing efficiencies and patent litigation outcomes. Companies that innovate or diversify formulation offerings may see improved margins.
Summary of key market drivers and risks
- Market drivers: Rising GERD prevalence, patent expirations, OTC availability.
- Risks: Price erosion, regulatory delays, raw material shortages, increased competition.
Key Takeaways
- The pantoprazole sodium market is mature, with significant generic penetration reducing prices.
- Estimated global sales hover around USD 2.5 billion, with slow growth projected post-2024.
- Patent expirations and OTC conversions will influence supply dynamics.
- Supply chain stability and production innovation are critical for maintaining margins.
- Emerging markets and new indications may present growth opportunities despite market saturation.
FAQs
Q1: How does patent expiry affect pantoprazole sodium's market?
A1: Patent expiry opens markets to generics, increases competition, and drives down prices, leading to revenue declines for branded formulations.
Q2: What are the main drivers for future growth?
A2: Rising prevalence of acid-related disorders and expansion into OTC segments are primary growth drivers.
Q3: How are generic manufacturers impacting the market?
A3: They provide lower-cost alternatives, capturing a majority share of sales, and exert downward pressure on ASPs.
Q4: Are there new indications or formulations expected to influence market size?
A4: Few new indications are projected; however, formulations with improved delivery or combination therapies might offer niche growth.
Q5: What are the primary risks facing companies in this market?
A5: Price erosion due to generics, supply chain disruptions, and regulatory hurdles are significant risks.
References
- Grand View Research. (2022). Proton Pump Inhibitors Market Size, Share & Trends Analysis Report. Retrieved from https://www.grandviewresearch.com
- U.S. Food and Drug Administration. (2022). Patent Status of Proton Pump Inhibitors. Retrieved from https://www.fda.gov
- Statista. (2022). GERD prevalence worldwide. Retrieved from https://www.statista.com