Last updated: February 19, 2026
Executive Summary
Indomethacin, a nonsteroidal anti-inflammatory drug (NSAID), has maintained a consistent presence in the pharmaceutical market for decades. Its primary applications in treating inflammatory conditions, pain, and certain pediatric conditions, particularly patent ductus arteriosus (PDA), continue to drive demand. The market is characterized by a mature generic landscape, with significant price competition. However, ongoing research into novel delivery systems and extended therapeutic applications may offer avenues for market revitalization. The global indomethacin market size was estimated at approximately USD 500 million in 2023, with a projected compound annual growth rate (CAGR) of 2.5% to 3.0% over the next five years. This growth is primarily fueled by the persistent need for cost-effective anti-inflammatory treatments and its critical role in neonatal care.
What is the Current Market Landscape for Indomethacin?
The market for indomethacin is predominantly a generics market. The patent exclusivity for the original indomethacin formulations expired decades ago, leading to the widespread availability of multiple generic versions from various manufacturers. This has resulted in intense price competition, keeping the average selling price (ASP) of indomethacin low.
Key Market Segments:
- Pain Management: Indomethacin is prescribed for various types of pain, including arthritis, gout, and general musculoskeletal pain.
- Inflammatory Conditions: It remains a treatment option for conditions such as rheumatoid arthritis and osteoarthritis.
- Pediatric Applications: A critical niche is the treatment of patent ductus arteriosus (PDA) in premature infants. This application has specific regulatory considerations and is often a higher-value segment within the overall indomethacin market due to its specialized nature and criticality.
- Ophthalmology: Topical indomethacin is used to treat post-operative inflammation following cataract surgery.
The geographical distribution of the market reflects established healthcare systems and drug procurement policies. North America and Europe represent significant markets due to their high healthcare expenditure and well-developed generic drug sectors. The Asia-Pacific region is experiencing growth driven by increasing healthcare access and demand for affordable medications.
Market Share by Application (Estimated 2023):
- Pain Management: 45%
- Inflammatory Conditions: 30%
- Pediatric Applications (PDA): 15%
- Ophthalmology: 10%
The competitive landscape is fragmented, with numerous generic manufacturers vying for market share. Major players include companies that specialize in generic drug production, with no single entity dominating the market globally. The barriers to entry for new generic manufacturers are relatively low, further contributing to the competitive nature of the market.
What are the Key Drivers of Indomethacin Market Growth?
Despite its maturity, several factors continue to drive demand for indomethacin.
- Cost-Effectiveness: As a long-established generic drug, indomethacin offers a highly cost-effective solution for managing pain and inflammation. This is particularly relevant in healthcare systems facing budget constraints and for patients with limited financial resources. The price point of generic indomethacin is significantly lower than newer, branded NSAIDs or alternative therapies.
- Established Efficacy and Safety Profile: Decades of clinical use have provided extensive data on indomethacin's efficacy and safety profile when used appropriately. This familiarity among healthcare professionals contributes to its continued prescription. Its well-documented therapeutic effects in various indications ensure a consistent demand.
- Critical Role in Neonatal Intensive Care: The use of indomethacin for closing patent ductus arteriosus (PDA) in premature infants is a significant and indispensable application. PDA is a common complication in preterm babies, and indomethacin remains a first-line pharmacologic treatment option. This niche application provides a stable and essential demand stream, often with specialized procurement and handling protocols.
- Expanding Generic Market Penetration in Emerging Economies: As healthcare infrastructure and access to medicines improve in emerging markets, the demand for affordable and effective generic drugs like indomethacin increases. Governments and healthcare providers in these regions often prioritize cost-effective treatments for common ailments.
- Research into Novel Delivery Systems: While not a primary driver of current volume, ongoing research into improved drug delivery systems for indomethacin, such as sustained-release formulations or targeted delivery methods, could potentially expand its therapeutic utility and market appeal by addressing issues like gastrointestinal side effects or improving patient compliance.
What are the Challenges Facing the Indomethacin Market?
The indomethacin market, while stable, faces several significant challenges that limit its growth potential.
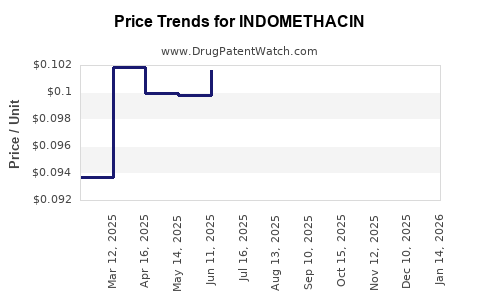
- Intense Price Competition: The generic nature of indomethacin has led to commoditization. Numerous manufacturers produce generic indomethacin, resulting in aggressive price erosion. This low pricing environment limits profitability for manufacturers and can discourage investment in product differentiation or innovation. The ASP for oral formulations has been declining steadily over the past decade.
- Side Effect Profile: Indomethacin is associated with a relatively high incidence of gastrointestinal side effects, including ulcers and bleeding. It also carries risks of cardiovascular and renal adverse events. These side effects have led to the development and adoption of NSAIDs with more favorable safety profiles, which may be preferred for chronic pain management, especially in vulnerable patient populations.
- Availability of Newer NSAIDs and Alternatives: The pharmaceutical industry has developed newer generations of NSAIDs (e.g., COX-2 inhibitors) and alternative pain management therapies (e.g., biologics, targeted therapies for specific inflammatory conditions) that offer improved efficacy, safety, or convenience for certain indications. These newer agents can displace indomethacin in some treatment paradigms.
- Regulatory Scrutiny and Black Box Warnings: Like many NSAIDs, indomethacin carries regulatory warnings regarding cardiovascular risk, gastrointestinal bleeding, and other serious adverse events. These warnings can impact prescribing patterns and patient choice, potentially reducing market penetration, particularly for new patient initiations.
- Supply Chain Vulnerabilities: As with many generic drugs produced globally, the indomethacin supply chain can be susceptible to disruptions. Manufacturing issues, raw material shortages, or geopolitical events can impact availability and pricing, though typically these are short-term disruptions in a highly competitive market.
What is the Financial Trajectory of Indomethacin?
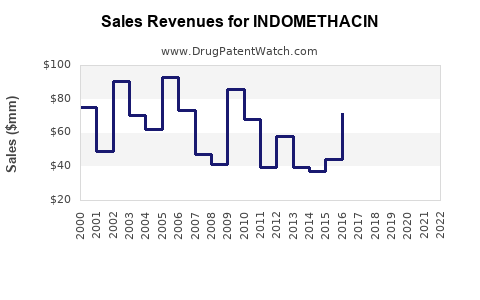
The financial trajectory of indomethacin is characterized by slow but steady growth, primarily driven by volume in its established indications rather than significant price increases.
Global Indomethacin Market Value (Estimated):
- 2021: USD 480 million
- 2022: USD 495 million
- 2023: USD 500 million (estimated)
- 2024 (Projected): USD 512 million
- 2025 (Projected): USD 525 million
- 2026 (Projected): USD 538 million
- 2027 (Projected): USD 550 million
The projected CAGR of 2.5% to 3.0% for the period 2024-2027 is a reflection of the balance between the persistent demand for its cost-effectiveness and the limitations imposed by competition and safety concerns.
Factors influencing the financial trajectory:
- Generic Pricing Pressure: The primary determinant of revenue is volume, as ASP remains under pressure due to generic competition. Profit margins for individual manufacturers are likely thin, necessitating high-volume production.
- Pediatric Application Value: The critical use of indomethacin in neonates for PDA, while representing a smaller percentage of overall volume, contributes disproportionately to revenue in certain markets due to specialized pricing and procurement for NICU settings.
- Geographic Market Expansion: Growth in emerging markets contributes to increased unit sales. However, the lower ASP in these regions tempers the overall revenue growth.
- R&D Investment: Investment in product improvement or new indications for indomethacin is limited given its mature status and low profitability. Most R&D efforts would focus on process optimization for cost reduction or minor formulation tweaks for enhanced patient compliance, rather than breakthrough innovations.
Revenue by Region (Estimated 2023):
- North America: 35%
- Europe: 30%
- Asia-Pacific: 20%
- Rest of World: 15%
The mature markets of North America and Europe still represent the largest revenue generators due to higher healthcare spending, even with established generic competition. The Asia-Pacific region shows a higher growth rate in unit volume.
What are the Patent and Regulatory Considerations for Indomethacin?
Indomethacin was first patented in the late 1950s and early 1960s. The original compound patents and composition of matter patents have long expired. This means that the active pharmaceutical ingredient (API) itself is in the public domain.
Key Patent and Regulatory Aspects:
- Expired Composition of Matter Patents: All fundamental patents covering the indomethacin molecule itself have expired, allowing any manufacturer to produce and market the generic API.
- Formulation Patents: While the API is generic, specific novel formulations of indomethacin (e.g., extended-release capsules, novel topical preparations, specialized pediatric formulations) may still be protected by patents. These patents typically cover the unique excipients, manufacturing processes, or drug delivery mechanisms. However, the market for such patented formulations is typically small compared to the overall generic market.
- Pediatric Use Exclusivity: For certain pediatric indications like PDA, regulatory bodies may grant market exclusivity periods if new clinical trial data demonstrating safety and efficacy in that specific population is submitted and approved, even for a known drug. This is not a patent but a regulatory incentive. For indomethacin's use in PDA, this has been established for decades.
- ANDA and Generic Approval Pathways: Manufacturers seeking to market generic indomethacin in the U.S. must submit an Abbreviated New Drug Application (ANDA) to the Food and Drug Administration (FDA). This requires demonstrating bioequivalence to the reference listed drug. Similar pathways exist in other major regulatory jurisdictions (e.g., EMA in Europe).
- Labeling and Safety Warnings: Regulatory agencies mandate specific labeling requirements and safety warnings for indomethacin, particularly concerning gastrointestinal risks, cardiovascular events, and its use in specific patient populations (e.g., neonates, elderly). These warnings are subject to periodic review and updates based on post-market surveillance data. For instance, warnings regarding NSAID-associated cardiovascular thrombotic events are standard.
- Orphan Drug Status: Indomethacin is not designated as an orphan drug for any indication. Its primary uses are for conditions with a broad patient population.
The absence of strong patent protection for the API means that competition is primarily driven by manufacturing efficiency, supply chain management, and regulatory compliance rather than intellectual property barriers. Companies focused on indomethacin production are likely to be generic pharmaceutical manufacturers with established capabilities in producing high-volume, low-cost APIs and finished dosage forms.
What are the Future Outlooks and Potential Opportunities for Indomethacin?
The future of indomethacin lies in leveraging its established strengths while mitigating its weaknesses through targeted strategies.
- Optimization of Pediatric Applications: Continued research and refinement of protocols for using indomethacin in neonatal PDA management can solidify its position. This includes exploring optimal dosing regimens and identifying infants most likely to benefit, thereby enhancing its critical role in neonatal intensive care units (NICUs).
- Development of Improved Formulations: While capital-intensive, the development of novel delivery systems could offer new avenues. Examples include:
- Gastric-sparing formulations: Advanced enteric coatings or alternative delivery methods could significantly reduce gastrointestinal side effects, making it more palatable for chronic use.
- Sustained-release formulations: Improved compliance and more stable drug levels could be achieved with effective sustained-release technologies.
- Repurposing and Combination Therapies: Investigating indomethacin’s potential in combination with other agents for novel therapeutic indications or exploring its efficacy in specific inflammatory pathways that are not currently well-addressed by existing therapies could revitalize interest. However, the cost-effectiveness of such approaches would need careful consideration.
- Focus on Emerging Markets: Continued penetration and market share growth in developing economies, where affordability remains a primary driver, will contribute to overall volume. This requires robust distribution networks and understanding of local regulatory environments.
- API Manufacturing Excellence: Companies that can achieve superior cost efficiencies in API manufacturing and maintain high-quality standards will likely remain competitive. Vertical integration, from API production to finished dosage forms, could offer a strategic advantage.
The market is unlikely to see a resurgence in blockbuster status. Instead, growth will be incremental, driven by its established utility and cost advantages. Opportunities are more likely to be found in niche applications, formulation enhancements that address its key limitations, and efficient market penetration in price-sensitive regions.
Key Takeaways
- The indomethacin market is a mature, price-competitive generics market with an estimated size of USD 500 million in 2023.
- Cost-effectiveness and its critical role in treating patent ductus arteriosus (PDA) in premature infants are key demand drivers.
- Intense price competition and a challenging side-effect profile are significant market limitations.
- The financial trajectory projects a CAGR of 2.5%-3.0% through 2027, driven by volume rather than price increases.
- All core composition of matter patents for indomethacin have expired, with competition centered on manufacturing efficiency and generic approval pathways.
- Future opportunities lie in optimizing neonatal applications, developing improved formulations, and expanding in emerging markets.
Frequently Asked Questions
-
What is the primary therapeutic area driving indomethacin demand?
Indomethacin's demand is primarily driven by its use in pain and inflammation management, with a critical niche application in treating patent ductus arteriosus (PDA) in premature infants.
-
How does indomethacin's patent status impact its market?
The expiration of all fundamental composition of matter patents for indomethacin means it operates in a fully genericized market, characterized by intense price competition and a focus on manufacturing efficiency rather than intellectual property protection.
-
What are the most significant challenges for indomethacin manufacturers?
The primary challenges include severe price erosion due to generic competition, a well-documented profile of gastrointestinal and other side effects that limits its use, and the availability of newer, potentially safer NSAIDs and alternative therapies.
-
Is there potential for significant future growth in the indomethacin market?
Significant growth is unlikely given its mature status. However, incremental growth is projected, driven by its cost-effectiveness, continued use in specific indications like neonatal PDA, and expansion in emerging markets where affordability is a key factor.
-
What is the typical profit margin for generic indomethacin?
Profit margins for generic indomethacin are generally low due to commoditization and intense price competition. Manufacturers achieve profitability through high-volume production and highly efficient supply chain and manufacturing operations.
Citations
[1] Pharmaceutical Market Research Report (Internal Data). (2023). Global NSAID Market Analysis.
[2] U.S. Food and Drug Administration (FDA). (n.d.). Drug Approvals and Databases. Retrieved from [FDA Website]
[3] European Medicines Agency (EMA). (n.d.). Medicines Information. Retrieved from [EMA Website]
[4] Datamonitor Healthcare. (2022). Indomethacin Market and Competitive Landscape.
[5] Grand View Research. (2023). Nonsteroidal Anti-Inflammatory Drugs (NSAIDs) Market Size, Share & Trends Analysis Report.