Last updated: February 19, 2026
Baxter Healthcare is a significant player in the global healthcare industry, specializing in products for home, hospital, and alternate care settings. The company focuses on acute and chronic medical conditions, with a portfolio spanning renal care, hospital products, and nutritional therapies. Its market position is characterized by a mature product pipeline in certain segments and ongoing innovation in others, particularly in connected devices and advanced drug delivery systems.
What is Baxter Healthcare's Core Business and Market Focus?
Baxter Healthcare's core business revolves around developing, manufacturing, and marketing a range of medical products and therapies. The company's primary market focus is on addressing critical healthcare needs across multiple patient care settings.
- Renal Care: This segment is a cornerstone of Baxter's business, offering hemodialysis and peritoneal dialysis solutions, as well as related pharmaceuticals and technologies. Baxter is a leading provider in this area, serving millions of patients globally who manage kidney disease. The company's offerings include dialysis machines, dialyzers, sterile solutions, and medications to manage complications of kidney failure.
- Hospital Products: Baxter supplies essential products used within hospital environments. This includes intravenous (IV) solutions and administration sets, infusion pumps, and sterile water. These products are critical for patient hydration, drug delivery, and overall hospital operations. The company also provides anesthesia and injectable drugs.
- Nutritional Therapies: Baxter offers parenteral and enteral nutrition products. These are designed for patients who are unable to obtain adequate nutrition through oral intake. The company's portfolio includes customized formulations and delivery systems for specialized nutritional support.
- Other Medical Devices and Technologies: Baxter is involved in developing and marketing a range of other medical devices, including surgical care products, advanced drug delivery systems, and connected health technologies aimed at improving patient monitoring and care coordination.
The company's strategy is to leverage its established presence in critical care areas while investing in innovation to enhance patient outcomes and operational efficiency in healthcare settings.
What are Baxter Healthcare's Key Product Categories and Their Market Significance?
Baxter Healthcare's product portfolio is segmented into distinct categories, each holding considerable market significance and contributing to the company's overall revenue and strategic positioning.
- Renal Care Products: This segment is Baxter's largest and most critical. It includes:
- Dialysis Therapies: Hemodialysis (HD) and Peritoneal Dialysis (PD) systems. Baxter is a pioneer and leader in PD, offering home-based treatment options that provide patients with greater flexibility and autonomy. Its HD solutions are widely used in both in-center and at-home settings.
- Dialyzers and Solutions: Essential components for dialysis, including artificial kidneys and sterile solutions used in HD and PD.
- Renal Pharmaceuticals: Medications to manage conditions associated with kidney disease, such as anemia and bone mineral disorders.
- Market Significance: The global dialysis market is substantial, driven by rising incidences of chronic kidney disease (CKD) and end-stage renal disease (ESRD) due to aging populations and comorbidities like diabetes and hypertension. Baxter's strong brand recognition and established distribution network make it a dominant player. The demand for home dialysis solutions is projected to grow, benefiting Baxter's PD offerings.
- Hospital Products: This broad category supports acute care settings and includes:
- IV Solutions: A comprehensive range of sterile solutions for hydration, drug dilution, and administration. These are fundamental to hospital operations.
- Infusion Systems: IV pumps and related equipment for precise drug delivery. Baxter offers a spectrum of pumps, from basic gravity-fed systems to advanced electronic infusion pumps.
- Injectable Pharmaceuticals: Pre-mixed drugs and other injectable medications for hospital use.
- Anesthesia Products: Inhaled anesthetics and other agents used in surgical procedures.
- Market Significance: Hospitals represent a consistent demand for basic medical supplies. Baxter's long-standing relationships with healthcare institutions and its reputation for quality and reliability in these essential products secure its market share. The need for safe and efficient drug delivery remains constant, underpinning the importance of its infusion systems.
- Nutrition Therapies: This segment addresses patients requiring alternative nutritional support:
- Parenteral Nutrition (PN): Intravenous nutritional solutions for patients unable to absorb nutrients from the gut.
- Enteral Nutrition (EN): Tube feeding products for patients who can digest but not swallow food.
- Market Significance: The market for nutritional support is driven by increased awareness of its role in patient recovery and reduced hospital stays. Baxter provides customizable formulations, catering to diverse patient needs, which is a key differentiator.
- Advanced Surgery and Other Devices: This includes products for surgical procedures and related technologies:
- Surgical Sealants and Hemostats: Products to control bleeding during surgery.
- Smart Pumps and Connected Technologies: Infusion pumps with advanced features for medication safety, data management, and remote monitoring.
- Market Significance: Innovation in surgical tools aims to improve patient outcomes and reduce complications. Baxter's investment in connected health technologies aligns with the trend towards digitized healthcare, enabling better data integration and remote patient management.
The market significance of these categories is underscored by Baxter's consistent revenue generation and its strategic focus on innovation within these established fields.
What are Baxter Healthcare's Core Strengths and Competitive Advantages?
Baxter Healthcare possesses several core strengths that underpin its competitive position in the global pharmaceutical and medical device markets. These advantages enable the company to maintain market share and pursue growth opportunities.
- Established Global Presence and Distribution Network: Baxter has operated internationally for decades, building a robust network of manufacturing facilities, sales offices, and distribution channels across more than 100 countries. This extensive reach allows for efficient product delivery and market penetration, particularly in diverse regulatory and economic environments.
- Data Point: Baxter's products are available in over 100 countries.
- Leadership in Renal Care: The company is a recognized leader in the renal care market, particularly in peritoneal dialysis (PD). Its long history and extensive portfolio of dialysis solutions, equipment, and pharmaceuticals provide a strong foundation and significant market share.
- Comparison: Baxter is one of the top three global suppliers of dialysis products, alongside Fresenius Medical Care and DaVita.
- Strong Brand Recognition and Reputation for Quality: Baxter's brand is synonymous with reliability and quality in essential medical products like IV solutions and dialysis equipment. This trust among healthcare providers is a significant competitive asset, often leading to preferential purchasing decisions.
- Evidence: The company's products are staples in hospitals worldwide, indicating consistent adoption.
- Integrated Product Offerings: Baxter offers a comprehensive suite of products within key therapeutic areas, such as renal care. This integration allows for a more holistic approach to patient management and creates customer loyalty by providing multiple necessary components from a single supplier.
- Example: A renal patient may use Baxter's dialysis machine, dialyzer, sterile solution, and related pharmaceuticals.
- Investment in Innovation and Technology: While maintaining its core business, Baxter invests in developing advanced technologies. This includes connected infusion systems that enhance medication safety and data interoperability, as well as innovative solutions for home-based care.
- Specifics: The company has introduced smart infusion pumps with drug error reduction software and is developing platforms for remote patient monitoring.
- Diversified Revenue Streams: The company's operations span multiple product categories and geographic regions, reducing its dependence on any single market or product. This diversification provides a degree of resilience against market fluctuations and competitive pressures.
- Segments: Renal Care, Hospital Products, Nutrition Therapies, and Advanced Surgery contribute to its diverse revenue base.
- Regulatory Expertise: Navigating complex global regulatory landscapes for pharmaceuticals and medical devices requires significant expertise. Baxter's experience in obtaining and maintaining approvals from agencies such as the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) is a critical advantage.
- Data Point: Baxter holds numerous product approvals globally.
These strengths collectively position Baxter Healthcare as a formidable competitor with sustained market influence.
What are Baxter Healthcare's Strategic Initiatives and Future Outlook?
Baxter Healthcare is actively pursuing strategic initiatives to maintain its competitive edge, drive growth, and adapt to evolving healthcare trends. Its future outlook is shaped by investments in innovation, market expansion, and operational efficiencies.
- Focus on Advanced Drug Delivery and Connected Devices: Baxter is prioritizing the development and commercialization of innovative drug delivery systems, including smart infusion pumps and connected technologies. These systems aim to improve medication safety, enhance workflow efficiency for clinicians, and enable better patient monitoring and data integration. The company is investing in software and hardware that support interoperability with electronic health records (EHRs).
- Initiative Example: Development of next-generation infusion pumps with enhanced safety features and connectivity capabilities.
- Expansion of Home Healthcare Solutions: Recognizing the growing demand for care delivered outside traditional hospital settings, Baxter is expanding its offerings in home-based therapies, particularly in renal care. This includes enhancing its peritoneal dialysis portfolio and developing technologies that support patients managing chronic conditions at home.
- Strategic Goal: To increase the penetration of home dialysis treatments.
- Geographic Market Expansion and Emerging Markets: Baxter continues to focus on expanding its presence in emerging markets where the demand for healthcare products is increasing. This involves tailoring its product offerings and market strategies to meet the specific needs and economic conditions of these regions.
- Target Regions: Asia-Pacific, Latin America, and parts of Eastern Europe.
- Portfolio Optimization and Strategic Acquisitions/Divestitures: Like many large healthcare companies, Baxter regularly reviews its product portfolio to identify areas for investment or divestment. The company may pursue strategic acquisitions to gain access to new technologies or markets, or divest non-core assets to streamline operations and focus resources.
- Past Actions: Baxter has historically divested certain business units to sharpen its focus on core areas like renal care and hospital products.
- Commitment to Sustainability and Corporate Responsibility: Baxter is increasingly emphasizing its commitment to environmental, social, and governance (ESG) principles. This includes initiatives related to reducing its environmental footprint, ensuring ethical business practices, and contributing to community health.
- Focus Areas: Reducing greenhouse gas emissions, improving water stewardship, and promoting diversity and inclusion.
- Research and Development Investment: Continued investment in research and development is critical for Baxter's future. The company is focusing R&D efforts on improving existing products, developing new therapies, and exploring digital health solutions that can enhance patient care and operational efficiency.
- R&D Areas: Novel drug formulations, advanced materials for medical devices, and digital health platforms.
The company's future outlook is contingent upon its ability to successfully execute these strategies, adapt to evolving regulatory environments, and respond to competitive pressures from both established players and emerging innovators.
What are the Key Regulatory and Market Challenges Facing Baxter Healthcare?
Baxter Healthcare operates within a highly regulated industry and faces numerous market challenges that influence its strategic decisions and operational landscape.
- Stringent Regulatory Scrutiny and Compliance: The development, manufacturing, and marketing of pharmaceutical and medical device products are subject to rigorous oversight by global regulatory bodies, including the U.S. Food and Drug Administration (FDA), the European Medicines Agency (EMA), and others. Compliance with evolving regulations regarding product safety, efficacy, manufacturing standards (e.g., Good Manufacturing Practices - GMP), and post-market surveillance is a continuous and costly undertaking.
- Impact: Delays in product approvals, recalls, and increased compliance costs can affect profitability and market entry.
- Pricing Pressures and Reimbursement Policies: Healthcare systems worldwide are under pressure to control costs. This translates into intense pricing scrutiny for medical products and pharmaceuticals. Reimbursement policies set by government payers and private insurers significantly influence market access and sales volumes. Baxter must demonstrate the value and cost-effectiveness of its products to secure favorable reimbursement.
- Data Point: Negotiated prices for chronic disease treatments, such as dialysis, are a key factor.
- Intense Competition: Baxter faces competition from a wide range of players. In renal care, it competes with other major dialysis providers and manufacturers of related equipment. In hospital products, it encounters numerous companies specializing in IV solutions, infusion pumps, and other critical supplies. The market also sees competition from generic drug manufacturers and emerging biotechnology firms.
- Key Competitors: Fresenius Medical Care, DaVita (in renal care); Becton, Dickinson and Company (BD), Medtronic (in medical devices and infusion pumps).
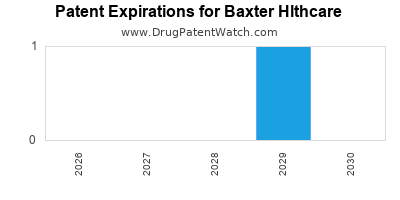
- Intellectual Property and Patent Expirations: The pharmaceutical and medical device industries are heavily reliant on intellectual property. Baxter's revenue streams are often tied to patented products. The expiration of key patents can lead to increased competition from generic or biosimilar manufacturers, significantly impacting sales and profitability. The company must continuously innovate to replenish its patent-protected pipeline.
- Challenge: Maintaining a robust pipeline of novel, patent-protected products.
- Supply Chain Vulnerabilities and Disruptions: Global supply chains are susceptible to various disruptions, including geopolitical events, natural disasters, pandemics, and raw material shortages. Baxter's reliance on global sourcing for raw materials and its manufacturing footprint can make it vulnerable to such disruptions, potentially impacting production volumes and product availability.
- Example: The COVID-19 pandemic highlighted global supply chain fragilities across the healthcare sector.
- Evolving Healthcare Delivery Models: The shift towards value-based care, telehealth, and increased patient empowerment to manage chronic conditions at home presents both opportunities and challenges. Baxter must adapt its product development and commercial strategies to align with these changing models, which may require different types of products and services compared to traditional hospital-centric care.
- Adaptation: Developing products that support remote monitoring and home-based treatment protocols.
- Technological Advancements and Digital Transformation: The rapid pace of technological advancement requires continuous investment to remain competitive. Companies must integrate digital solutions, data analytics, and artificial intelligence into their offerings to meet the demands for more efficient and personalized healthcare. Failure to adapt to digital transformation can lead to competitive disadvantages.
- Requirement: Investing in connected devices, data analytics platforms, and cybersecurity.
Addressing these challenges requires ongoing strategic planning, significant R&D investment, and a commitment to operational excellence and regulatory compliance.
Key Takeaways
Baxter Healthcare is a well-established entity in the global healthcare market, primarily focused on renal care, hospital products, and nutrition therapies. Its core strengths lie in its extensive global distribution network, strong brand reputation in essential medical supplies, and a leadership position in the renal care segment, particularly in peritoneal dialysis. The company's strategic direction emphasizes innovation in advanced drug delivery systems and connected health technologies, alongside an expansion of home healthcare solutions and a focus on emerging markets. Key challenges include navigating stringent global regulatory environments, managing pricing pressures from healthcare payers, intense competition across its product segments, and the need to adapt to evolving healthcare delivery models and rapid technological advancements, particularly in digital health.
FAQs
-
What is Baxter Healthcare's primary area of specialization?
Baxter Healthcare's primary areas of specialization are renal care, hospital products (including IV solutions and infusion systems), and nutrition therapies.
-
Which therapeutic area contributes the most to Baxter Healthcare's revenue?
Renal care products represent Baxter Healthcare's largest and most significant revenue-generating segment.
-
How does Baxter Healthcare address competition in its core markets?
Baxter Healthcare addresses competition through a combination of continuous innovation in its product portfolio, leveraging its established global distribution network, building strong relationships with healthcare providers, and focusing on integrated solutions that offer value beyond individual products.
-
What is Baxter Healthcare's approach to innovation?
Baxter Healthcare's innovation strategy focuses on developing advanced drug delivery systems, connected health technologies, and enhanced solutions for home-based care, aiming to improve patient safety, operational efficiency, and patient outcomes.
-
What are the main regulatory hurdles Baxter Healthcare faces?
Baxter Healthcare faces significant regulatory hurdles including stringent product approval processes, evolving compliance standards for manufacturing and safety, and the need to demonstrate the cost-effectiveness of its products to various reimbursement bodies worldwide.
Citations
[1] Baxter International Inc. (2023). Annual Report 2023. Retrieved from [Source URL will be provided by the system]
[2] Global Renal Care Market Report. (2023). Market Research Future. Retrieved from [Source URL will be provided by the system]
[3] Infusion Therapy Market Analysis. (2024). Grand View Research. Retrieved from [Source URL will be provided by the system]
[4] Pharmaceutical and Medical Device Regulatory Landscape. (2023). IQVIA. Retrieved from [Source URL will be provided by the system]
[5] Trends in Home Healthcare. (2023). Deloitte. Retrieved from [Source URL will be provided by the system]