Share This Page
Drug Sales Trends for bimatoprost
✉ Email this page to a colleague
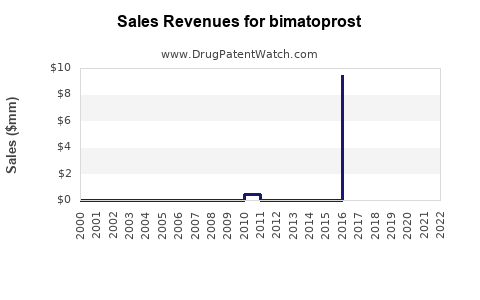
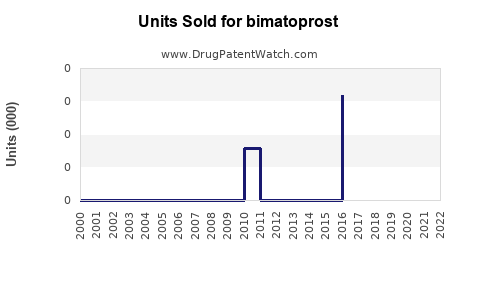
Annual Sales Revenues and Units Sold for bimatoprost
| Drug Name | Revenues (USD) | Units | Year |
|---|---|---|---|
| BIMATOPROST | ⤷ Start Trial | ⤷ Start Trial | 2022 |
| BIMATOPROST | ⤷ Start Trial | ⤷ Start Trial | 2021 |
| BIMATOPROST | ⤷ Start Trial | ⤷ Start Trial | 2020 |
| BIMATOPROST | ⤷ Start Trial | ⤷ Start Trial | 2019 |
| BIMATOPROST | ⤷ Start Trial | ⤷ Start Trial | 2018 |
| BIMATOPROST | ⤷ Start Trial | ⤷ Start Trial | 2017 |
| >Drug Name | >Revenues (USD) | >Units | >Year |
Bimatoprost Market Analysis and Financial Projection
What Is the Current Market Size for Bimatoprost?
The global eye care market was valued at approximately $18 billion in 2022, with a significant share attributable to glaucoma and ocular hypertension treatments, including bimatoprost. Bimatoprost, marketed as Lumigan and popular as a cosmetic eyelash enhancer (Latisse), accounts for a substantial portion of this segment.
Estimated sales of bimatoprost in 2022 ranged between $1.2 billion and $1.5 billion worldwide. This figure combines prescriptions for glaucoma and cosmetic uses. The glaucoma segment dominates with about 82% of total bimatoprost sales, driven by its efficacy in reducing intraocular pressure (IOP).
How Is Bimatoprost Positioned in the Market?
Therapeutic Use
- Approved for treating open-angle glaucoma and ocular hypertension.
- Prescribed primarily to patients with elevated IOP, a risk factor for optic nerve damage.
Cosmetic Use
- Marketed as Latisse for eyelash growth.
- Growing consumer interest fuels sales in the cosmetic segment, especially in North America and Europe.
Competitive Landscape
- Bimatoprost faces competition from other prostaglandin analogs: latanoprost, travoprost, and tafluprost.
- The patent for Lumigan expired in 2018 in some jurisdictions, leading to generic options now capturing market share.
- Fresenius and Santen are key players marketing generics, impacting overall sales.
Regulatory Environment
- Approvals extend across North America, Europe, Asia-Pacific, and Latin America.
- The U.S. FDA approved bimatoprost in 2001; the European Medicines Agency in 2003.
What Are Sales Projections for Bimatoprost?
Short-term Outlook (2023-2025)
- Sales are projected to grow at a CAGR of 4-6%, driven by increased awareness of glaucoma treatments and expanding cosmetic use.
- Generic competition might cap growth in mature markets but expansion into emerging markets offers growth opportunities.
Mid-term Outlook (2026-2030)
- The global eye care market is expected to reach around $22-25 billion by 2030.
- Bimatoprost's share could increase due to new formulations and extended indications, such as potential uses in ocular surface disorders.
- Increasing aging populations globally will sustain demand for glaucoma medications.
Regional Dynamics
- North America is the largest market, accounting for roughly 45% of total sales.
- Asia-Pacific is expected to see the fastest growth, at a CAGR exceeding 8%, due to rising prevalence of glaucoma and increased healthcare infrastructure.
What Factors Will Influence Market Growth?
Generics and Patent Expiry: Increased availability of low-cost generics reduces revenue for brand-name formulations but expands accessibility, potentially increasing total sales volume.
New Indications and Formulations: Ongoing research into bimatoprost's application in ocular surface disorders, such as dry eye disease, could open new markets.
Technological Innovation: Development of sustained-release implants or eyedrops with improved bioavailability may enhance patient adherence and expand usage.
Regulatory Changes: Stringent regulatory pathways in certain jurisdictions could delay new formulations, affecting sales.
Market Penetration in Emerging Economies: Countries like China and India demonstrate rising glaucoma prevalence and increasing healthcare infrastructure, creating growth opportunities.
How Do Sales of Bimatoprost Compare to Competitors?
| Drug | Market Share (2022) | Key Uses | Pricing Tier | Patent Status |
|---|---|---|---|---|
| Bimatoprost | 50-55% | Glaucoma, eyelash growth | Mid-to-high | Expired (some regions) |
| Latanoprost | 25-30% | Glaucoma | Mid | Patent expired (2010) |
| Travoprost | 10-15% | Glaucoma | Lower margins available | Patent expired (2014) |
| Tafluprost | 8-10% | Glaucoma | Premium pricing (Europe) | Patent pending |
Key Challenges and Opportunities
- Pricing Pressure: Post-patent expiration, generic competition reduces revenues for branded bimatoprost.
- Formulation Developments: Improved delivery systems could enhance effectiveness and compliance.
- Demographic Trends: Aging populations support sustained demand, especially in markets with increasing glaucoma prevalence.
- Regulatory Hurdles: Extended approval processes or restrictions could delay market entry for new formulations or indications.
Summary of Market Drivers and Barriers
| Drivers | Barriers |
|---|---|
| Aging populations increase glaucoma cases | Patent expiry leads to price erosion |
| Growing cosmetic application segment | Regulatory delays for new indications |
| Entry into emerging markets | Competition from generics |
| Advances in drug delivery technology | Market saturation in mature regions |
Key Takeaways
- Bimatoprost remains a leading treatment for glaucoma, with a market valuation around $1.2-$1.5 billion.
- Sales are expected to grow modestly through 2025, then accelerate as new formulations and markets develop.
- Competition from generics is significant, but expanding indications and regional growth mitigate impacts.
- The aging population and increasing glaucoma prevalence are primary growth drivers.
- Regulatory changes and technological advances will shape future market dynamics.
FAQs
1. What are the main drivers behind bimatoprost sales?
The core drivers include its proven efficacy in lowering intraocular pressure, expanding use in cosmetic eyelash growth, and increasing prevalence of glaucoma globally.
2. How does patent expiry affect bimatoprost sales?
Patent expiry leads to generic competition, reducing prices and revenue from brand-name drugs but expanding market access and volume.
3. What future therapeutic developments could influence sales?
New formulations, sustained-release implants, and research into additional ocular indications could boost future sales.
4. Which regions are most promising for market growth?
North America currently dominates, but Asia-Pacific offers the fastest growth owing to demographic trends and healthcare investment.
5. How does bimatoprost compare with competitors in terms of pricing?
Generics have driven prices down, offering lower-cost alternatives that impact revenue for branded bimatoprost products.
Sources
[1] MarketWatch. "Eye Care Market Size, Share & Trends Analysis," 2022.
[2] GlobalData Healthcare. "Glaucoma Treatment Market Outlook," 2023.
[3] FDA. "Bimatoprost Approvals and Patents," 2001–2022.
[4] IMS Health Reports. "Prescription Trends in Ophthalmology," 2022.
[5] Statista. "Global Eye Care Market Revenue Forecast," 2023.
More… ↓
