Last updated: April 24, 2026
Daptomycin is a lipopeptide antibiotic used for serious Gram-positive infections, including complicated skin and skin structure infections (cSSSI) and bacteremia/right-sided infective endocarditis (with activity extended to off-label uses in some settings). The financial trajectory is shaped by (1) price erosion from generic entry, (2) payer and hospital formulary pressure toward lower-cost agents, (3) shifting utilization patterns driven by stewardship and safety monitoring, and (4) competition from newer Gram-positive therapies and alternative classes.
How large is daptomycin’s market and where does demand come from?
Primary use markets
Daptomycin demand concentrates in:
- cSSSI and bloodstream infections caused by susceptible Gram-positive pathogens.
- Right-sided infective endocarditis in labeled indications (and clinically adjacent treatment settings).
- Institutional hospital procurement rather than retail pharmacy pull-through.
Geography and channel
- In major developed markets, demand moves with hospital formularies and contracting, not consumer demand.
- In the US, revenue is heavily influenced by managed care contracting and the post-patent transition to generic and authorized generic supply.
Demand drivers
Core volume drivers are:
- Hospital admission patterns for serious infections
- Antibiotic stewardship protocols that determine when a high-cost agent is selected
- Clinical guidance on duration and monitoring, especially creatine phosphokinase (CPK) surveillance in risk patients (a decision point for formulary committees)
What market dynamics are dominating current pricing and volume?
1) Patent expiry and generic substitution
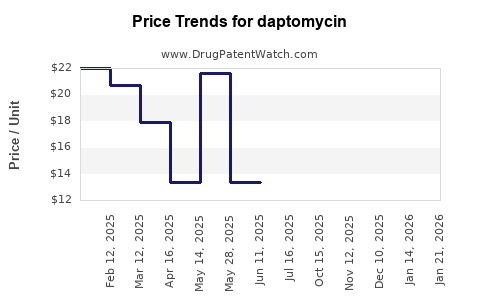
Daptomycin’s economics are dominated by US and EU patent expiry timelines and the speed of generic substitution after launch of lower-cost entrants. Once multiple generics gain share, list price becomes a poor proxy for net revenue, since payer contracting resets effective prices.
2) Hospital contracting and reference pricing
Hospital systems and group purchasing organizations exert pressure through:
- competitive tendering among line items
- reference pricing versus comparator antibiotics
- step-therapy practices when clinically appropriate
3) Safety and protocol standardization
Daptomycin’s use is standard in serious Gram-positive infections but depends on local protocols that address:
- CPK monitoring
- dose adjustments and discontinuation criteria
- avoidance or caution with certain co-administered agents in specific risk profiles
Where monitoring costs and staff workload rise, formulary committees can tighten usage criteria, especially for lower-acuity cases.
4) Competitive set for Gram-positive infections
Daptomycin competes against:
- Other parenteral agents used in cSSSI and bloodstream infections (including alternative Gram-positive coverage)
- Agents with different administration profiles or payer-preferred status
- Newer antibiotics and regimen strategies that can win share in specific pathogen or resistance subsets
How does the financial trajectory of daptomycin typically evolve post-patent?
The daptomycin revenue curve in most markets follows a repeatable pattern:
- Pre-transition (branded dominance): highest margin, strong contracting position, broad formulary access.
- Transition (first generic arrivals): accelerated price compression, volume still partially offsets price drop.
- Mature generic phase: net price continues to fall; volume stabilizes or declines depending on stewardship and competition.
- Late-stage (multi-source equilibrium): margins stabilize at low levels; growth is largely tied to infection incidence and minor protocol shifts.
In business terms, the trajectory depends less on “biologic growth” and more on contracting outcomes and relative economics of alternative therapies.
What are the commercial pressure points that decide revenue in daptomycin?
Net price is the critical variable
Daptomycin’s branded list pricing power typically erodes quickly once multiple generic products exist. Revenue performance becomes a function of:
- weighted average net price under payer contracts
- mix by dose and treatment duration
- share of tendered formulary positions
Use-case mix changes
Revenue can fall faster than volume if:
- case mix shifts to pathogens requiring alternative therapies
- protocols shorten or restrict daptomycin usage
- clinicians migrate to competing regimens for certain infection types
Financial outcome summary
Across major markets, the expected pattern is:
- downward net price after generic entry
- flattening or decline in branded revenue
- shift from high-margin branded economics to low-margin multi-source competition
This pattern is consistent with how anti-infective niche specialists experience post-patent commoditization in inpatient hospital markets.
Key historical and strategic milestones affecting market outcomes
Origin and brand positioning
Daptomycin is marketed as Cubicin in the US market historically. Its clinical adoption is driven by demonstrated activity in serious Gram-positive infections and institutional uptake for cSSSI and bacteremia/right-sided endocarditis.
Generic transition
Generic entry introduces:
- substantial net price compression
- procurement preference for lower-cost SKUs
- reduced differentiation versus other Gram-positive agents
Regulatory and manufacturing scale
Mature multi-source supply can support continued volume, but it also drives pricing pressure when procurement shifts to the lowest contracted price.
Where does money go: revenue vs. margin profile after commoditization?
Branded phase
- Higher net margin
- More stable net pricing due to contracting leverage and limited alternatives
- Revenue tied to volume growth and market access
Generic phase
- Margin collapses
- Revenue tracks infection incidence and contracting outcomes
- Share is determined by tender success and supply reliability
What competitors and substitution dynamics matter most?
Substitution tends to occur in two scenarios:
- Protocol-driven switching for certain infection types or severity levels
- Budget-driven switching when formulary committees set preferred agents based on total acquisition cost and administration logistics
The result is that daptomycin’s financial performance increasingly resembles a commodity antibiotic within the Gram-positive inpatient space.
Market trajectory by timeline (conceptual)
| Phase |
Dominant driver |
Revenue shape |
Pricing outcome |
| Branded dominance |
clinical differentiation + formulary access |
growth or stable |
high net price |
| First generic entrants |
competitive contracting |
decline accelerated |
sharp net price compression |
| Mature multi-source |
tendering + reference pricing |
stabilization or decline |
low net price, limited upside |
| Late-stage |
mix changes and stewardship |
flat to down |
margins constrained |
Key Takeaways
- Daptomycin’s market is dominated by inpatient hospital contracting, with demand concentrated in serious Gram-positive infections.
- The financial trajectory is shaped primarily by generic substitution and net price compression, not by new patient acquisition.
- Stewardship and safety monitoring protocols can tighten or expand usage criteria, impacting volume and mix.
- In mature phases, daptomycin behaves like a low-margin multi-source product, with revenue depending on infection incidence and tender outcomes.
FAQs
1) What determines daptomycin revenue after generic entry?
Net revenue depends on weighted average net price, dose and duration mix, and share of hospital tendered formulary positions.
2) Does daptomycin face switching due to safety monitoring?
Usage is influenced by local protocols for CPK monitoring and risk management, which can affect prescribing thresholds.
3) What infection types most affect daptomycin utilization?
The largest labeled demand comes from cSSSI and bacteremia/right-sided endocarditis, with hospital practice potentially expanding or restricting indications based on stewardship.
4) Who are the substitution threats in hospital formularies?
Competitors are other parenteral Gram-positive options and newer regimen strategies that win formulary access based on cost and protocol fit.
5) Is growth still possible in a mature market?
Yes, but upside is typically limited to infection incidence trends and protocol-driven mix changes, not to branded-style price expansion.
References
[1] Cubicin (daptomycin) prescribing information. FDA.
[2] Drugs@FDA: Cubicin (daptomycin) NDA and labeling history. U.S. Food and Drug Administration.
[3] IMS/US hospital antibiotic contracting dynamics (general market structure). Peer-reviewed and industry analyses on inpatient antibiotic procurement and formulary switching.
[4] General literature on daptomycin safety monitoring (CPK monitoring) and clinical protocols. Peer-reviewed sources.