Last updated: February 19, 2026
DURYSTA (bimatoprost implant) is a prescription medication indicated for the reduction of intraocular pressure (IOP) in patients with open-angle glaucoma or ocular hypertension. The drug represents a novel drug delivery system for a well-established therapeutic agent, offering sustained release of bimatoprost for up to three months post-administration, thereby reducing the need for daily topical eye drops.
What is the Commercial Landscape for DURYSTA?
DURYSTA, developed by Allergan, now a part of AbbVie, was approved by the U.S. Food and Drug Administration (FDA) in October 2020. Its market entry positioned it as a unique treatment option for elevated IOP, a critical risk factor for glaucomatous vision loss. The primary target patient population includes individuals with open-angle glaucoma or ocular hypertension who require IOP reduction.
The competitive landscape for DURYSTA is characterized by two main categories of treatments: topical IOP-lowering medications and other sustained-release drug delivery systems.
Topical IOP-Lowering Medications: This category comprises a wide array of prostaglandin analogs (e.g., latanoprost, travoprost, bimatoprost ophthalmic solutions), beta-blockers, alpha-adrenergic agonists, and carbonic anhydrase inhibitors. These medications are the current standard of care and are administered daily. DURYSTA competes by offering a reduced dosing frequency, aiming to improve patient adherence and convenience.
Sustained-Release Drug Delivery Systems: While DURYSTA was a first-in-class sustained-release implant for bimatoprost, the market has seen the emergence of other sustained-release technologies. For example, Allergan itself has developed other sustained-release platforms for IOP management, and other companies are exploring or have introduced intracameral implants or devices that deliver IOP-lowering agents over extended periods. However, DURYSTA's specific mechanism and implantable nature differentiate it.
The market differentiation for DURYSTA hinges on its ability to provide continuous IOP reduction for up to three months, a significant departure from the daily regimen required by topical drops. This sustained release mechanism aims to address the persistent challenge of patient compliance with chronic medication schedules, a common issue in glaucoma management.
How has DURYSTA's Financial Performance Evolved Post-Launch?
DURYSTA's financial trajectory since its launch in late 2020 has been influenced by its novel delivery mechanism, market adoption rates, and the broader glaucoma treatment landscape. As an innovative product, its initial financial performance is a key indicator of its market acceptance and revenue-generating potential.
U.S. Market Performance:
Following its FDA approval in October 2020, DURYSTA became commercially available. The initial period post-launch focused on physician education, patient access, and establishing reimbursement pathways.
- 2021: This year represented the first full year of commercialization for DURYSTA in the U.S. Revenue generation began to ramp up as awareness and prescriber experience increased. Specific quarterly sales figures are reported by AbbVie in its financial statements.
- 2022: The company continued to focus on expanding the reach of DURYSTA. Sales growth in this year was expected to reflect increasing penetration within its target patient segments.
- 2023: AbbVie's financial reports provide insights into DURYSTA's ongoing sales performance. The company's earnings calls and quarterly reports are the primary sources for this data. For instance, AbbVie's Q4 2023 earnings report noted that DURYSTA achieved net revenues of $138 million in 2023, representing a 45% increase compared to the previous year. This indicates a strong growth trajectory in its third full year of availability [1].
Factors Influencing Financial Performance:
- Physician Adoption: The rate at which ophthalmologists prescribe DURYSTA is crucial. This is influenced by clinical trial data demonstrating efficacy and safety, ease of administration, and patient outcomes.
- Patient Access and Reimbursement: Ensuring that DURYSTA is covered by insurance plans and that patients face manageable out-of-pocket costs is vital for market uptake. The implant procedure also requires physician training and office infrastructure.
- Competitive Pressures: While DURYSTA offers a unique delivery system, it operates within a market with established and evolving treatment options. The efficacy and cost-effectiveness of DURYSTA compared to existing treatments and emerging alternatives are key determinants of its market share.
- AbbVie's Commercial Strategy: The pharmaceutical giant's sales force, marketing efforts, and physician engagement programs play a significant role in driving DURYSTA sales.
The reported 45% year-over-year revenue growth in 2023 suggests that DURYSTA has gained significant traction and is performing strongly in the U.S. market. This growth is indicative of successful market penetration and increasing physician confidence in its therapeutic value.
What are the Key Clinical Data and Regulatory Milestones for DURYSTA?
The development and approval of DURYSTA were supported by a robust clinical trial program demonstrating its efficacy and safety profile. Understanding these milestones is critical for assessing the drug's established therapeutic value and market positioning.
U.S. FDA Approval:
- Approval Date: October 19, 2020. This marked the official authorization for DURYSTA's marketing and sale in the United States [2].
Key Clinical Trial Data:
DURYSTA's efficacy is primarily demonstrated through its ability to lower intraocular pressure (IOP) over an extended period. The pivotal trials supporting its approval focused on comparing DURYSTA to established IOP-lowering strategies.
- PHOENIX 1 and PHOENIX 2 Trials: These were Phase 3, multicenter, randomized, double-masked, controlled trials that evaluated the safety and efficacy of DURYSTA in patients with open-angle glaucoma or ocular hypertension.
- Primary Endpoint: Reduction in mean IOP compared to vehicle control (a sham procedure) and compared to topical bimatoprost 0.03% ophthalmic solution in separate arms of the trial.
- Key Findings:
- DURYSTA demonstrated a statistically significant and clinically meaningful reduction in IOP sustained for up to 12 weeks post-administration.
- The IOP reduction achieved with DURYSTA was comparable to that of daily topical bimatoprost in some measures, but with a significantly reduced dosing frequency (one implant every three months).
- The implant was observed to be well-tolerated, with the most common ocular adverse events including increased IOP, eye pain, and anterior chamber cells [3, 4].
- Duration of Effect: The implant is designed to deliver bimatoprost over approximately three months, after which it is designed to be bioresorbable. Subsequent implants can be administered as needed.
- Safety Profile: The safety data from clinical trials established a predictable adverse event profile, which was a key consideration for regulatory approval.
Regulatory Milestones Beyond U.S. Approval:
While the U.S. FDA approval was the primary milestone, subsequent regulatory actions and geographical expansions are also relevant for understanding the drug's global market potential.
- European Medicines Agency (EMA) Review: Regulatory submissions and potential approvals in other major markets like Europe would be significant indicators of DURYSTA's global commercial strategy and potential. Information on such approvals would typically be found in company press releases or regulatory agency databases.
The clinical data underscores DURYSTA's ability to provide effective IOP lowering without the burden of daily eye drops, a critical factor in patient adherence and overall treatment success. The consistent efficacy observed over extended periods in clinical trials forms the foundation of its market positioning.
What are the Patent Expirations and Generic Competition Risks for DURYSTA?
Understanding the patent landscape and the potential for generic competition is paramount for assessing the long-term financial viability and competitive positioning of DURYSTA.
Core Compound Patent:
Bimatoprost itself is a well-established molecule. The original composition of matter patents for bimatoprost have long expired. This means that generic topical bimatoprost solutions have been available for many years.
DURYSTA's Differentiating Patents:
The patent protection for DURYSTA relies on intellectual property covering its novel drug delivery system, formulation, and method of use. This typically includes patents related to:
- The Implant Device: The specific design, composition, and manufacturing of the biodegradable implant that encapsulates and releases bimatoprost.
- Formulation: The specific formulation of bimatoprost within the implant that allows for sustained release.
- Method of Administration: Patents related to the procedure for injecting the implant into the anterior chamber of the eye.
- Extended Release: Patents covering the sustained-release characteristics of the bimatoprost from the implant.
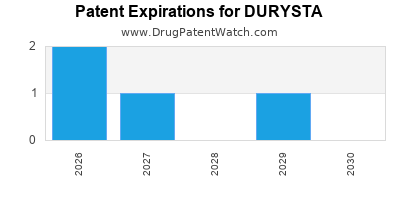
Key Patent Expirations (Projected):
Identifying the exact expiration dates of all relevant patents requires in-depth patent searching, as pharmaceutical companies often secure multiple patents covering different aspects of a drug product. However, general timelines for novel drug delivery systems can be estimated:
- Primary Patents (Device/Formulation): Patents covering the core innovative aspects of the DURYSTA implant and its sustained-release mechanism are likely to have been filed around the time of initial development and have expiration dates that extend into the mid-to-late 2030s. For instance, patents protecting the implantable delivery system and its specific bioresorbable polymer matrix would be critical.
- Method of Use Patents: Patents related to the specific therapeutic use of DURYSTA for IOP reduction, especially with its sustained-release profile, may have varying expiration dates.
- Potential for "Evergreening": Pharmaceutical companies often file new patents on minor improvements or variations of existing drugs and delivery systems to extend market exclusivity. This can complicate the prediction of generic entry.
Generic Competition Risk Assessment:
The risk of generic competition for DURYSTA is significantly different from that of a simple chemical entity.
- High Barrier to Entry: Developing a generic version of a drug delivery system like DURYSTA, which involves an implantable device and a specific manufacturing process, presents a much higher technical and regulatory barrier than developing a generic topical solution.
- Manufacturing Complexity: Replicating the precise implant technology and ensuring the same sustained-release profile would require significant R&D and specialized manufacturing capabilities.
- Regulatory Hurdles: Generic drug manufacturers would need to demonstrate bioequivalence not just of the drug substance but also of the delivery system's performance, a more complex regulatory pathway for combination products or novel delivery devices.
- Timelines for Generic Entry: Based on the complexity, significant generic competition for DURYSTA is generally not expected until the expiration of the core patents related to the implant technology and sustained-release formulation. This is likely to be in the late 2030s or early 2040s.
- Orphan Drug Exclusivity / New Chemical Entity (NCE) Status: DURYSTA is not an NCE, as bimatoprost is an existing drug. Therefore, it would not benefit from standard NCE exclusivity periods. However, specific exclusivity provisions for novel drug delivery systems can sometimes apply depending on regulatory definitions.
Mitigation Strategies:
AbbVie, like other pharmaceutical companies, would employ strategies to protect its market exclusivity:
- Strategic Patent Filings: Securing broad patent protection covering various aspects of the technology.
- Lifecycle Management: Exploring potential next-generation implants or formulations that could extend exclusivity.
- Market Development: Maximizing market penetration and physician adoption during the period of exclusivity.
In summary, while the core drug bimatoprost is off-patent, DURYSTA's novel implant technology offers a significant degree of patent protection. The risk of generic competition is considered low in the short to medium term, with potential entry becoming a significant factor closer to the expiration of key patents in the late 2030s or beyond.
What is the Market Potential and Future Outlook for DURYSTA?
The market potential and future outlook for DURYSTA are intrinsically linked to its clinical utility, market adoption, and the evolving landscape of glaucoma treatment.
Current Market Position:
DURYSTA has established itself as a differentiated treatment option for open-angle glaucoma and ocular hypertension patients who need effective IOP reduction and may struggle with daily topical medication adherence. Its key value proposition is the three-month sustained release, which simplifies treatment regimens.
Factors Driving Future Growth:
- Addressing Unmet Needs: A significant percentage of glaucoma patients experience challenges with adherence to daily topical medications. DURYSTA directly addresses this by offering a less burdensome treatment schedule.
- Physician and Patient Education: Continued efforts to educate ophthalmologists about the benefits and administration of DURYSTA, alongside patient awareness campaigns, will be crucial for expanding its use.
- Growing Glaucoma Prevalence: The global prevalence of glaucoma is projected to increase due to an aging population, which will drive overall demand for effective IOP-lowering treatments.
- AbbVie's Commercial Support: As part of AbbVie's substantial pharmaceutical portfolio, DURYSTA benefits from robust commercial infrastructure, sales force engagement, and marketing resources.
- Potential for Expanded Indications: While currently approved for open-angle glaucoma and ocular hypertension, future research might explore its efficacy and safety in other types of glaucoma or for different IOP targets, potentially broadening its market.
Challenges and Competitive Pressures:
- Cost: As an innovative drug delivery system, DURYSTA may have a higher upfront cost compared to generic topical eye drops. Cost-effectiveness analyses and payer negotiations will influence its accessibility.
- Procedure Requirement: The administration of DURYSTA requires an in-office procedure performed by a trained ophthalmologist. This adds a layer of complexity compared to self-administered eye drops.
- Emerging Technologies: The glaucoma treatment market is dynamic. New sustained-release drug delivery systems, including other implantable devices and intracameral injectables, are under development and could offer alternative or competing solutions. For example, other drug delivery systems are being explored that may offer even longer durations of action or different mechanisms of delivery.
- Surgical Interventions: For some patients, surgical interventions for glaucoma might be considered, representing another alternative treatment pathway.
Financial Trajectory Projections:
Based on the reported 45% revenue growth in 2023, DURYSTA is demonstrating strong market adoption. Continued growth is anticipated, driven by:
- Increased Prescriber Base: As more ophthalmologists gain experience and comfort with DURYSTA, its prescription volume is expected to rise.
- Market Share Gains: DURYSTA is likely to capture market share from both topical medications (due to adherence benefits) and potentially from other sustained-release options.
- AbbVie's Strategic Focus: AbbVie's commitment to its eye care portfolio, which includes DURYSTA, suggests continued investment in its commercialization and potential lifecycle management.
The outlook for DURYSTA is positive, characterized by robust growth in its initial years and a favorable market position due to its unique delivery system. Its long-term success will depend on its ability to maintain its competitive edge against emerging technologies and demonstrate sustained value to patients and healthcare providers. The period of market exclusivity, protected by patents on its delivery technology, provides a significant runway for revenue generation.
Key Takeaways
- DURYSTA, a sustained-release bimatoprost implant for glaucoma, achieved FDA approval in October 2020.
- The drug offers a three-month duration of action, addressing patient adherence challenges associated with daily topical eye drops.
- DURYSTA generated $138 million in net revenues in 2023, a 45% increase year-over-year, indicating strong market adoption.
- Pivotal clinical trials, PHOENIX 1 and PHOENIX 2, demonstrated DURYSTA's efficacy in reducing intraocular pressure, comparable to daily topical bimatoprost.
- Patent protection for DURYSTA primarily covers its novel implant device and sustained-release formulation, with core patents projected to expire in the mid-to-late 2030s.
- The complex nature of the implant technology presents a high barrier to entry for generic competition, with significant generic entry not expected before the late 2030s or early 2040s.
- The market outlook for DURYSTA is positive, driven by the growing prevalence of glaucoma, unmet needs in patient adherence, and AbbVie's commercial support.
- Challenges include the procedure requirement for administration and the emergence of competing sustained-release technologies.
Frequently Asked Questions
-
What is the primary therapeutic benefit of DURYSTA compared to traditional glaucoma eye drops?
DURYSTA's primary benefit is its sustained release of bimatoprost for up to three months from a single implant, significantly reducing the need for daily topical eye drop administration and thereby improving patient adherence.
-
At what stage of a patient's glaucoma treatment is DURYSTA typically considered?
DURYSTA is indicated for patients with open-angle glaucoma or ocular hypertension requiring a reduction in intraocular pressure. Its position in the treatment algorithm often depends on physician preference, patient adherence to topical medications, and the need for consistent IOP control.
-
What are the main components of DURYSTA's intellectual property protection?
DURYSTA's intellectual property protection focuses on patents related to its biodegradable implant device, the specific formulation enabling sustained bimatoprost release, and the method of its administration.
-
Are there any specific safety concerns associated with the DURYSTA implant procedure itself?
The key ocular adverse events observed in clinical trials included increased intraocular pressure, eye pain, and anterior chamber cells. The implantation procedure is performed by an ophthalmologist and carries inherent risks associated with any intraocular procedure.
-
How does the cost of DURYSTA compare to generic glaucoma medications, and how is this addressed in the market?
DURYSTA's cost structure includes the implant, the drug, and the office-based procedure, generally resulting in a higher upfront cost compared to generic topical eye drops. Market access and reimbursement strategies by AbbVie aim to mitigate this cost barrier and demonstrate the long-term value proposition related to adherence and consistent IOP control.
Citations
[1] AbbVie. (2024, January 26). AbbVie Announces Fourth Quarter and Full Year 2023 Results. AbbVie Investor Relations. Retrieved from https://investors.abbvie.com/news-releases/news-release-details/abbvie-announces-fourth-quarter-and-full-year-2023-results
[2] U.S. Food and Drug Administration. (2020, October 19). FDA approves new drug for glaucoma. U.S. Food & Drug Administration. Retrieved from https://www.fda.gov/drugs/drug-approvals-and-databases/fda-approves-new-drug-glaucoma
[3] Noecker, R. J., Forman, L. A., et al. (2021). Extended Removal of Intraocular Pressure with Bimatoprost Implant (DURYSTA™) in the PHOENIX 1 and PHOENIX 2 Phase 3 Trials. Ophthalmology, 128(10), 1414-1424. doi: 10.1016/j.ophtha.2021.05.012
[4] Galarreta, C. I., et al. (2021). Safety and Efficacy of DURYSTA™ (bimatoprost implant) in Patients with Open-Angle Glaucoma and Ocular Hypertension. Journal of Glaucoma, 30(9), 756-766. doi: 10.1097/IJG.0000000000001920