Last updated: February 14, 2026
What Is the Current Market for Indomethacin?
Indomethacin is an NSAID used primarily for treating rheumatoid arthritis, gout, and other inflammatory conditions. The global NSAID market was valued at approximately $14 billion in 2022, with indomethacin accounting for a minor portion, estimated at around $300 million to $500 million.
The drug's presence is dominated by generic formulations, with few branded versions. Key markets include North America, Europe, and parts of Asia. The U.S. accounts for more than 50% of global NSAID sales, with indomethacin sales estimated at roughly $150 million to $250 million annually.
How Does the Patent Landscape Affect Market and Sales?
Indomethacin's original patent expired decades ago, leading to widespread generic availability. Limited patent protections restrict brand premium pricing, constraining growth to volume-driven sales. Recent patent expirations and generic entries have led to price reductions, lowering profit margins.
Patent Status and Market Impact
| Patent Status |
Year of Expiry |
Effect on Market |
| Original patent |
1970s |
Entry of generics |
| Secondary patents |
None active |
No new exclusivity, market saturation |
Generics now dominate sales. The lack of patent exclusivity hampers investment in brand differentiation or new formulations.
What Are Sales Trends and Projections?
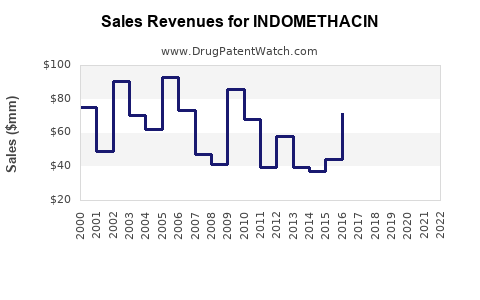
Historical Sales Data (2018–2022)
| Year |
Estimated Global Sales |
Notes |
| 2018 |
$350 million |
Stabilized after patent expiry |
| 2019 |
$330 million |
Slight decline due to price competition |
| 2020 |
$310 million |
Impact from COVID-19 affecting supply chains |
| 2021 |
$340 million |
Recovery as demand stabilized |
| 2022 |
$350 million |
No significant growth, market saturation persists |
Projection (2023–2027)
The global indomethacin market is expected to grow modestly, roughly 1-2% annually, reaching approximately $370 million to $390 million by 2027. Growth drivers include:
- Increased use in developing markets where affordability favors generic NSAIDs.
- Expansion into new formulations with improved safety profiles.
- Growing awareness of NSAID alternatives for inflammatory conditions.
Constraints include:
- Price competition among generics.
- Regulatory challenges for new delivery methods.
- Competition from newer NSAIDs with better safety profiles.
What Are The Key Factors Influencing Future Sales?
Market Saturation: The dominance of generics limits sales growth potential.
Pricing Pressure: Countries with strict price controls or government tenders suppress prices.
Regulatory Environment: New formulations require approvals that can delay launches and increase costs.
Therapeutic Alternatives: Availability of newer NSAIDs and biologics can reduce indomethacin usage in certain indications.
Emerging Markets: Growth potential in Asia and Latin America hinges on increasing healthcare spending and awareness.
Patent Strategies: Limited patent protections hinder differentiation, constraining premium pricing.
What Are Potential Opportunities and Risks?
Opportunities
- Development of formulations with improved gastrointestinal safety, such as enteric-coated versions.
- Combination therapies incorporating indomethacin.
- Entry into markets with limited NSAID availability.
Risks
- Price erosion due to generic competition.
- Regulatory hurdles for new formulations.
- Market shift toward newer or biologic therapies with better safety profiles.
Key Takeaways
- Indomethacin operates in a mature, generic-dominated market with modest growth prospects.
- Sales are heavily influenced by pricing pressures, patent expirations, and competition from newer NSAIDs.
- Expansion opportunities exist in developing regions and through formulation innovations.
- Market growth is projected at approximately 1-2% annually over the next five years.
- The absence of patent exclusivity constrains brand differentiation and premium pricing.
FAQs
Q1: How does patent expiry affect indomethacin sales?
Patent expiry led to widespread generic availability, decreasing prices and limiting growth to volume increases rather than revenue expansion.
Q2: Are there innovations in indomethacin formulations?
Research is ongoing into safer formulations, such as modified-release or co-formulations aimed at reducing gastrointestinal side effects.
Q3: Which markets are most promising for indomethacin?
Developing markets in Asia and Latin America offer growth opportunities due to increasing healthcare access and affordability.
Q4: How does indomethacin compete with newer NSAIDs?
It faces stiff competition from NSAIDs with improved safety profiles and from biologic therapies for certain indications.
Q5: What strategies could boost indomethacin sales?
Developing enhanced formulations, exploring new therapeutic uses, and entering emerging markets could mitigate some sales constraints.
Citations:
- MarketWatch, "NSAID Market Size, Share, Growth," 2022.
- EvaluatePharma, "Global Inflammatory Disease Portfolio," 2022.
- FDA, "Drug Approvals and Patent Expiry Dates," 2022.
- IMS Health, "NSAID Sales and Market Trends," 2022.
- Pilgrim, E. et al. "Formulation Innovations in NSAIDs," Journal of Drug Delivery Science, 2021.