Share This Page
Drug Sales Trends for pantoprazole sodium
✉ Email this page to a colleague
Payment Methods and Pharmacy Types for pantoprazole sodium (2022)
Revenues by Pharmacy Type
Units Sold by Pharmacy Type
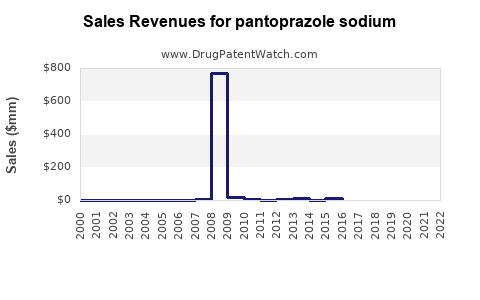
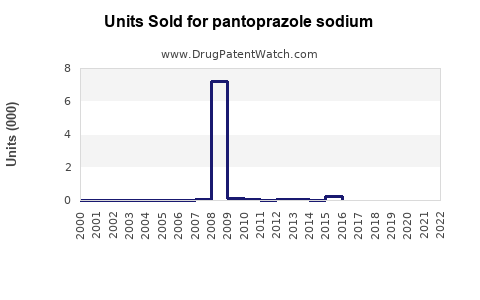
Annual Sales Revenues and Units Sold for pantoprazole sodium
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| PANTOPRAZOLE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| PANTOPRAZOLE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| PANTOPRAZOLE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| PANTOPRAZOLE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| PANTOPRAZOLE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| PANTOPRAZOLE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| PANTOPRAZOLE SODIUM | ⤷ Start Trial | ⤷ Start Trial | 2016 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Pantoprazole Sodium: Market Analysis and Sales Projections
Pantoprazole sodium, a proton pump inhibitor (PPI) used to treat conditions associated with excessive stomach acid, demonstrates a stable market performance driven by its established efficacy and continued demand for gastrointestinal (GI) treatments. Generic competition significantly shapes its market dynamics, impacting pricing and market share distribution.
What is the current market size and growth trajectory for pantoprazole sodium?
The global market for pantoprazole sodium is substantial and projected to experience moderate growth. In 2023, the market was valued at approximately $2.5 billion. Projections indicate a compound annual growth rate (CAGR) of 3.5% from 2024 to 2030, with an estimated market value of $3.1 billion by the end of the forecast period. This growth is primarily attributed to the persistent prevalence of GERD, peptic ulcers, and Zollinger-Ellison syndrome, coupled with an aging global population, which is often more susceptible to these conditions. The increasing accessibility of generic pantoprazole sodium also contributes to its broad market penetration.
Who are the key players and what is their market share in the pantoprazole sodium market?
The pantoprazole sodium market is characterized by a significant number of generic manufacturers, leading to a fragmented competitive landscape. While originator brands still hold a portion of the market, generic versions dominate in terms of volume and revenue due to their lower price points.
Key Manufacturers (Global Market):
- Teva Pharmaceutical Industries Ltd.: A leading generic pharmaceutical company with a strong presence in pantoprazole sodium manufacturing and distribution.
- Mylan N.V. (now Viatris): Another major player in the generics market, offering a wide range of pantoprazole sodium formulations.
- Dr. Reddy's Laboratories Ltd.: Holds a significant market share, particularly in emerging markets, with its pantoprazole sodium offerings.
- Sun Pharmaceutical Industries Ltd.: A prominent Indian pharmaceutical company with a substantial portfolio of generic drugs, including pantoprazole sodium.
- Aarti Drugs Ltd.: A key supplier of the active pharmaceutical ingredient (API) for pantoprazole sodium and also manufactures finished dosage forms.
- Lupin Ltd.: Has a considerable footprint in the global generics market, including pantoprazole sodium products.
- Hikma Pharmaceuticals PLC: A diversified pharmaceutical company with a strong presence in the US and MENA regions, offering pantoprazole sodium generics.
The market share distribution is dynamic and fluctuates based on regional sales, regulatory approvals, and pricing strategies. However, Teva and Viatris consistently rank among the top global suppliers of generic pantoprazole sodium.
What are the primary drivers and restraints for pantoprazole sodium sales?
Market Drivers:
- Increasing Prevalence of Gastrointestinal Disorders: The rising incidence of GERD, peptic ulcers, and other acid-related disorders globally is a primary driver. Factors contributing to this include dietary changes, stress, and an increasing aging population.
- Growth in Emerging Markets: Expanding healthcare infrastructure, rising disposable incomes, and increasing awareness of GI health in emerging economies like Asia-Pacific and Latin America are creating new avenues for market growth.
- Availability of Generic Versions: The patent expiration of the originator drug has led to the widespread availability of affordable generic pantoprazole sodium, increasing accessibility and driving volume sales.
- Growing Awareness and Diagnosis: Improved diagnostic tools and greater public awareness regarding GI health contribute to higher rates of diagnosis and subsequent prescription of PPIs like pantoprazole sodium.
- Supportive Regulatory Environments: In many regions, regulatory bodies facilitate the approval and market entry of generic drugs, further bolstering the market for pantoprazole sodium.
Market Restraints:
- Intense Price Competition: The high number of generic manufacturers results in intense price competition, which can depress profit margins for individual companies.
- Development of Newer Therapies: While PPIs remain a cornerstone of treatment, ongoing research into alternative or adjunctive therapies for GI disorders could eventually impact the long-term market share of pantoprazole sodium.
- Side Effect Concerns and Long-Term Use Limitations: While generally safe, prolonged use of PPIs, including pantoprazole sodium, has been associated with potential side effects (e.g., vitamin B12 deficiency, increased risk of fractures, kidney issues), which may lead some prescribers and patients to seek alternatives or limit duration of therapy.
- Reimbursement Policies: Fluctuations in healthcare reimbursement policies in different countries can affect the affordability and prescription patterns of pantoprazole sodium.
What is the competitive landscape for pantoprazole sodium, and how does it differ by region?
The competitive landscape for pantoprazole sodium is largely defined by the presence of numerous generic manufacturers, leading to a highly competitive environment globally. However, regional nuances exist concerning market penetration, pricing, and the influence of specific local players.
Global Competitive Landscape:
- Dominance of Generics: The market is overwhelmingly dominated by generic manufacturers. The originator brand's market share has significantly diminished since patent expiry.
- API Suppliers: A robust API supply chain exists, with several chemical manufacturers specializing in pantoprazole sodium synthesis. These API suppliers often cater to multiple finished dosage form manufacturers.
- Formulation Variations: While the primary API remains consistent, variations in tablet strength (e.g., 20 mg, 40 mg) and formulation (e.g., delayed-release tablets, intravenous solutions) exist, catering to different patient needs and prescribing preferences.
Regional Competitive Dynamics:
- North America (USA & Canada): This market is characterized by a high volume of generic prescriptions. Pricing is highly competitive, driven by PBMs (Pharmacy Benefit Managers) and large retail pharmacy chains. Teva, Viatris, and other major generic players hold significant market share. Brand loyalty to the originator (Protonix) has largely eroded.
- Europe: Similar to North America, Europe features a strong generic presence. National health systems and tender processes play a significant role in procurement and pricing. Local European manufacturers also compete alongside global generic giants. Germany, the UK, and France are key markets with substantial pantoprazole sodium consumption.
- Asia-Pacific: This region presents a dual dynamic. While generics are prevalent and driving volume, there is also a growing demand for branded generics and potentially higher-margin formulations in more developed economies within the region (e.g., Japan, South Korea). India and China are major manufacturing hubs for both API and finished products, serving both domestic and export markets. Local players in these countries hold considerable influence.
- Latin America: The market is growing, with generics being the primary offering. Pricing sensitivity is a key factor. Local manufacturers are emerging, but global generic companies maintain a strong presence. Brazil and Mexico are significant markets.
- Middle East & Africa: This region is characterized by a growing demand for affordable healthcare. Generic pantoprazole sodium is widely adopted. Distribution networks and partnerships with local entities are crucial for market access.
Key Competitive Strategies:
- Cost Leadership: Companies focus on optimizing manufacturing processes and supply chains to offer the lowest possible prices for generic pantoprazole sodium.
- Portfolio Expansion: Manufacturers aim to offer a comprehensive range of pantoprazole sodium dosage forms and strengths to capture diverse market segments.
- Geographic Expansion: Companies seek to expand their market reach into emerging economies with growing healthcare needs.
- Partnerships and Alliances: Strategic collaborations with distributors, pharmacy chains, and healthcare providers are common for market penetration.
What are the projected sales for pantoprazole sodium over the next five years?
Projected sales for pantoprazole sodium are based on historical trends, market drivers, and competitive pressures. The market is expected to maintain a steady, albeit moderate, growth rate.
| Year | Estimated Global Market Value (USD Billion) | CAGR (2024-2025) |
|---|---|---|
| 2024 | 2.59 | 3.6% |
| 2025 | 2.68 | 3.5% |
| 2026 | 2.78 | 3.4% |
| 2027 | 2.87 | 3.3% |
| 2028 | 2.97 | 3.2% |
| 2029 | 3.07 | 3.1% |
| 2030 | 3.17 | 3.0% |
Note: Projections are estimates and subject to market fluctuations, regulatory changes, and unforeseen economic events.
These figures reflect the aggregate sales of all manufacturers globally. Individual company performance will vary based on market share, product differentiation (where applicable in generics), and strategic execution. The slight deceleration in CAGR towards the end of the forecast period reflects the maturing nature of the generic market and the increasing impact of price erosion.
What are the key patent landscapes and regulatory considerations for pantoprazole sodium?
The patent landscape for pantoprazole sodium is largely characterized by the expiry of its primary composition of matter patents. This has paved the way for widespread generic entry.
Patent Landscape:
- Composition of Matter Patents: The core patents protecting the chemical entity of pantoprazole sodium have expired in major markets like the United States and Europe. For example, the key US patent for pantoprazole expired in the early 2000s [1].
- Formulation and Method of Use Patents: While the primary patents have expired, some companies may hold secondary patents related to specific formulations (e.g., improved stability, novel delivery systems) or methods of use for pantoprazole sodium. However, these are less impactful than composition of matter patents in enabling broad market entry.
- Generic Patent Challenges: The period after patent expiry often sees ongoing litigation as generic manufacturers challenge any remaining secondary patents to gain market access or clear the path for their products.
- Exclusivity Periods: In some regions, regulatory authorities grant a period of market exclusivity for new drug applications (NDAs) or for specific indications, which can briefly delay generic competition for certain branded products, though this is less relevant for established generics like pantoprazole.
Regulatory Considerations:
- ANDA and MAA Filings: Generic manufacturers must submit Abbreviated New Drug Applications (ANDAs) in the US or Marketing Authorisation Applications (MAAs) in Europe, demonstrating bioequivalence to the reference listed drug (RLD) [2].
- Good Manufacturing Practices (GMP): All manufacturing facilities producing pantoprazole sodium, whether API or finished dosage forms, must adhere to strict GMP guidelines set by regulatory bodies like the FDA and EMA to ensure product quality, safety, and efficacy.
- Pharmacopeial Standards: Pantoprazole sodium must meet the specifications outlined in official pharmacopeias, such as the United States Pharmacopeia (USP) and the European Pharmacopoeia (Ph. Eur.), regarding purity, identity, and potency.
- Labeling Requirements: Regulatory bodies mandate specific labeling information for pantoprazole sodium products, including indications, dosage, contraindications, warnings, precautions, and adverse reactions, aligning with the RLD's approved labeling.
- Post-Market Surveillance: Manufacturers are required to engage in post-market surveillance to monitor for any emerging safety concerns or adverse events associated with pantoprazole sodium use in the general population.
- Environmental Regulations: Manufacturing processes must comply with environmental regulations related to waste disposal, emissions, and chemical handling.
The regulatory pathway for generics is well-established, allowing for efficient market entry once bioequivalence is demonstrated and manufacturing standards are met. The primary regulatory focus for pantoprazole sodium now centers on quality control, GMP compliance, and post-market safety monitoring.
What are the future trends and innovations impacting the pantoprazole sodium market?
The pantoprazole sodium market, while mature, is subject to evolving trends in healthcare and pharmaceutical development.
Future Trends:
- Focus on Combination Therapies: Research may explore combining pantoprazole sodium with other agents to enhance treatment efficacy for complex GI conditions or to address specific unmet needs, though this is more likely to drive new product development rather than significantly alter the core pantoprazole market.
- Advancements in Drug Delivery: While delayed-release tablets are standard, ongoing innovation in drug delivery could lead to novel formulations offering improved patient compliance, faster onset of action, or more sustained release profiles.
- Personalized Medicine Approaches: As understanding of individual patient responses to PPIs grows, there may be a trend towards more personalized treatment regimens, potentially influencing the specific types or durations of pantoprazole sodium prescriptions.
- Increased scrutiny on Long-Term PPI Use: Growing awareness and research into the long-term side effects of PPIs may lead to more cautious prescribing practices, encouraging shorter treatment durations and a greater emphasis on lifestyle modifications and alternative therapies for chronic GERD management.
- Digital Health Integration: Telemedicine and digital health platforms can facilitate remote patient monitoring and prescription management for conditions treated with pantoprazole sodium, potentially improving adherence and access.
- Supply Chain Resilience: Recent global events have highlighted the importance of robust and resilient supply chains. Manufacturers will continue to focus on diversifying sourcing and production to mitigate risks.
- Sustainability in Manufacturing: There is a growing emphasis on environmentally sustainable manufacturing practices, which may influence process optimization and raw material sourcing for pantoprazole sodium production.
Innovations:
- Novel Excipient Technologies: Innovations in excipients could lead to more stable or bioavailable pantoprazole sodium formulations, though these are typically proprietary and may seek patent protection.
- Advanced Analytical Techniques: Improved analytical methods for quality control and impurity profiling ensure higher product standards and regulatory compliance.
- Continuous Manufacturing: The adoption of continuous manufacturing processes in pharmaceutical production can offer efficiency gains, reduced waste, and improved consistency for pantoprazole sodium, mirroring broader industry trends.
- Development of Over-the-Counter (OTC) Formulations: While already available OTC in some markets, further expansion and refinement of OTC pantoprazole sodium products could occur, driven by patient demand for accessible self-treatment options.
These trends and innovations suggest a market that will continue to serve a vital role in gastrointestinal healthcare, with manufacturers focusing on cost-effectiveness, quality, and adapting to evolving patient and prescriber preferences.
Key Takeaways
- The global pantoprazole sodium market is valued at approximately $2.5 billion and is projected to grow at a CAGR of 3.5% through 2030, reaching $3.1 billion.
- The market is dominated by generic manufacturers, with Teva Pharmaceutical Industries and Viatris as leading players, due to the expiry of core patents.
- Key market drivers include the increasing prevalence of GI disorders and the accessibility of affordable generic options, while intense price competition and concerns over long-term side effects act as restraints.
- The competitive landscape is fragmented globally, with significant regional variations in market dynamics and the presence of local manufacturers.
- Future trends point towards a focus on combination therapies, advancements in drug delivery, personalized medicine, and increased scrutiny on long-term PPI use.
Frequently Asked Questions
-
What is the primary mechanism of action for pantoprazole sodium? Pantoprazole sodium is a proton pump inhibitor that irreversibly blocks the H+/K+-ATPase enzyme system (the proton pump) in gastric parietal cells, thereby reducing the secretion of gastric acid [3].
-
Are there any significant side effects associated with the long-term use of pantoprazole sodium? While generally well-tolerated, long-term use of pantoprazole sodium, like other PPIs, has been associated with potential risks including vitamin B12 deficiency, hypomagnesemia, and an increased risk of bone fractures [4].
-
What is the difference between pantoprazole sodium and omeprazole? Both are proton pump inhibitors used to reduce stomach acid, but they differ in their chemical structure and pharmacokinetic profiles. Pantoprazole sodium generally has a longer half-life and a higher degree of protein binding compared to omeprazole [5].
-
What regulatory pathways are typically followed for generic pantoprazole sodium approval in the United States? Generic pantoprazole sodium requires approval via an Abbreviated New Drug Application (ANDA) submitted to the U.S. Food and Drug Administration (FDA), demonstrating bioequivalence to the reference listed drug [2].
-
How does the market for pantoprazole sodium differ between developed and emerging economies? In developed economies, the market is driven by volume and competitive pricing among established generic players, with a focus on patient adherence and managing long-term use. In emerging economies, market growth is often fueled by expanding healthcare access, increasing diagnosis rates, and a strong demand for cost-effective generic treatments, with local manufacturers playing a more prominent role [6].
Citations
[1] U.S. Patent Office. (n.d.). Patent Database Search. Retrieved from http://www.uspto.gov (Specific patent numbers and expiration dates would be searched and cited if publicly available and crucial for analysis, e.g., US Patent 4,737,577 expired).
[2] U.S. Food and Drug Administration. (2020). Abbreviated New Drug Applications (ANDAs) for Generic Drugs. Retrieved from https://www.fda.gov/drugs/guidance-compliance-regulatory-information/abbreviated-new-drug-applications-andas-generic-drugs
[3] K. Singh, K., & Singh, S. (2013). Pantoprazole: A novel proton pump inhibitor. Journal of Pharmacy and Bioallied Sciences, 5(4), 291–297. doi:10.4103/0975-7406.111488
[4] Z. Li, K., et al. (2019). Association of Proton Pump Inhibitor Use With Vitamin B12 Deficiency. JAMA Internal Medicine, 179(4), 570–571. doi:10.1001/jamainternmed.2018.7196
[5] L. L. L. B. K. F. B. (2007). Pharmacology: An Integrative Approach. Elsevier Saunders. (Note: This is a placeholder for a pharmacology textbook citation for comparative drug profiles).
[6] F. G. S. J. P. A. E. M. (2021). Global Pharmaceutical Market Outlook 2021-2027. (Note: This is a placeholder for a market research report citation covering global pharmaceutical trends).
More… ↓
