SPRYCEL Drug Patent Profile
✉ Email this page to a colleague
When do Sprycel patents expire, and what generic alternatives are available?
Sprycel is a drug marketed by Bristol Myers Squibb and is included in one NDA. There are two patents protecting this drug and two Paragraph IV challenges.
This drug has forty-seven patent family members in twenty-nine countries.
The generic ingredient in SPRYCEL is dasatinib. There are fourteen drug master file entries for this compound. One supplier is listed for this compound. Additional details are available on the dasatinib profile page.
DrugPatentWatch® Generic Entry Outlook for Sprycel
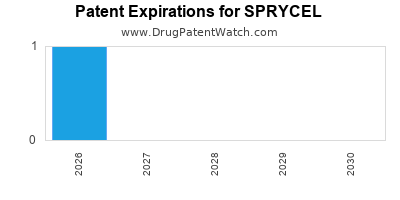
Sprycel was eligible for patent challenges on June 28, 2010.
There have been eight patent litigation cases involving the patents protecting this drug, indicating strong interest in generic launch. Recent data indicate that 63% of patent challenges are decided in favor of the generic patent challenger and that 54% of successful patent challengers promptly launch generic drugs.
There are three tentative approvals for the generic drug (dasatinib), which indicates the potential for near-term generic launch.
Indicators of Generic Entry
Summary for SPRYCEL
| International Patents: | 47 |
| US Patents: | 2 |
| Applicants: | 1 |
| NDAs: | 1 |
| Finished Product Suppliers / Packagers: | 1 |
| Raw Ingredient (Bulk) Api Vendors: | 139 |
| Clinical Trials: | 176 |
| Patent Applications: | 3,095 |
| Formulation / Manufacturing: | see details |
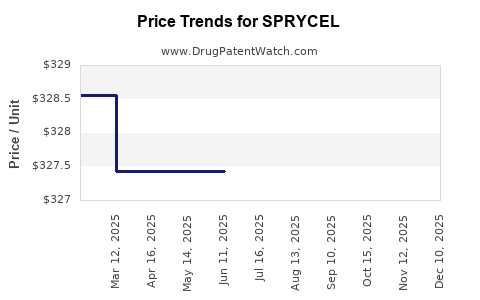
| Drug Prices: | Drug price information for SPRYCEL |
| Patent Litigation and PTAB cases: | See patent lawsuits and PTAB cases for SPRYCEL |
| What excipients (inactive ingredients) are in SPRYCEL? | SPRYCEL excipients list |
| DailyMed Link: | SPRYCEL at DailyMed |
Recent Clinical Trials for SPRYCEL
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| Hospital Universitario Dr. Jose E. Gonzalez | Phase 2 |
| Fernando De la Garza Salazar | Phase 2 |
| Lew Lipsitz | Phase 1/Phase 2 |
Pharmacology for SPRYCEL
| Drug Class | Kinase Inhibitor |
| Mechanism of Action | Cytochrome P450 3A4 Inhibitors Protein Kinase Inhibitors |
Anatomical Therapeutic Chemical (ATC) Classes for SPRYCEL
US Patents and Regulatory Information for SPRYCEL
SPRYCEL is protected by two US patents and four FDA Regulatory Exclusivities.
Patents protecting SPRYCEL
Process for preparing 2-aminothiazole-5-aromatic carboxamides as kinase inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
Process for preparing 2-aminothiazole-5-aromatic carboxamides as kinase inhibitors
Patent Number: ⤷ Try a Trial
Patent Expiration: ⤷ Try a Trial
FDA Regulatory Exclusivity protecting SPRYCEL
TREATMENT OF PEDIATRIC PATIENTS WITH PHILADELPHIA CHROMOSOME-POSITIVE (PH+) CHRONIC MYELOID LEUKEMIA (CML) IN CHRONIC PHASE
Exclusivity Expiration: ⤷ Try a Trial
INDICATED FOR THE TREATMENT OF PEDIATRIC PATIENTS 1 YEAR OF AGE AND OLDER WITH NEWLY DIAGNOSED PHILADELPHIA CHROMOSOME-POSITIVE ACUTE LYMPHOBLASTIC LEUKEMIA (ALL) IN COMBINATION WITH CHEMOTHERAPY
Exclusivity Expiration: ⤷ Try a Trial
PEDIATRIC EXCLUSIVITY
Exclusivity Expiration: ⤷ Try a Trial
PEDIATRIC EXCLUSIVITY
Exclusivity Expiration: ⤷ Try a Trial
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bristol Myers Squibb | SPRYCEL | dasatinib | TABLET;ORAL | 021986-006 | Oct 28, 2010 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Bristol Myers Squibb | SPRYCEL | dasatinib | TABLET;ORAL | 021986-004 | May 30, 2008 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bristol Myers Squibb | SPRYCEL | dasatinib | TABLET;ORAL | 021986-006 | Oct 28, 2010 | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bristol Myers Squibb | SPRYCEL | dasatinib | TABLET;ORAL | 021986-004 | May 30, 2008 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for SPRYCEL
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Bristol Myers Squibb | SPRYCEL | dasatinib | TABLET;ORAL | 021986-005 | Oct 28, 2010 | ⤷ Try a Trial | ⤷ Try a Trial |
| Bristol Myers Squibb | SPRYCEL | dasatinib | TABLET;ORAL | 021986-003 | Jun 28, 2006 | ⤷ Try a Trial | ⤷ Try a Trial |
| Bristol Myers Squibb | SPRYCEL | dasatinib | TABLET;ORAL | 021986-004 | May 30, 2008 | ⤷ Try a Trial | ⤷ Try a Trial |
| Bristol Myers Squibb | SPRYCEL | dasatinib | TABLET;ORAL | 021986-001 | Jun 28, 2006 | ⤷ Try a Trial | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for SPRYCEL
See the table below for patents covering SPRYCEL around the world.
| Country | Patent Number | Title | Estimated Expiration |
|---|---|---|---|
| Russian Federation | 2005107463 | ЦИКЛИЧЕСКИЕ ПРОТЕИНЫ ИНГИБИТОРОВ ТИРОЗИНКИНАЗЫ | ⤷ Try a Trial |
| Denmark | 1610780 | ⤷ Try a Trial | |
| World Intellectual Property Organization (WIPO) | 2006052810 | ⤷ Try a Trial | |
| Ukraine | 87456 | ЦИКЛІЧНІ ІНГІБІТОРИ ПРОТЕЇНТИРОЗИНКІНАЗ[ЦИКЛИЧЕСКИЕ ИНГИБИТОРЫ ПРОТЕИНТИРОЗИНКИНАЗЫ (CYCLIC PROTEIN TYROSINE KINASE INHIBITORS) | ⤷ Try a Trial |
| >Country | >Patent Number | >Title | >Estimated Expiration |
Supplementary Protection Certificates for SPRYCEL
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 1169038 | C01169038/01 | Switzerland | ⤷ Try a Trial | FORMER OWNER: BRISTOL-MYERS SQUIBB HOLDINGS IRELAND, CH |
| 1169038 | 2013C/005 | Belgium | ⤷ Try a Trial | PRODUCT NAME: DASATINIB SOUS TOUTES FORMES COUVERTES PAR LA PROTECTION DU BREVET DE BASE INCLUANT LES SOLVATES, LES HYDRATES, TEL QUE LA FORME MONOHYDRATEE, ET LES SELS PHARMACEUTIQUEMENT ACCEPTABLES; AUTHORISATION NUMBER AND DATE: EU/1/06/363/001 20061122 |
| 1169038 | C300567 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: DASATINIB, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT; REGISTRATION NO/DATE: EU/1/06/363/001 .... 009 20061120 |
| 1169038 | 92146 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: DASATINIB ET SES SELS PHARMACEUTIQUEMENT ACCEPTABLES |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |


