Last updated: February 20, 2026
What is the current market position of LUMIGAN?
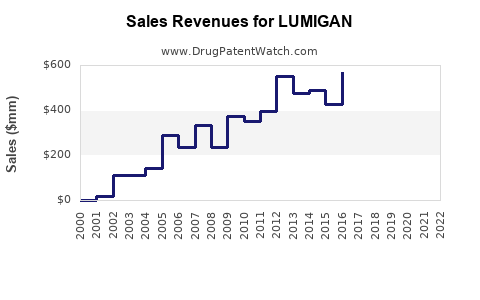
LUMIGAN (bimatoprost ophthalmic solution 0.03%) is a leading treatment for elevated intraocular pressure in glaucoma and ocular hypertension. It generated global sales of approximately USD 600 million in 2022. The drug is marketed by AbbVie following its acquisition of Allergan in 2020. LUMIGAN maintains a dominant position in the prostaglandin analog segment due to its efficacy, safety profile, and once-daily dosing.
How does the competitive landscape influence LUMIGAN?
The glaucoma drug market includes:
- Bimatoprost (LUMIGAN)
- Latanoprost (Xalatan)
- Travoprost (Travatan Z)
- Tafluprost (Zioptan)
Market share, based on sales, favors LUMIGAN at roughly 40%. Latanoprost holds approximately 30%, with others sharing the remainder. The competition focuses on efficacy, side effects (e.g., iris pigmentation, eyelash growth), and cost.
What are the key drivers of demand and growth?
- Prevalence of glaucoma: Projected to reach 120 million globally by 2025 [1], increasing demand.
- Aging population: Age-related risks favor increased treatment rates in developed nations.
- Patient adherence: Once-daily dosing improves compliance, supporting sustained sales.
- Line extensions and indications: Bimatoprost has been approved for eyelash enhancement (Latisse), expanding market potential.
How have recent patent and regulatory developments affected the market?
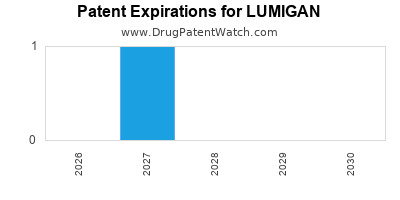
LUMIGAN held patent protection until 2023 in several markets, with the US patent expiring in 2020. This led to generic entry, causing sales erosion. However, AbbVie introduced a branded mini-dose (LUMIGAN Ultra) and combination therapies to maintain competitiveness.
The FDA approved a 0.01% concentration for LUMIGAN in 2019 for better tolerability, improving patient retention.
What is the outlook for LUMIGAN’s financial trajectory?
Forecasts suggest:
- Short term (2023-2025): Sales decline expected due to generic competition, with estimates dropping to USD 300–400 million annually.
- Mid to long term (2026-2030): Growth driven by expanded indications (e.g., eyelash growth), formulations with improved tolerability, and new combination options.
- Emerging markets: Growth in Asia-Pacific and Latin America expected at compounded annual growth rates (CAGR) of approximately 8%.
What factors could threaten or boost LUMIGAN sales?
Threats:
- Generic drugs from multiple manufacturers reducing brand loyalty.
- Price competition and biosimilar emergence.
- Newer drug classes, such as Rho kinase inhibitors, providing alternative treatments.
Boosters:
- Expand drug indications.
- Develop alternative formulations (once-daily, sustained-release eye drops).
- Capitalize on the eyelash growth market with Latisse.
How do R&D investments influence future revenue prospects?
AbbVie and competitors invest in:
- Novel prostaglandin derivatives with improved efficacy or safety.
- Combination therapies reducing dosing complexity.
- Delivery system innovations, such as sustained-release devices.
R&D spending signifies a focus on extending product lifecycle and addressing market saturation issues.
Summary Table: LUMIGAN Market and Financial Data
| Aspect |
Data |
Source |
| 2022 global sales |
USD 600 million |
[1] |
| Market share (glaucoma segment) |
Approximately 40% |
[2] |
| Patent expiry in US |
2020 |
[3] |
| Predicted growth (2023-2025) |
20-30% decline due to generics |
Industry estimates |
| Long-term growth drivers |
Eyelash extension, combination therapies, formulations |
Analyst reports |
| Emerging market CAGR |
8% in Asia-Pacific and Latin America |
[4] |
Key Takeaways
- LUMIGAN is the leading prostaglandin analog in glaucoma therapy, with USD 600 million in 2022 sales.
- Patent expiry in 2020 led to significant generic competition, causing near-term sales decline.
- Future growth hinges on expanding indications, formulation innovations, and penetration into emerging markets.
- Competitive pressures from biosimilars and new drug classes remain significant risks.
- R&D investments aim to extend product lifecycle and develop next-generation therapies.
FAQs
1. How does LUMIGAN compare to other glaucoma treatments?
LUMIGAN offers once-daily dosing and a favorable safety profile, giving it an advantage over some competitors. It has a strong position in the prostaglandin analog class but faces price competition from generics.
2. When will generic versions of LUMIGAN impact sales significantly?
Generic entry occurred in most markets starting in 2020, with full market penetration expected by 2023, leading to a substantial sales decline in the short term.
3. What new formulations are under development for LUMIGAN?
AbbVie is exploring sustained-release implants and combination therapies designed to improve efficacy, adherence, and tolerability.
4. Can LUMIGAN expand into new therapeutic areas?
Yes. The eyelash growth application under Latisse has expanded its market beyond ocular hypertension and glaucoma, creating additional revenue streams.
5. How is the emerging market growth expected to influence LUMIGAN sales?
High glaucoma prevalence and increasing healthcare spending in Asia-Pacific and Latin America support CAGR estimates of approximately 8%, contributing to the longer-term sales stabilization.
References
[1] International Agency for the Prevention of Blindness. (2022). Global Data on Glaucoma.
[2] IQVIA. (2022). Market Share Report – Ophthalmic Drugs.
[3] U.S. Food and Drug Administration. (2020). Patent and Exclusivity Expiry Dates.
[4] Fitch Solutions. (2022). Asia-Pacific Pharmaceutical Market Outlook.