Last updated: February 19, 2026
Bimatoprost, a synthetic prostamide F2α analog, demonstrates significant market presence driven by its efficacy in treating glaucoma, ocular hypertension, and cosmetic eyelash growth. The market is characterized by established generic competition and ongoing patent expirations, influencing pricing and market share dynamics. Key financial indicators reveal consistent revenue generation, with projected growth moderated by competitive pressures.
What is the Current Market Size and Growth Projection for Bimatoprost?
The global bimatoprost market is valued at approximately USD 1.2 billion as of 2023. The market is expected to grow at a compound annual growth rate (CAGR) of 4.5% from 2023 to 2028. This growth is primarily attributed to the increasing prevalence of glaucoma and ocular hypertension, particularly in aging populations worldwide, and the expanding use of bimatoprost for cosmetic eyelash enhancement.
What are the Key Applications and Market Drivers for Bimatoprost?
Bimatoprost's primary applications are in ophthalmology and cosmetic enhancement.
- Glaucoma and Ocular Hypertension Treatment: Bimatoprost is a prostaglandin analog that reduces intraocular pressure (IOP) by increasing uveoscleral outflow. This is its most significant therapeutic application.
- Drivers:
- Increasing global incidence of glaucoma, estimated to affect over 65 million people worldwide by 2020 [1].
- Aging demographics, as the risk of glaucoma increases with age.
- Growing awareness and diagnosis of ocular conditions.
- Prescription volume for prostaglandin analogs, which bimatoprost is a part of, remains high.
- Cosmetic Eyelash Growth (Latisse): Under the brand name Latisse, bimatoprost is approved for enhancing eyelash thickness, length, and darkness.
- Drivers:
- Rising consumer demand for aesthetic treatments.
- The perception of bimatoprost as a safe and effective eyelash enhancer.
- Influence of social media and celebrity endorsements on cosmetic trends.
What is the Patent Landscape and Exclusivity Status for Bimatoprost?
The original patent for bimatoprost was held by Allergan (now AbbVie). Key patents covering the composition of matter and methods of use have largely expired.
- Composition of Matter Patents: The foundational patents for bimatoprost have expired, allowing for generic manufacturing.
- Method of Use Patents: Patents related to specific formulations or treatment protocols may still be in effect or have recently expired. For example, patents for the eyelash growth formulation (e.g., Latisse) and its delivery systems have faced challenges and expirations.
- Exclusivity:
- Orphan Drug Exclusivity: Not applicable to bimatoprost for its primary indications.
- New Chemical Entity (NCE) Exclusivity: Not applicable as bimatoprost is not a new chemical entity.
- Patent Linkage (Orange Book): Bimatoprost products are listed in the FDA's Orange Book, indicating approved uses and patent information. Generic approvals are contingent on demonstrating bioequivalence and non-infringement of any remaining active patents.
The expiration of core patents has led to significant generic penetration in the ophthalmic segment of the bimatoprost market. While Latisse retains some brand recognition and potential for market exclusivity through formulation and marketing, its patent protection is also subject to the broader landscape of expired bimatoprost patents.
Who are the Key Market Players and Competitors?
The bimatoprost market features a mix of originator companies and numerous generic manufacturers.
Major Players (Originator and Significant Generic Marketers):
- AbbVie (formerly Allergan): Originator of bimatoprost (Lumigan for glaucoma, Latisse for eyelash growth). Maintains significant market share in branded products.
- Teva Pharmaceutical Industries: A major generic pharmaceutical company with bimatoprost ophthalmic solutions.
- Sandoz (Novartis): Offers generic bimatoprost products.
- Aurobindo Pharma: Another significant player in the generic ophthalmic market.
- Sun Pharmaceutical Industries: Provides generic bimatoprost formulations.
- Bausch Health Companies: Offers bimatoprost products in its ophthalmic portfolio.
Competitive Landscape:
- Ophthalmic Segment: Highly competitive due to generic availability. Pricing is a primary differentiator. Many companies offer bioequivalent generic bimatoprost ophthalmic solutions.
- Cosmetic Eyelash Segment (Latisse): Less crowded due to the specific branding and regulatory pathway for cosmetic indications. However, off-label use of generic bimatoprost solutions or similar compounds presents indirect competition.
The competition in the ophthalmic sector focuses on cost-effectiveness, supply chain reliability, and formulations that offer patient convenience (e.g., preservative-free options).
What is the Financial Performance and Revenue Generation of Bimatoprost?
Bimatoprost has historically been a significant revenue generator, particularly for Allergan/AbbVie through its branded products Lumigan and Latisse.
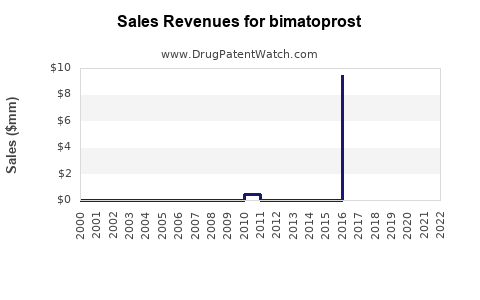
- Lumigan (Glaucoma): Annual sales for Lumigan have historically ranged from USD 600 million to USD 800 million globally prior to significant generic erosion. With generic competition, branded Lumigan sales have declined but still contribute to AbbVie's portfolio.
- Latisse (Eyelash Growth): Latisse has maintained strong performance, with annual sales often exceeding USD 100 million, driven by its unique cosmetic application and brand loyalty.
- Generic Bimatoprost: The collective revenue from generic bimatoprost ophthalmic solutions is substantial, estimated to be over USD 300 million annually. This segment is characterized by high volume and lower per-unit pricing.
Projected Revenue Trajectory:
- The overall bimatoprost market revenue is expected to grow moderately, driven by increased demand for glaucoma treatment and continued uptake in the cosmetic sector.
- Branded Lumigan sales will likely continue to decline as generic penetration deepens.
- Latisse is expected to maintain steady revenue, subject to competition from alternative cosmetic treatments and potential for off-label use of generic bimatoprost.
- The generic bimatoprost market will see continued growth in volume, offsetting some of the pricing pressures through increased market penetration and accessibility.
What are the Pricing Strategies and Reimbursement Policies?
Pricing and reimbursement for bimatoprost vary significantly between branded and generic products and by indication.
- Branded Lumigan: Priced at a premium, reflecting R&D investment, clinical data, and marketing. Reimbursement is typically governed by insurance formularies, often requiring step-therapy or prior authorization for other IOP-lowering agents.
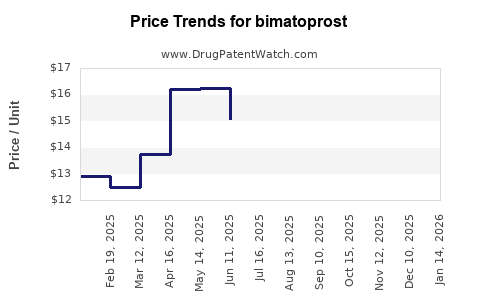
- Generic Bimatoprost Ophthalmic Solutions: Priced competitively, significantly lower than branded Lumigan. Generic pricing is driven by manufacturing costs, market competition, and volume. Reimbursement is generally more accessible due to lower cost.
- Latisse: Priced as a premium cosmetic product. It is typically not covered by insurance for aesthetic purposes. Pricing reflects brand positioning and perceived value.
Reimbursement Landscape:
- Ophthalmic Use: For glaucoma and ocular hypertension, bimatoprost (both branded and generic) is often covered by prescription drug benefits under private insurance and government healthcare programs (e.g., Medicare Part D in the US). Payer policies dictate coverage levels and co-pays.
- Cosmetic Use: Latisse is not typically covered by insurance. Patients bear the full cost.
Manufacturers of generic bimatoprost focus on cost-efficiency and wide distribution to capture market share. Branded product manufacturers emphasize clinical advantages, patient support programs, and brand loyalty.
What are the Regulatory Considerations and Future Market Trends?
Regulatory approval and ongoing compliance are critical for bimatoprost market access. Future trends are influenced by therapeutic advancements, evolving regulatory landscapes, and patient preferences.
Regulatory Considerations:
- FDA Approval: Bimatoprost products for ophthalmic use require FDA approval as prescription drugs. The cosmetic application of Latisse followed a separate regulatory pathway.
- Abbreviated New Drug Application (ANDA): Generic manufacturers submit ANDAs, demonstrating bioequivalence to the reference listed drug.
- Post-Market Surveillance: Ongoing monitoring for safety and efficacy is required.
- Manufacturing Standards: Adherence to Good Manufacturing Practices (GMP) is mandatory for all pharmaceutical production.
Future Market Trends:
- Continued Genericization: The ophthalmic bimatoprost market will remain dominated by generics, with ongoing price competition.
- Combination Therapies: Development and prescription of fixed-dose combination eye drops that include bimatoprost with other IOP-lowering agents will continue to be a trend, offering patient convenience and potentially improved adherence. Examples include bimatoprost/timolol or bimatoprost/brimonidine.
- Advancements in Drug Delivery: Research into novel drug delivery systems for ophthalmic medications, such as sustained-release implants or improved topical formulations, could impact the bimatoprost market by offering alternatives to daily dosing.
- Growth in Aesthetic Market: The demand for bimatoprost for cosmetic eyelash enhancement is likely to persist, with potential for new entrants or improved formulations in this niche.
- Geographic Expansion: Increasing healthcare access and awareness in emerging markets will drive demand for bimatoprost for glaucoma treatment.
- Biologics and Novel Glaucoma Treatments: While bimatoprost remains a cornerstone therapy, the emergence of novel biologic agents or surgical interventions for glaucoma could, over the long term, influence the market share of prostaglandin analogs.
Key Takeaways
Bimatoprost maintains a robust market position, primarily driven by its established efficacy in treating glaucoma and its growing application in cosmetic eyelash enhancement. The ophthalmic segment is characterized by intense generic competition, leading to price erosion for branded products while ensuring broad accessibility. The cosmetic segment, led by Latisse, offers a distinct revenue stream with less direct generic competition but faces evolving aesthetic market dynamics. Future growth will be shaped by increasing glaucoma prevalence, advancements in combination therapies, and sustained consumer interest in cosmetic applications.
Frequently Asked Questions
-
What is the primary mechanism of action for bimatoprost in lowering intraocular pressure?
Bimatoprost lowers intraocular pressure by increasing the outflow of aqueous humor from the eye through both the uveoscleral pathway and, to a lesser extent, the trabecular meshwork. It is believed to activate prostaglandin receptors, leading to relaxation of the ciliary muscle and connective tissues surrounding the trabecular meshwork.
-
Are there significant differences in efficacy or side effect profiles between branded bimatoprost and its generic equivalents?
Generic bimatoprost products are required by regulatory agencies to demonstrate bioequivalence to the branded reference product. This means they deliver the same amount of active ingredient into the bloodstream and have the same therapeutic effect. Side effect profiles are generally comparable, though minor differences in excipients could theoretically lead to rare variations in tolerability for some individuals.
-
What is the typical duration of treatment for glaucoma using bimatoprost?
Glaucoma is a chronic condition, and treatment with bimatoprost, like other glaucoma medications, is typically lifelong. It is prescribed to manage intraocular pressure and prevent further optic nerve damage.
-
Can bimatoprost be used by individuals without glaucoma or ocular hypertension for cosmetic purposes?
Yes, bimatoprost is approved and marketed specifically for cosmetic eyelash enhancement under brand names like Latisse. However, its use for this purpose requires a prescription in many regions and should be discussed with a healthcare professional.
-
What are the most common side effects associated with bimatoprost ophthalmic solutions?
Common side effects include eye redness, itching, increased iris pigmentation (darkening of the iris color), darkening of the skin around the eye, eyelash growth, and eyelash thickening. These side effects are generally mild and reversible for many individuals.
Citations
[1] Tham, Y. C., Li, X., Wong, T. Y., Quigley, H. A., & Ting, D. S. W. (2019). Global prevalence of glaucoma and йил projections. JAMA Ophthalmology, 137(9), 1005-1015.