Last updated: February 19, 2026
Sildenafil citrate, primarily known for its brand-name formulation Viagra, holds a significant position in the pharmaceutical market for treating erectile dysfunction (ED) and, in specific formulations, pulmonary arterial hypertension (PAH) under the brand name Revatio. The market for sildenafil citrate is characterized by established brand recognition, extensive patent litigation, and a mature generic landscape. Its financial trajectory is influenced by market penetration, competition from other phosphodiesterase type 5 (PDE5) inhibitors, and the ongoing development of alternative treatments.
What is the Market Size and Growth Projection for Sildenafil Citrate?
The global market for sildenafil citrate has matured considerably. While precise, up-to-the-minute market size figures are proprietary and fluctuate, industry estimates place the global market for ED treatments, where sildenafil citrate is a dominant player, in the billions of U.S. dollars annually. The U.S. market alone for ED drugs has historically been valued at several billion dollars.
Growth projections for sildenafil citrate are generally modest, reflecting its status as a well-established, off-patent medication. The growth is primarily driven by:
- Aging Global Population: Increased prevalence of conditions associated with ED in aging demographics.
- Growing Awareness and Reduced Stigma: Increased patient and physician awareness of ED as a treatable condition, alongside a gradual reduction in societal stigma.
- Emergence of Generic Competition: The widespread availability of affordable generic sildenafil citrate has increased accessibility and market volume, even if it has reduced revenue for the originator.
- Potential for New Indications: While less prominent for sildenafil citrate, ongoing research into new therapeutic applications for PDE5 inhibitors could influence future market dynamics.
The PAH segment, while smaller than the ED market, represents a stable revenue stream for sildenafil citrate (as Revatio). The U.S. Food and Drug Administration (FDA) approved Revatio for PAH in June 2005 [1]. The PAH market is influenced by factors such as diagnostic rates, treatment guidelines, and the development of competing PAH therapies.
The overall growth rate for the sildenafil citrate market is projected to be in the low single digits annually, largely reflecting the volume-driven nature of the generic market rather than significant price appreciation.
What are the Key Intellectual Property and Patent Expirations for Sildenafil Citrate?
The intellectual property landscape for sildenafil citrate has been extensively litigated. The original patent for sildenafil citrate, filed by Pfizer Inc., expired in the United States in April 2020 [2]. This marked the end of market exclusivity for the branded product, Viagra.
Prior to this, Pfizer engaged in significant patent litigation to defend its exclusivity. Key aspects of the IP strategy and litigation included:
- Original Compound Patents: These covered the sildenafil molecule itself.
- Formulation Patents: Patents related to specific formulations of sildenafil, including those designed for improved delivery or efficacy.
- Method of Use Patents: Patents covering the use of sildenafil for specific medical conditions, such as ED and PAH.
- Patent Linkage (Orange Book): The FDA's "Orange Book" lists patents that cover approved drug products. Disputes over these patents often determine the timeline for generic market entry.
The expiration of the primary patents has led to:
- Widespread Generic Entry: Numerous pharmaceutical companies now manufacture and market generic versions of sildenafil citrate.
- Increased Price Competition: Generic competition has dramatically reduced the average selling price of sildenafil citrate compared to the branded Viagra.
- Continued Legal Challenges: While the core patents have expired, disputes can arise over secondary patents, formulation patents, or manufacturing processes.
The patent expiration timeline for sildenafil citrate represents a significant shift from a monopoly market to a highly competitive generic environment.
Who are the Major Market Players and Competitors?
The competitive landscape for sildenafil citrate is divided into the originator (Pfizer) and a broad spectrum of generic manufacturers.
Originator:
- Pfizer Inc.: Holds the original patents and marketed Viagra (for ED) and Revatio (for PAH). While facing generic competition for Viagra, Revatio continues to be a key product.
Major Generic Manufacturers:
The market is highly fragmented with numerous companies producing generic sildenafil citrate. These include, but are not limited to:
- Teva Pharmaceutical Industries Ltd.
- Mylan N.V. (now part of Viatris Inc.)
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Cipla Ltd.
- Accord Healthcare (part of Intas Pharmaceuticals)
- Amneal Pharmaceuticals LLC
These companies compete primarily on price, manufacturing efficiency, and distribution networks. Their market share is dynamic, influenced by regulatory approvals, production capacity, and market access strategies.
Competitors within the PDE5 Inhibitor Class:
Beyond generic sildenafil citrate, the market for ED treatments includes other PDE5 inhibitors, which represent direct therapeutic competition:
- Tadalafil (Cialis by Eli Lilly and Company): Known for its longer duration of action. The U.S. patent for Cialis expired in September 2018, leading to generic availability [3].
- Vardenafil (Levitra by Bayer AG, Staxyn by GSK): Also a PDE5 inhibitor with a distinct pharmacokinetic profile.
- Avanafil (Stendra by Vivus Inc.): A newer generation PDE5 inhibitor marketed for faster onset of action.
These branded and generic competitors offer alternative treatment options, influencing prescribing patterns and patient choice.
What are the Regulatory and Clinical Landscape Considerations?
The regulatory and clinical landscape for sildenafil citrate is well-established, given its long history on the market.
Regulatory Approvals:
- U.S. FDA: Approved Viagra for erectile dysfunction in March 1998. Approved Revatio for pulmonary arterial hypertension in June 2005 [1]. Generic sildenafil citrate for both indications is also approved.
- European Medicines Agency (EMA): Approved Viagra in Europe in 1998. Generic versions are also available.
- Other Jurisdictions: Sildenafil citrate is approved for ED and PAH in numerous countries worldwide, with regulatory pathways varying by region.
Clinical Landscape:
- Erectile Dysfunction (ED): Sildenafil citrate is a first-line oral therapy for ED. Its efficacy is well-documented, with clinical trials demonstrating significant improvements in erectile function. Treatment guidelines generally recommend PDE5 inhibitors as the initial pharmacological approach. Factors influencing treatment choice include patient comorbidities, co-administered medications (potential for drug interactions), and patient preference regarding onset of action and duration.
- Pulmonary Arterial Hypertension (PAH): Revatio is used to improve exercise ability in patients with PAH. It is part of a broader therapeutic landscape for PAH, which includes prostacyclin analogs, endothelin receptor antagonists, and other PDE inhibitors. Treatment decisions for PAH are complex and guided by patient severity, disease etiology, and response to therapy.
- Safety Profile: Sildenafil citrate has a well-characterized safety profile. Common side effects include headache, flushing, dyspepsia, nasal congestion, and visual disturbances. Contraindications include concomitant use of nitrates due to the risk of severe hypotension. Drug interactions, particularly with CYP3A4 inhibitors, are also a key consideration.
- Off-Label Use: While primarily approved for ED and PAH, sildenafil citrate has been investigated and used off-label for other conditions, such as Raynaud's phenomenon and high-altitude pulmonary edema. However, these uses do not contribute significantly to the overall market value or revenue.
The regulatory environment favors generic competition following patent expiry, ensuring broad access. Clinical guidelines continue to position sildenafil citrate as a cornerstone therapy in its approved indications.
What are the Financial Performance and Revenue Drivers?
The financial performance of sildenafil citrate has evolved significantly due to patent expirations and the rise of generic competition.
Branded Product (Viagra):
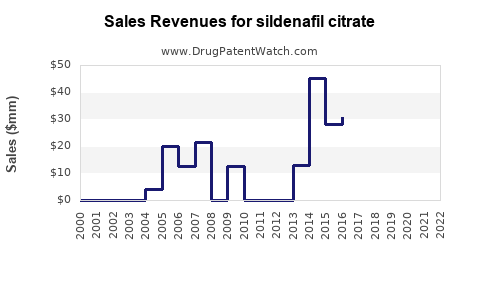
- Peak Revenue: Prior to patent expiry, Viagra was a blockbuster drug for Pfizer, generating billions of dollars in annual revenue. For instance, in 2011, Viagra sales were approximately $1.9 billion [4].
- Post-Exclusivity Decline: Following the introduction of generic sildenafil citrate, branded Viagra sales have experienced a substantial decline. Pfizer still markets Viagra, but its revenue is now considerably lower, primarily driven by brand loyalty and specific market segments.
- Revatio Revenue: Revatio, marketed for PAH, continues to generate revenue for Pfizer, although this market segment is smaller than ED.
Generic Sildenafil Citrate:
- Volume-Driven Market: The revenue generated by generic sildenafil citrate is largely driven by sales volume rather than price. The low cost of generic alternatives makes sildenafil citrate highly accessible to a broad patient population.
- Manufacturer Revenue: The collective revenue generated by all generic manufacturers is substantial, reflecting the high prescription volume. However, individual generic manufacturers achieve profitability through efficient production and high-volume sales.
- Pricing Dynamics: Generic pricing is highly competitive. Manufacturers must balance production costs with market prices to maintain margins. Price erosion is common as more generic players enter the market.
- Distribution Channels: Revenue is distributed across various channels, including retail pharmacies, mail-order pharmacies, and hospital formularies.
Key Financial Drivers:
- Prescription Volume: The primary driver for generic sildenafil citrate revenue is the number of prescriptions filled.
- Pricing: While prices are low, minor fluctuations in generic pricing can impact overall market revenue.
- Market Access: The ability of manufacturers to secure formulary placement and favorable reimbursement from payers is critical for sales volume.
- Global Reach: The expansion of healthcare access and increasing diagnoses in emerging markets contribute to global sales volume.
The financial trajectory for sildenafil citrate has shifted from high-margin branded sales to high-volume, lower-margin generic sales.
What are the Future Outlook and Emerging Trends?
The future outlook for sildenafil citrate is characterized by continued market stability in its established indications, with potential for modest growth driven by demographic factors and increasing awareness.
Key Outlook Factors:
- Sustained Generic Dominance: Generic sildenafil citrate will remain the primary offering for ED treatment due to its affordability and established efficacy.
- Demographic Tailwinds: The aging global population will continue to support demand for ED treatments.
- Technological Advancements in Delivery: While not a significant trend for sildenafil citrate currently, innovation in drug delivery systems could potentially emerge for PDE5 inhibitors, though this is more likely to impact newer compounds.
- Competition from Other PDE5 Inhibitors: Tadalafil and other PDE5 inhibitors will continue to compete, with generic versions of these drugs also readily available.
- Emerging Therapies for ED: Research into non-pharmacological treatments for ED, such as shockwave therapy and regenerative medicine approaches, may offer alternatives, but these are unlikely to displace oral medications in the near to medium term for the majority of patients.
- PAH Market Dynamics: The Revatio market for PAH will remain subject to the development of new, potentially more targeted PAH therapies. However, sildenafil citrate's established efficacy and cost-effectiveness position it to remain a treatment option, particularly in certain patient populations or healthcare systems.
- Potential for Repurposing: While less likely for sildenafil citrate given its wide availability, continued research into PDE5 inhibition mechanisms could uncover new therapeutic avenues for related compounds.
Emerging Trends:
- Digital Health and Telemedicine: Increased adoption of telemedicine platforms may facilitate easier access to prescriptions for ED medications, potentially increasing prescription volumes.
- Focus on Cardiovascular Health: Growing recognition of the link between ED and cardiovascular disease may lead to more comprehensive patient screening and management, indirectly supporting the market for ED treatments.
- Supply Chain Resilience: Global events have highlighted the importance of robust pharmaceutical supply chains, a factor that will remain critical for generic manufacturers.
The market for sildenafil citrate is expected to remain a significant segment of the pharmaceutical industry, characterized by consistent demand and fierce generic competition.
Key Takeaways
- Sildenafil citrate (Viagra, Revatio) is a mature pharmaceutical product with a well-established market for erectile dysfunction (ED) and pulmonary arterial hypertension (PAH).
- The U.S. patent for Viagra expired in April 2020, leading to widespread generic competition and a shift from a branded monopoly to a highly price-competitive generic market.
- The global market for sildenafil citrate is driven by volume rather than price, with modest growth projected due to an aging population and increasing awareness of ED.
- Key competitors include Pfizer (originator) and numerous generic manufacturers such as Teva, Viatris, and Dr. Reddy's. Direct therapeutic competition comes from other PDE5 inhibitors like tadalafil and vardenafil.
- Regulatory approval and well-documented clinical efficacy in ED and PAH solidify sildenafil citrate's position in treatment guidelines.
- Future outlook points to sustained generic dominance, driven by consistent demand and demographic trends, with telemedicine potentially influencing prescription access.
Frequently Asked Questions
-
When did the main patent for Viagra expire in the United States?
The main patent for sildenafil citrate (Viagra) expired in the United States in April 2020.
-
What are the primary medical conditions for which sildenafil citrate is approved?
Sildenafil citrate is approved for the treatment of erectile dysfunction (ED) under the brand name Viagra and for pulmonary arterial hypertension (PAH) under the brand name Revatio.
-
How has the expiration of sildenafil citrate patents affected its market price?
The expiration of patents has led to significant price reductions due to the entry of generic manufacturers, making sildenafil citrate much more affordable.
-
Are there other drugs in the same class as sildenafil citrate for treating ED?
Yes, other phosphodiesterase type 5 (PDE5) inhibitors are available for treating ED, including tadalafil (Cialis) and vardenafil (Levitra).
-
What is the projected growth rate for the sildenafil citrate market?
The market for sildenafil citrate is projected to experience low single-digit annual growth, primarily driven by volume.
Citations
[1] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA Website - specific approval details are publicly available through FDA drug database searches, e.g., Revatio approval history.]
[2] U.S. Patent and Trademark Office. (n.d.). Patent Search. Retrieved from [USPTO Website - public patent records for sildenafil citrate.]
[3] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from [FDA Website - specific approval details for tadalafil (Cialis) generic entry.]
[4] Pfizer Inc. (2012). Pfizer Reports Fourth Quarter and Full-Year 2011 Results. Retrieved from [Pfizer Investor Relations Website - historical financial reports.]