SIVEXTRO Drug Patent Profile
✉ Email this page to a colleague
When do Sivextro patents expire, and what generic alternatives are available?
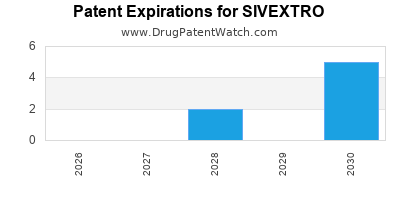
Sivextro is a drug marketed by Cubist Pharms Llc and is included in two NDAs. There are seven patents protecting this drug.
This drug has seventy-nine patent family members in thirty-nine countries.
The generic ingredient in SIVEXTRO is tedizolid phosphate. One supplier is listed for this compound. Additional details are available on the tedizolid phosphate profile page.
DrugPatentWatch® Generic Entry Outlook for Sivextro
Sivextro was eligible for patent challenges on June 20, 2018.
By analyzing the patents and regulatory protections it appears that the earliest date
for generic entry will be February 3, 2030. This may change due to patent challenges or generic licensing.
Indicators of Generic Entry
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for SIVEXTRO?
- What are the global sales for SIVEXTRO?
- What is Average Wholesale Price for SIVEXTRO?
Summary for SIVEXTRO
| International Patents: | 79 |
| US Patents: | 7 |
| Applicants: | 1 |
| NDAs: | 2 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 62 |
| Clinical Trials: | 5 |
| Patent Applications: | 147 |
| Drug Prices: | Drug price information for SIVEXTRO |
| What excipients (inactive ingredients) are in SIVEXTRO? | SIVEXTRO excipients list |
| DailyMed Link: | SIVEXTRO at DailyMed |
DrugPatentWatch® Estimated Loss of Exclusivity (LOE) Date for SIVEXTRO
Generic Entry Dates for SIVEXTRO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
POWDER;INTRAVENOUS |
Generic Entry Dates for SIVEXTRO*:
Constraining patent/regulatory exclusivity:
NDA:
Dosage:
TABLET;ORAL |
*The generic entry opportunity date is the latter of the last compound-claiming patent and the last regulatory exclusivity protection. Many factors can influence early or later generic entry. This date is provided as a rough estimate of generic entry potential and should not be used as an independent source.
Recent Clinical Trials for SIVEXTRO
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Assistance Publique - Hôpitaux de Paris | Phase 2 |
| Los Angeles Biomedical Research Institute | Phase 2 |
| Lundquist Institute for Biomedical Innovation at Harbor-UCLA Medical Center | Phase 2 |
Pharmacology for SIVEXTRO
| Drug Class | Oxazolidinone Antibacterial |
| Mechanism of Action | Breast Cancer Resistance Protein Inhibitors |
US Patents and Regulatory Information for SIVEXTRO
SIVEXTRO is protected by seven US patents and one FDA Regulatory Exclusivity.
Based on analysis by DrugPatentWatch, the earliest date for a generic version of SIVEXTRO is ⤷ Start Trial.
This potential generic entry date is based on patent 10,442,829.
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cubist Pharms Llc | SIVEXTRO | tedizolid phosphate | POWDER;INTRAVENOUS | 205436-001 | Jun 20, 2014 | RX | Yes | Yes | 8,426,389 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Cubist Pharms Llc | SIVEXTRO | tedizolid phosphate | POWDER;INTRAVENOUS | 205436-001 | Jun 20, 2014 | RX | Yes | Yes | 10,065,947 | ⤷ Start Trial | Y | ⤷ Start Trial | |||
| Cubist Pharms Llc | SIVEXTRO | tedizolid phosphate | POWDER;INTRAVENOUS | 205436-001 | Jun 20, 2014 | RX | Yes | Yes | 9,624,250 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| Cubist Pharms Llc | SIVEXTRO | tedizolid phosphate | TABLET;ORAL | 205435-001 | Jun 20, 2014 | RX | Yes | Yes | 9,624,250 | ⤷ Start Trial | Y | Y | ⤷ Start Trial | ||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for SIVEXTRO
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Merck Sharp & Dohme B.V. | Sivextro | tedizolid phosphate | EMEA/H/C/002846Sivextro is indicated for the treatment of acute bacterial skin and skin structure infections (ABSSSI) in adults and adolescents 12 years of age and older. | Authorised | no | no | no | 2015-03-23 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
International Patents for SIVEXTRO
When does loss-of-exclusivity occur for SIVEXTRO?
Based on analysis by DrugPatentWatch, the following patents block generic entry in the countries listed below:
African Regional IP Organization (ARIPO)
Patent: 87
Patent: Crystalline form of (R)-3-(-4(2-(2-methyltetrazol-5-YL)pyridin-5-YL)-3-fluorophenyl)-5-hydroxymethyloxazolidin-2-one dihydrogen phosphate
Estimated Expiration: ⤷ Start Trial
Australia
Patent: 10210627
Patent: Crystalline form of R)-3-(4-(2-(2-methyltetrazol-5-yl)pyridin-5-yl)-3-fluorophenyl)-5-hydroxymethyl oxazolidin-2-one dihydrogen phosphate
Estimated Expiration: ⤷ Start Trial
Brazil
Patent: 1008829
Patent: forma cristalina de dihidrogenofosfato de (r)-3-(4-(2-(2-metiltetrazol-5-il) piridin-5-il)-3-fluorofenil)-5-hidroximetil oxazolidin-2-ona
Estimated Expiration: ⤷ Start Trial
Canada
Patent: 51392
Patent: FORME CRISTALLINE DU DIHYDROGENOPHOSPHATE DE (R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE (CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE)
Estimated Expiration: ⤷ Start Trial
Chile
Patent: 11001855
Patent: Partículas cristalinas que comprenden al compuesto fosfato de dihidrógeno de (r)-3-(4-(2-(2-metiltetrazol-5-il)piridin-5-il)-3-fluorofenil)-5-hidroximetil oxazolidin-2-ona; proceso de preparación; mezcla que la comprende; composición farmacéutica; y su uso para tratar una infección bacteriana.
Estimated Expiration: ⤷ Start Trial
China
Patent: 2439006
Patent: Crystalline form of r)-3-(4-(2-(2-methyltetrazol-5-yl)pyridin-5-yl)-3-fluorophenyl)-5-hydroxymethyl oxazolidin-2-one dihydrogen phosphate
Estimated Expiration: ⤷ Start Trial
Patent: 7082790
Patent: 种噁唑烷酮化合物的晶型 (Crystalline form of R)-3-(4-(2-(2-methyltetrazol-5-yl)pyridin-5-yl)-3-fluorophenyl)-5-hydroxymethyl oxazolidin-2-one dihydrogen phosphate)
Estimated Expiration: ⤷ Start Trial
Colombia
Patent: 20071
Patent: Forma cristalina del fosfato de dihidrogeno r)-3-(4-(2-(2-metiltetrazol-5-il)piridin-5-il)-3-fluorofenil)-5-hidroximetil oxazolidin-2-ona
Estimated Expiration: ⤷ Start Trial
Costa Rica
Patent: 110464
Patent: FORMA CRISTALINA DEL FOSFATO DE DIHIDROGENO R)-3-(4-(2-(2-METILTETRAZOL-5-IL)PIRIDIN-5-IL)-3-FLUOROFENIL)-5- HIDROXIMETIL OXAZOLIDIN-2-ONA
Estimated Expiration: ⤷ Start Trial
Cuba
Patent: 089
Patent: FORMA CRISTALINA DEL FOSFATO DE DIHIDRÓGENO R)-3-(4-(2-(2-METILTRETAZOL-5-IL)-3-FLUOROFENIL)-5-HIDROXIMETIL OXAZOLIDIN-2-ONA
Estimated Expiration: ⤷ Start Trial
Patent: 110155
Patent: FORMA CRISTALINA DEL FOSFATO DE DIHIDRÓGENO R)-3-(4-(2-(2-METILTRETAZOL-5-IL)-3-FLUOROFENIL)-5-HIDROXIMETIL OXAZOLIDIN-2-ONA
Estimated Expiration: ⤷ Start Trial
Dominican Republic
Patent: 011000251
Patent: FORMA CRISTALINA DEL FOSFATO DE DIHIDROGENO R)-3-(4-(2-(2-METILTETRAZOL-5-IL) PIRIDIN-5-IL)-3-FLUOROFENIL)-5-HIDROXIMETIL OXAZOLIDIN-2-ONA
Estimated Expiration: ⤷ Start Trial
Ecuador
Patent: 11011285
Patent: FORMA CRISTALINA DEL FOSFATO DE DIHIDROGENO R)-3-(4-(2-(2-METILTETRAZOL-5-IL)PIRIDIN-5-IL)-3-FLUOROFENIL)-5-HIDROXIMETIL OXAZOLIDIN-2-ONA
Estimated Expiration: ⤷ Start Trial
European Patent Office
Patent: 93808
Patent: FORME CRISTALLINE DU DIHYDROGÉNOPHOSPHATE DE (R)-3-(4-(2-(2-MÉTHYLTÉTRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHÉNYL)-5-HYDROXYMÉTHYL OXAZOLIDIN-2-ONE (CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE)
Estimated Expiration: ⤷ Start Trial
Israel
Patent: 4401
Patent: צורה גבישית של (r)-3-(4-(2-(2-מתילטטראזול-5-יל)פירידין-5-יל)-3-פלואורופניל)-5-הידרוקסימתיל אוקסאזולידין-2-און דיהידרוגן פוספאט (Crystalline form of (r)-3-(4-(2-(2-methyltetrazol-5-yl)pyridin-5-yl)-3-fluorophenyl)-5-hydroxymethyl oxazolidin-2-one dihydrogen phosphate)
Estimated Expiration: ⤷ Start Trial
Japan
Patent: 84705
Estimated Expiration: ⤷ Start Trial
Patent: 12516894
Estimated Expiration: ⤷ Start Trial
Malaysia
Patent: 6354
Patent: CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE
Estimated Expiration: ⤷ Start Trial
Mexico
Patent: 4300
Patent: FORMA CRISTALINA DEL FOSFATO DE DIHIDROGENO R) -3- (4- (2- (2-METILTETRAZOL-5-IL) PIRIDIN-5-IL) -3-FLUOROFENIL) -5-HIDROXIMETIL OXAZOLIDIN-2-ONA. (CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE)
Estimated Expiration: ⤷ Start Trial
Patent: 11008093
Patent: FORMA CRISTALINA DEL FOSFATO DE DIHIDROGENO R)-3-(4-(2-(2-METILTET RAZOL-5-IL)PIRIDIN-5-IL)-3-FLUOROFENIL)-5-HIDROXIMETIL OXAZOLIDIN-2-ONA. (CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-Y L)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE.)
Estimated Expiration: ⤷ Start Trial
Patent: 20011773
Patent: FORMA CRISTALINA DEL FOSFATO DE DIHIDROGENO R)-3-(4-(2-(2-METILTET RAZOL-5-IL)PIRIDIN-5-IL)-3-FLUOROFENIL)-5-HIDROXIMETIL OXAZOLIDIN-2-ONA. (CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-Y L)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE.)
Estimated Expiration: ⤷ Start Trial
Morocco
Patent: 092
Patent: FORME CRISTALLINE DU DIHYDROGÉNOPHOSPHATE DE (R)-3-(4-(2-(2-MÉTHYLTÉTRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHÉNYL)-5-HYDROXYMÉTHYL OXAZOLIDIN-2-ONE
Estimated Expiration: ⤷ Start Trial
New Zealand
Patent: 4408
Patent: Crystalline form of r)-3-(4-(2-(2-methyltetrazol-5-yl)pyridin-5-yl)-3-fluorophenyl)-5-hydroxymethyl oxazolidin-2-one dihydrogen phosphate
Estimated Expiration: ⤷ Start Trial
Patent: 0458
Patent: Crystalline form of r)-3-(4-(2-(2-methyltetrazol-5-yl)pyridin-5-yl)-3-fluorophenyl)-5-hydroxymethyl oxazolidin-2-one dihydrogen phosphate
Estimated Expiration: ⤷ Start Trial
Patent: 2289
Patent: Crystalline form of r)-3-(4-(2-(2-methyltetrazol-5-yl)pyridin-5-yl)-3-fluorophenyl)-5-hydroxymethyl oxazolidin-2-one dihydrogen phosphate
Estimated Expiration: ⤷ Start Trial
Peru
Patent: 120585
Patent: FORMA CRISTALINA DEL FOSFATO DE DIHIDROGENO R)-3-(4-(2-(2-METILTETRAZOL-5-IL) PIRIDIN-5-IL)-3-FLUOROFENSIL)-5-HIDROXIMETIL OXAZOLIDIN-2-ONA
Estimated Expiration: ⤷ Start Trial
Philippines
Patent: 014500092
Patent: CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE
Estimated Expiration: ⤷ Start Trial
Russian Federation
Patent: 55928
Patent: Кристаллические частицы для приготовления твердых лекарственных форм для лечения бактериальных инфекций, реакционная смесь, содержащая такие частицы, и фармацевтическая композиция для лечения бактериальных инфекций (CRYSTALLINE PARTICLES FOR PREPARATION OF SOLID DOSAGE FORMS FOR TREATMENT OF BACTERIAL INFECTIONS, REACTION MIXTURE CONTAINING SUCH PARTICLES, AND PHARMACEUTICAL COMPOSITION FOR TREATING BACTERIAL INFECTIONS)
Estimated Expiration: ⤷ Start Trial
Patent: 11136537
Patent: КРИСТАЛЛИЧЕСКАЯ ФОРМА (R)-3-(4-(2-МЕТИЛТЕТРАЗОЛ-5-ИЛ)ПИРИДИН-5-ИЛ)-3-ФТОРФЕНИЛ)-5-ГИДРОКСИМЕТИЛ ОКСАЗОЛИДИН-2-ОН ДИВОДОРОД ФОСФАТА
Estimated Expiration: ⤷ Start Trial
Singapore
Patent: 201500207Q
Patent: CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE
Estimated Expiration: ⤷ Start Trial
Patent: 3497
Patent: CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE
Estimated Expiration: ⤷ Start Trial
South Africa
Patent: 1106412
Patent: CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE
Estimated Expiration: ⤷ Start Trial
Patent: 1306536
Patent: CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE
Estimated Expiration: ⤷ Start Trial
South Korea
Patent: 1739923
Estimated Expiration: ⤷ Start Trial
Patent: 1918678
Estimated Expiration: ⤷ Start Trial
Patent: 110120311
Patent: CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE
Estimated Expiration: ⤷ Start Trial
Patent: 170040371
Patent: R)-3--5-히드록시메틸 옥사졸리딘-2-온 디히드로겐 포스페이트의 결정형 (-3-4-2-2--5--5--3--5- -2- Crystalline form of R-3-4-2-2-methyltetrazol-5-ylpyridin-5-yl-3-fluorophenyl-5-hydroxymethyl oxazolidin-2-one dihydrogen phosphate)
Estimated Expiration: ⤷ Start Trial
Patent: 170135984
Patent: R)-3--5-히드록시메틸 옥사졸리딘-2-온 디히드로겐 포스페이트의 결정형 (-3-4-2-2--5--5--3--5- -2- Crystalline form of R-3-4-2-2-methyltetrazol-5-ylpyridin-5-yl-3-fluorophenyl-5-hydroxymethyl oxazolidin-2-one dihydrogen phosphate)
Estimated Expiration: ⤷ Start Trial
Spain
Patent: 34724
Estimated Expiration: ⤷ Start Trial
Tunisia
Patent: 11000381
Patent: CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE
Estimated Expiration: ⤷ Start Trial
Ukraine
Patent: 4068
Estimated Expiration: ⤷ Start Trial
Generics may enter earlier, or later, based on new patent filings, patent extensions, patent invalidation, early generic licensing, generic entry preferences, and other factors.
See the table below for additional patents covering SIVEXTRO around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Morocco | 33092 | FORME CRISTALLINE DU DIHYDROGÉNOPHOSPHATE DE (R)-3-(4-(2-(2-MÉTHYLTÉTRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHÉNYL)-5-HYDROXYMÉTHYL OXAZOLIDIN-2-ONE | ⤷ Start Trial |
| New Zealand | 547928 | Novel oxazolidinone derivatives | ⤷ Start Trial |
| World Intellectual Property Organization (WIPO) | 2010091131 | ⤷ Start Trial | |
| Tunisia | 2011000381 | CRYSTALLINE FORM OF R)-3-(4-(2-(2-METHYLTETRAZOL-5-YL)PYRIDIN-5-YL)-3-FLUOROPHENYL)-5-HYDROXYMETHYL OXAZOLIDIN-2-ONE DIHYDROGEN PHOSPHATE | ⤷ Start Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for SIVEXTRO
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1699784 | 675 | Finland | ⤷ Start Trial | |
| 1699784 | 300762 | Netherlands | ⤷ Start Trial | PRODUCT NAME: TEDIZOLID, DESGEWENST IN DE VORM VAN EEN ESTER, IN HET BIJZONDER EEN FOSFAAT, OF EEN FARMACEUTISCH AANVAARDBAAR ZOUT ERVAN; REGISTRATION NO/DATE: EU/1/15/991 20150323 |
| 1699784 | 2015/048 | Ireland | ⤷ Start Trial | PRODUCT NAME: TEDIZOLID, OPTIONALLY IN THE FORM OF AN ESTER, IN PARTICULAR A PHOSPHATE, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REGISTRATION NO/DATE: EU/1/15/991 20150323 |
| 1699784 | 1590047-5 | Sweden | ⤷ Start Trial | PRODUCT NAME: TEDIZOLID, OPTIONALLY IN THE FORM OF AN ESTER, IN PARTICULAR A PHOSPHATE, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; REG. NO/DATE: EU/1/15/991 20150325 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Market Dynamics and Financial Trajectory for SIVEXTRO (Xeflim Functional Sodium)
More… ↓
Make Better Decisions: Try a trial or see plans & pricing
Drugs may be covered by multiple patents or regulatory protections. All trademarks and applicant names are the property of their respective owners or licensors. Although great care is taken in the proper and correct provision of this service, thinkBiotech LLC does not accept any responsibility for possible consequences of errors or omissions in the provided data. The data presented herein is for information purposes only. There is no warranty that the data contained herein is error free. We do not provide individual investment advice. This service is not registered with any financial regulatory agency. The information we publish is educational only and based on our opinions plus our models. By using DrugPatentWatch you acknowledge that we do not provide personalized recommendations or advice. thinkBiotech performs no independent verification of facts as provided by public sources nor are attempts made to provide legal or investing advice. Any reliance on data provided herein is done solely at the discretion of the user. Users of this service are advised to seek professional advice and independent confirmation before considering acting on any of the provided information. thinkBiotech LLC reserves the right to amend, extend or withdraw any part or all of the offered service without notice.