Cubist Pharms Llc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for CUBIST PHARMS LLC, and what generic alternatives to CUBIST PHARMS LLC drugs are available?
CUBIST PHARMS LLC has six approved drugs.
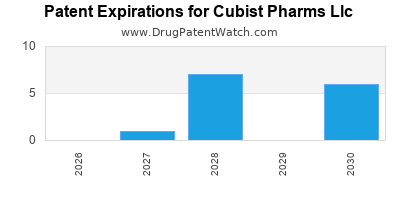
There are twenty-nine US patents protecting CUBIST PHARMS LLC drugs.
There are three hundred and forty-one patent family members on CUBIST PHARMS LLC drugs in fifty-three countries and fifty-five supplementary protection certificates in eighteen countries.
Summary for Cubist Pharms Llc
| International Patents: | 341 |
| US Patents: | 29 |
| Tradenames: | 5 |
| Ingredients: | 4 |
| NDAs: | 6 |
Drugs and US Patents for Cubist Pharms Llc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cubist Pharms Llc | SIVEXTRO | tedizolid phosphate | TABLET;ORAL | 205435-001 | Jun 20, 2014 | RX | Yes | Yes | 9,624,250 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Cubist Pharms Llc | DIFICID | fidaxomicin | FOR SUSPENSION;ORAL | 213138-001 | Jan 24, 2020 | RX | Yes | Yes | 7,906,489*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Cubist Pharms Llc | ZERBAXA | ceftolozane sulfate; tazobactam sodium | POWDER;INTRAVENOUS | 206829-001 | Dec 19, 2014 | RX | Yes | Yes | 8,685,957 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Cubist Pharms Llc | ZERBAXA | ceftolozane sulfate; tazobactam sodium | POWDER;INTRAVENOUS | 206829-001 | Dec 19, 2014 | RX | Yes | Yes | 9,872,906 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Cubist Pharms Llc | ZERBAXA | ceftolozane sulfate; tazobactam sodium | POWDER;INTRAVENOUS | 206829-001 | Dec 19, 2014 | RX | Yes | Yes | 10,028,963 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Cubist Pharms Llc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Cubist Pharms Llc | CUBICIN | daptomycin | POWDER;INTRAVENOUS | 021572-001 | Sep 12, 2003 | RE39071 | ⤷ Try a Trial |
| Cubist Pharms Llc | CUBICIN | daptomycin | POWDER;INTRAVENOUS | 021572-001 | Sep 12, 2003 | 6,852,689 | ⤷ Try a Trial |
| Cubist Pharms Llc | CUBICIN | daptomycin | POWDER;INTRAVENOUS | 021572-001 | Sep 12, 2003 | 5,912,226 | ⤷ Try a Trial |
| Cubist Pharms Llc | CUBICIN | daptomycin | POWDER;INTRAVENOUS | 021572-002 | Sep 12, 2003 | RE39071 | ⤷ Try a Trial |
| Cubist Pharms Llc | CUBICIN | daptomycin | POWDER;INTRAVENOUS | 021572-001 | Sep 12, 2003 | 8,058,238 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for CUBIST PHARMS LLC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | For Injection | 500 mg/vial | ➤ Subscribe | 2008-11-19 |
| ➤ Subscribe | Tablets | 200 mg | ➤ Subscribe | 2015-05-27 |
International Patents for Cubist Pharms Llc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| China | 105392485 | ⤷ Try a Trial |
| Israel | 214401 | ⤷ Try a Trial |
| Canada | 2626698 | ⤷ Try a Trial |
| European Patent Office | 2504353 | ⤷ Try a Trial |
| China | 102746353 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Cubist Pharms Llc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1699784 | PA2015032,C1699784 | Lithuania | ⤷ Try a Trial | PRODUCT NAME: TEDIZOLIDAS, PASIRINKTINAI ESTERIO PAVIDALU, YPAC FOSFATO ARBA JO FARMACINIU POZIURIU PRIIMTINA DRUSKA; REGISTRATION NO/DATE: EU/1/15/991 20150323 |
| 1115417 | 06C0022 | France | ⤷ Try a Trial | PRODUCT NAME: DAPTOMYCINE; REGISTRATION NO/DATE: EU/1/05/328/001-002 20060119 |
| 1539977 | 1590020-2 | Sweden | ⤷ Try a Trial | PRODUCT NAME: FIDAXOMICIN; TOTAL VALIDITY PERIOD: 16 JULY 2023 THROUGH 6 JUNE 2027 |
| 1699784 | 300762 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: TEDIZOLID, DESGEWENST IN DE VORM VAN EEN ESTER, IN HET BIJZONDER EEN FOSFAAT, OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT ERVAN; REGISTRATION NO/DATE: EU/1/15/991 20150323 |
| 1699784 | C 2015 035 | Romania | ⤷ Try a Trial | PRODUCT NAME: TEDIZOLIDOPTIONAL SUB FORMA DE ESTER, IN PARTICULAR FOSFATUL SAU O SARE ACCEPTABILA FARMACEUTIC AACESTUIA; NATIONAL AUTHORISATION NUMBER: EU/1/15/991; DATE OF NATIONAL AUTHORISATION: 20150323; NUMBER OF FIRST AUTHORISATION IN EUROPEAN ECONOMIC AREA (EEA): EU/1/15/991; DATE OF FIRST AUTHORISATION IN EEA: 20150323 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.