Last updated: February 19, 2026
What is the Current Market Position of SPRYCEL?
SPRYCEL (dasatinib) is a tyrosine kinase inhibitor approved for the treatment of Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) and Ph+ acute lymphoblastic leukemia (ALL). Its primary market position is as a second-line therapy for CML after failure of prior tyrosine kinase inhibitor (TKI) treatment, and as a first-line therapy for both CML and Ph+ ALL.
The global market for CML treatment is characterized by competition among several TKIs, including imatinib (Gleevec/Glivec), nilotinib (Tasigna), bosutinib (Bosulif), and ponatinib (Iclusig), alongside dasatinib. SPRYCEL's efficacy in achieving deep molecular responses and its broader activity spectrum compared to some earlier TKIs have supported its market penetration. However, the emergence of generic imatinib has significantly impacted the overall market dynamics, driving down treatment costs for the first-line setting and increasing pressure on branded therapies.
In the first-line CML setting, SPRYCEL competes directly with imatinib, nilotinib, and bosutinib. Clinical trial data, such as the DASISION trial, demonstrated comparable efficacy between SPRYCEL and imatinib in achieving major molecular response rates at 12 months, with a potentially faster achievement of response with SPRYCEL [1]. However, differences in tolerability profiles and long-term safety outcomes also influence physician prescribing habits and patient choice.
For the second-line CML indication, SPRYCEL is a well-established option following treatment failure with imatinib. Its ability to overcome certain imatinib-resistant mutations is a key differentiator. The market share in this segment is influenced by the availability of other second-generation TKIs and the evolving understanding of optimal treatment sequencing.
In Ph+ ALL, SPRYCEL is approved in combination with chemotherapy for newly diagnosed adult patients. This indication offers a distinct market segment, addressing a critical unmet need in a challenging hematological malignancy. The competition in Ph+ ALL treatment is less concentrated than in CML, with fewer approved TKIs specifically for this indication.
What is the Financial Performance of SPRYCEL?
SPRYCEL is a significant revenue generator for Bristol Myers Squibb (BMS). Its financial performance is influenced by prescription volumes, pricing strategies, patent exclusivity, and the competitive landscape.
| Year |
Net Sales (USD Billions) |
Percentage Change (%) |
| 2021 |
2.48 |
-3 |
| 2022 |
2.56 |
3.2 |
| 2023 |
2.65 |
3.5 |
Data source: Bristol Myers Squibb Annual Reports and SEC Filings.
The reported net sales indicate a steady revenue stream, with modest growth observed in recent years. This stability can be attributed to its established position in both first and second-line CML and its use in Ph+ ALL.
Key factors impacting SPRYCEL's financial trajectory include:
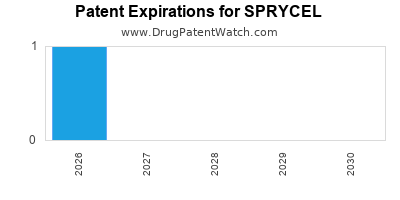
- Patent Expirations and Generic Competition: The primary and secondary patents protecting dasatinib have expired in major markets. Generic versions of dasatinib have entered the market, particularly in the United States and Europe, leading to price erosion and a decline in market share for the branded product in certain indications and geographies. For instance, generic dasatinib became available in the U.S. in late 2023. This is a significant headwind for future revenue growth of branded SPRYCEL.
- Pricing and Reimbursement: Pharmaceutical pricing is under continuous scrutiny globally. Payer negotiations and formulary placement play a critical role in determining net sales. As generic competition intensifies, pricing pressures are amplified, impacting profitability.
- Market Penetration and Expansion: Continued efforts to expand SPRYCEL's use in earlier lines of therapy and to gain approvals in new patient populations or geographical regions can drive revenue. However, the market for CML is relatively mature, with high awareness and established treatment pathways.
- Clinical Development and New Indications: While SPRYCEL has an established label, any new clinical trial data demonstrating improved outcomes or potential approval for novel applications could revitalize its market trajectory. However, the focus for many drug developers has shifted towards novel mechanisms of action for hematological malignancies.
- Competitive Landscape: The introduction of new TKIs or other novel therapies for CML and Ph+ ALL can directly impact SPRYCEL's market share and revenue. For example, advancements in CAR T-cell therapy for certain leukemias could indirectly affect the market for targeted therapies like SPRYCEL.
What are the Key Patent Expirations and Generic Entry Timelines?
Patent protection is a critical determinant of a drug's financial exclusivity. For SPRYCEL (dasatinib), the key patents have expired or are nearing expiration in major markets, paving the way for generic competition.
- United States: The primary U.S. compound patent for dasatinib expired. Generic versions of dasatinib became available in the U.S. in late 2023. BMS has historically engaged in patent litigation to defend its intellectual property, but the exclusivity period for the core compound has concluded.
- Europe: Similar to the U.S., the patent protection for dasatinib has expired in European Union member states. The availability of generics in Europe began prior to 2023 in many countries.
- Other Major Markets: Patent expirations in other significant pharmaceutical markets, such as Japan and Canada, have also occurred, leading to the introduction of generic dasatinib in those regions.
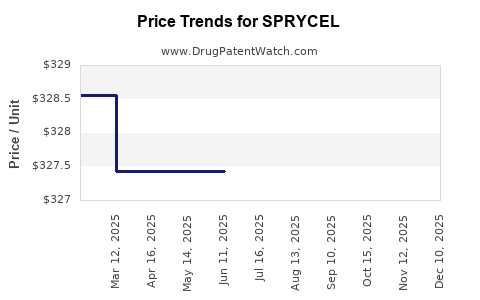
The timing of generic entry is crucial. Once generic versions are approved and marketed, they typically offer significant price reductions compared to the branded product, often ranging from 50% to 80% or more, depending on market dynamics and regulatory frameworks. This price erosion directly impacts the revenue generated by the branded drug.
Impact of Generic Entry:
- Sales Decline: The introduction of generics leads to a rapid decline in branded drug sales as prescribers and payers switch to the lower-cost alternative. This effect is often more pronounced in markets with strong generic uptake policies and lower pricing regulations.
- Market Share Shift: Generic manufacturers gain significant market share, capturing a substantial portion of the patient population previously treated with the branded drug.
- Pricing Pressure: Even for the branded product that remains on the market, the presence of generics forces significant price reductions to remain competitive, further impacting profitability.
- Lifecycle Management: Pharmaceutical companies often employ lifecycle management strategies, such as seeking new indications, developing new formulations, or combining drugs with other therapies, to extend the commercial life of a product beyond its initial patent exclusivity. However, for older molecules like dasatinib, these strategies may have limited long-term impact against a pervasive generic threat.
The transition from branded exclusivity to generic competition represents a significant shift in the financial trajectory of SPRYCEL, moving from a high-margin product to a more commoditized one.
What is the Competitive Landscape for SPRYCEL?
The competitive landscape for SPRYCEL is dynamic and multifaceted, involving established TKIs, emerging therapies, and generic alternatives.
Key Competitors in Chronic Myeloid Leukemia (CML):
- Imatinib (Gleevec/Glivec): The first-generation TKI, now largely available as a generic, remains a foundational therapy for CML, particularly in the first-line setting. Its long track record and low cost (for generics) make it a persistent competitor.
- Nilotinib (Tasigna): A second-generation TKI, nilotinib is approved for both first- and second-line CML treatment. It offers a different efficacy and tolerability profile compared to imatinib and dasatinib and has demonstrated superior outcomes in certain patient subgroups in head-to-head trials against imatinib [2].
- Bosutinib (Bosulif): Another second-generation TKI, bosutinib is approved for both first- and second-line CML. It is known for its efficacy in overcoming certain mutations resistant to earlier TKIs.
- Ponatinib (Iclusig): A third-generation TKI, ponatinib is approved for CML and Ph+ ALL in patients resistant to or intolerant of prior TKI therapy, including those with the T315I mutation. Its use is often reserved for more challenging or resistant cases due to its cardiovascular risk profile.
- Asciminib (Scemblix): A STAMP inhibitor, asciminib represents a novel mechanism of action for CML treatment. It is approved for patients with chronic phase CML previously treated with two or more TKIs and for those with the T315I mutation. Asciminib offers a distinct therapeutic option, particularly for patients who have failed or are intolerant to other TKIs [3].
Competition in Philadelphia Chromosome-Positive Acute Lymphoblastic Leukemia (Ph+ ALL):
- Ponatinib (Iclusig): Approved in combination with chemotherapy for newly diagnosed adult patients with Ph+ ALL.
- Nilotinib (Tasigna): Also approved in combination with chemotherapy for newly diagnosed adult patients with Ph+ ALL.
- Imatinib (Gleevec/Glivec): May be used in certain Ph+ ALL treatment regimens, although less frequently as a primary TKI component compared to newer agents.
Impact of Generic Dasatinib:
The most significant competitive force impacting branded SPRYCEL is the entry of generic dasatinib. This directly erodes market share and pricing power for the originator product across all approved indications where generics are available. Generic competition forces branded manufacturers to re-evaluate their pricing, market access strategies, and potential for product differentiation through new formulations or indications, though the latter is less feasible for older molecules.
Other Emerging Therapeutic Modalities:
- Allogeneic Stem Cell Transplantation (Allo-SCT): Remains a curative option for a subset of CML patients, particularly younger individuals with high-risk disease.
- Chimeric Antigen Receptor (CAR) T-cell Therapy: While not a primary treatment for CML, CAR T-cell therapy is a rapidly advancing modality in hematological malignancies and could impact the treatment landscape for certain types of ALL, indirectly affecting the market for TKIs in that disease space.
The competitive environment for SPRYCEL is characterized by a mix of established TKIs, novel agents with different mechanisms of action, and the increasing prevalence of generic alternatives, all of which exert downward pressure on its market share and financial performance.
What are the Future Market Projections for SPRYCEL?
Forecasting the future market trajectory of SPRYCEL involves considering the interplay of patent expirations, generic competition, evolving treatment paradigms, and ongoing clinical developments.
Key Factors Influencing Future Projections:
- Generic Erosion: The most significant driver of SPRYCEL's future market performance will be the continued impact of generic dasatinib. As generic penetration increases across all major markets, sales of branded SPRYCEL are projected to decline substantially. This decline will be more pronounced in regions where generic uptake is rapid and pricing pressures are intense.
- Life Cycle Management and Geographic Expansion: Bristol Myers Squibb may attempt to mitigate revenue losses through life cycle management strategies. This could include efforts to expand SPRYCEL's use in specific patient subpopulations, explore combination therapies, or focus on markets with delayed generic entry. However, the impact of these strategies is likely to be limited against broad generic availability.
- First-Line Market Share: In the first-line CML setting, generic imatinib and other TKIs like nilotinib and bosutinib, alongside generic dasatinib, will exert significant competitive pressure. The historical trend of TKIs moving to earlier lines of therapy may be curtailed for branded SPRYCEL due to cost considerations and the availability of generics.
- Second-Line and Refractory Disease: SPRYCEL will likely retain a role in the second-line CML setting and for patients with specific mutations where generic alternatives might be less immediately dominant. However, the emergence of novel agents like asciminib, with a distinct mechanism, offers a new alternative for patients who have failed prior therapies, potentially limiting SPRYCEL's long-term market share in these niches.
- Ph+ ALL Market: The Ph+ ALL indication provides a distinct market segment. The success of combination therapies involving SPRYCEL and chemotherapy will influence its trajectory here. However, this market is smaller than CML and also faces competition from other TKIs like ponatinib and nilotinib.
- Pricing Adjustments: To compete with generics and manage market access, Bristol Myers Squibb will likely implement significant price adjustments for branded SPRYCEL in markets where generics are available. This will reduce the average selling price and profit margins.
Quantitative Outlook:
While specific sales projections are proprietary to pharmaceutical companies and market research firms, the general trend for branded drugs post-patent expiration is a sharp decline in revenue. For SPRYCEL, the impact of generic competition is expected to lead to a significant year-over-year decrease in net sales starting from 2024, with sales potentially stabilizing at a much lower level in the mid-to-long term as it transitions to a smaller, niche market share primarily within generic-dominated landscapes. The cumulative impact of generic entry is projected to reduce branded SPRYCEL sales by over 70-80% within 3-5 years of widespread generic availability in major markets.
The company's strategic focus will likely shift towards its newer pipeline assets and established blockbuster drugs, with SPRYCEL becoming a legacy product with a diminishing but still present revenue stream from remaining branded prescriptions and markets.
Key Takeaways
- SPRYCEL (dasatinib) is an established TKI for CML and Ph+ ALL, facing significant generic competition following patent expirations.
- Net sales for SPRYCEL have been in the range of $2.5-2.6 billion annually, but this is expected to decline sharply due to generic entry.
- Key patent expirations for dasatinib have occurred in major markets, with generic versions available in the U.S. since late 2023 and in Europe prior to that.
- The competitive landscape includes imatinib (generic), nilotinib, bosutinib, ponatinib, and the novel STAMP inhibitor asciminib. Generic dasatinib is now a primary competitor.
- Future market projections indicate a substantial decline in branded SPRYCEL sales due to generic erosion, with a transition to a niche market presence.
Frequently Asked Questions
- When did generic versions of dasatinib become available in the United States?
Generic versions of dasatinib became available in the United States in late 2023.
- What is the primary indication for SPRYCEL?
SPRYCEL is approved for the treatment of Philadelphia chromosome-positive (Ph+) chronic myeloid leukemia (CML) and Ph+ acute lymphoblastic leukemia (ALL).
- How does asciminib (Scemblix) compete with SPRYCEL?
Asciminib represents a novel mechanism of action (STAMP inhibitor) and offers an alternative treatment option, particularly for patients who have failed or are intolerant to prior TKIs, thereby competing for market share in refractory CML.
- What is the expected impact of generic competition on branded SPRYCEL sales?
Generic competition is expected to lead to a significant and rapid decline in branded SPRYCEL sales due to price erosion and market share shift to lower-cost generic alternatives.
- Is SPRYCEL still considered a first-line treatment option for CML?
Yes, SPRYCEL is approved for first-line treatment of CML, but its market share in this setting is increasingly challenged by generic imatinib and other branded TKIs, as well as generic dasatinib itself.
Citations
[1] Kantarjian, H. M., Shah, N. P., Cortes, J., Apperley, J. F., Khoury, H. J., Duval, M., ... & Larson, R. A. (2012). Dasatinib versus imatinib in newly diagnosed chronic-phase chronic myeloid leukemia: 2-year follow-up of the randomized phase 3 DASISION trial. Blood, 119(24), 5637-5641.
[2] Hochhaus, A., Larson, R. A., Guilhot, F., Radich, J. P., Branford, S., Khoury, H. J., ... & Müller, M. C. (2016). Long-term outcomes of patients with chronic myeloid leukemia treated for first-line therapy with nilotinib or imatinib: 5-year results from the randomized ENESTnd trial. Leukemia, 30(5), 1044-1051.
[3] Cortes, J. E., Kim, D. W., Yeung, D. T., Xiao, Z., Chen, X., Ghiu, A., ... & Hochhaus, A. (2022). Asciminib, the first-in-class STAMP inhibitor, in chronic myeloid leukemia after ATP-competitive tyrosine kinase inhibitor failure: the phase 3 PACE trial. Cancer, 128(21), 3774-3785.