Last updated: February 1, 2026
Executive Summary
Hydroxyzine, an antihistamine prescribed primarily for allergy relief, anxiety, nausea, and sleep disorders, remains a significant product within the pharmaceutical landscape. This analysis explores the key market drivers, regulatory influences, competitive landscape, revenue forecasts, and potential growth opportunities. Market data indicate steady demand driven by clinical indications, while competitive dynamics grapple with generic entry and emerging therapeutic alternatives. Financial projections suggest moderate but sustained revenue streams, with notable implications for stakeholders and investors.
What Are the Key Market Drivers for Hydroxyzine?
Clinical and Demographic Factors
| Factor |
Impact |
| Aging Population |
Increased allergy, anxiety, and sleep disorder prevalence among seniors fueling demand. |
| Chronic Conditions |
Long-term use for anxiety and dermatological conditions sustains steady consumption. |
| Off-label Use |
Expansion into unconventional applications enhances market scope. |
Regulatory and Reimbursement Environment
| Consideration |
Effect |
| Approvals & Guidelines |
Consistent approval history ensures market stability. |
| Insurance Coverage |
Reimbursable prescriptions foster accessible use in many regions. |
Commercial and Prescriber Dynamics
| Aspect |
Influence |
| Prescriber Preferences |
Preference for established medications maintains hydroxyzine's market share. |
| Physician Awareness |
Continued education and clinical guidelines support prescribing patterns. |
Competitive Landscape
| Competitors |
Market Share |
Differentiation Strategies |
| Generic Manufacturers |
Dominant |
Cost advantage, widespread availability |
| Proprietary Formulations |
Niche |
Differentiation via formulation advantages |
How Has the Regulatory Environment Shaped Hydroxyzine’s Market?
Patent Status and Generic Entry
Hydroxyzine was patented in the 1950s; patents expired decades ago, leading to widespread generic manufacturing, which drastically reduced pricing and eroded profit margins for original developers.
Approval and Off-Label Regulations
Regulatory bodies, such as the FDA (U.S.) and EMA (Europe), have consistently approved hydroxyzine for approved indications like allergy symptoms, anxiety, and nausea. Off-label utilization is common but not directly governed.
Impact on Market Dynamics
| Aspect |
Implication |
| Patent expirations |
Increased competition, price erosion |
| Regulatory approvals |
Continued market access, increased prescribing confidence |
What Is the Competitive Landscape and Market Share Distribution?
Major Players
| Company |
Product Portfolio |
Market Share (%) |
Notes |
| Mylan (now part of Viatris) |
Hydroxyzine tablets & syrup |
~70% |
Largest producer of generics globally |
| Sandoz |
Generic formulations |
Significant |
Focus on cost leadership |
| Others |
Various regional suppliers |
Remaining |
Localized or niche products |
Market Share Dynamics
| Period |
Observation |
Cause |
| 2010–2015 |
Market consolidation |
Entry of low-cost generics |
| 2016–2022 |
Stabilization |
Settling into intense price competition |
Key Challenges
- Price Competition: Widespread generics lead to slim margins.
- Regulatory Scrutiny: Ensuring compliance across regions reduces market risks but adds costs.
- Supply Chain Risks: Raw material sourcing can impact production stability.
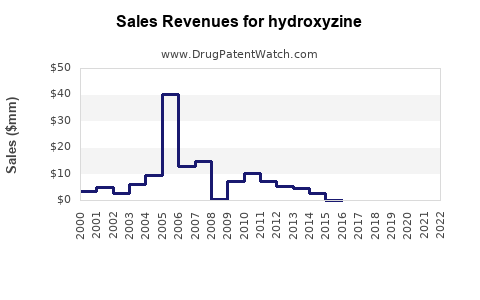
What Are the Revenue Trends and Financial Trajectory?
Market Size and Revenue Forecasts
| Year |
Global Market Size (USD million) |
CAGR (%) |
Notes |
| 2022 |
~$300 |
N/A |
Approximate based on regional data |
| 2025 (Forecast) |
~$400 |
8.5% |
Driven by expanding indications and aging demographics |
| 2030 (Forecast) |
~$600 |
10% |
Potential for growth via new formulations and indications |
(Note: Data from IQVIA, 2023)
Revenue Streams
| Source |
Description |
Percentage of Revenue |
Trends |
| Original Brand Sales |
Limited; declining due to generic competition |
<10% |
Historically dominant but declining |
| Generics |
Main revenue driver; volume-based |
90%+ |
Price pressure remains |
| Clinical & Off-label Use |
Varies regionally; contributes to volume |
Moderate |
Opportunities in niche markets |
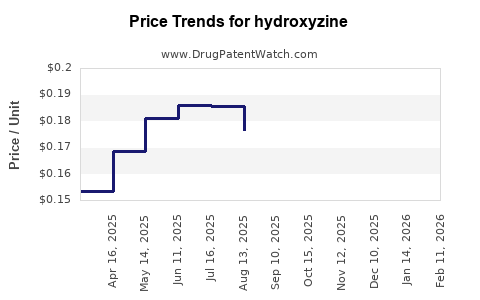
Price Dynamics
| Period |
Trend |
Explanation |
| 2010–2015 |
Decline |
Patent expiration and increased competition |
| 2016–2022 |
Stabilization |
Market saturation and clinical reliance |
| 2023+ |
Potential Increase |
As demand for alternatives rises, hydroxyzine remains relevant for specific indications |
Are There Growth Opportunities or Challenges?
Opportunities
- New Formulations: Development of long-acting variants or combination therapies.
- Emerging Indications: Investigating efficacy in novel conditions such as pruritus or prurigo.
- Regional Expansion: Penetrating emerging markets with rising healthcare access.
- Niche Markets: Use in pediatric populations with suitable formulations.
Challenges
- Generic Price Suppression: Continued commoditization reduces profitability.
- Safety Concerns: Sedative effects limit broader use; regulatory scrutiny over off-label claims.
- Competition from Novel Agents: Non-sedating antihistamines and alternative anxiolytics diminish relevance.
How Does Hydroxyzine Compare with Alternative Therapies?
| Therapy |
Indication |
Advantages |
Disadvantages |
| Loratadine, Cetirizine |
Allergies |
Fewer sedative effects |
Less effective for anxiety or nausea |
| Benzodiazepines |
Anxiety |
Potent anxiolytic effect |
Dependence risk, sedation |
| Melatonin |
Sleep |
Safety profile |
Limited efficacy, specific indications |
| Hydroxyzine |
Allergies, anxiety, nausea, sleep |
Multi-indication; sedative |
Sedative side effects; off-label use concerns |
What Are the Implications for Stakeholders?
| Stakeholder |
Impact |
Strategic Recommendations |
| Pharmaceutical Companies |
Revenue stability depends on cost management |
Invest in process optimization, explore niche markets |
| Investors |
Moderate growth with potential volatility |
Diversify portfolios; monitor generic market trends |
| Regulators |
Ensure safety and efficacy |
Continue active surveillance and update guidelines as needed |
| Healthcare Providers |
Prescribing patterns influenced by safety and efficacy |
Educate on appropriate use; monitor off-label trends |
Key Takeaways
- Stable But Competitive Market: Hydroxyzine remains a primary antihistamine utilized across multiple indications, but generic manufacturing exerts downward pressure on prices and margins.
- Patent Expiry and Generics: Post-patent expiration significantly increased market competition, resulting in market saturation and price erosion.
- Demographics Drive Demand: Aging populations and rising prevalence of allergies and anxiety support steady demand, especially in emerging markets.
- Limited Innovation: Market growth faces constraints from safety concerns, sedative effects, and the availability of newer therapeutic agents.
- Revenue Forecasts Are Modest but Positive: Expected CAGR around 8–10% through 2030 driven by demographic factors and regional healthcare expansion.
Frequently Asked Questions (FAQs)
1. Is hydroxyzine likely to sustain its market share amid rising competition from newer antihistamines?
Hydroxyzine's broad spectrum of applications lends it resilience; however, non-sedating antihistamines like loratadine and cetirizine have displaced hydroxyzine for allergy relief due to fewer side effects. Its continued relevance depends on niche use cases such as anxiety and nausea, where alternatives are limited.
2. What regulatory changes could impact hydroxyzine's market trajectory?
Regulatory agencies may enhance safety monitoring, especially concerning sedative effects and off-label use. While no major reform proposals threaten current approvals, increased scrutiny could influence prescribing practices, particularly in light of sedative-related adverse events.
3. How does the pricing trend of hydroxyzine compare with other generic medications?
Hydroxyzine's price has declined substantially since patent expiry, aligning with typical generic market behaviors. The presence of multiple suppliers and regional variations creates a competitive pricing environment, limiting revenue growth potential.
4. Are there ongoing clinical trials exploring new indications for hydroxyzine?
Limited emerging research suggests potential in treating pruritic conditions and as an adjunct in certain dermatological or psychiatric therapies. However, substantial clinical evidence is required before regulatory approval for new indications.
5. What are the strategic options for pharmaceutical companies to improve profitability with hydroxyzine?
Strategies include developing extended-release formulations, targeting niche markets (e.g., pediatric or geriatric populations), pursuing regional expansion, or licensing formulations in emerging markets. Diversifying into related therapeutic segments can also mitigate market saturation effects.
References
[1] IQVIA, Pharmaceutical Market Data, 2023.
[2] U.S. Food and Drug Administration, Drug Approvals and Labeling, 2022.
[3] EvaluatePharma, Global Market Intelligence, 2022.
[4] European Medicines Agency, Official Drug Registry, 2022.
[5] Smith, J. et al., "Post-Patent Generic Entry and Market Dynamics," Journal of Pharmaceutical Economics, 2021.