Last updated: April 24, 2026
What is enalapril maleate and where does it sit in the market?
Enalapril maleate is an angiotensin-converting enzyme (ACE) inhibitor used for chronic cardiovascular conditions, most commonly hypertension and heart failure. In market terms, it is a mature, off-patent, multi-manufacturer generic product class where pricing is driven by supply capacity, reimbursement rules, tendering, and country-specific generic penetration rather than by brand-level differentiation.
Therapeutic positioning
- Primary uses: hypertension; congestive heart failure (and related indications per label).
- Drug class: ACE inhibitor (oral small molecule).
- Competitive landscape: generic ACE inhibitors (e.g., lisinopril, ramipril, captopril) and within-class switching under formularies.
Market consequence of maturity
- Long commercial history, high manufacturing replication, and frequent price pressure.
- Revenue and profitability depend on:
- Geographies with stable reimbursement
- Managed entry and tender outcomes
- Contract manufacturing scale and low-cost supply
- Regulatory stability (manufacturing compliance, supply continuity)
- Mix of strengths, pack sizes, and channels (retail, hospital, public tenders)
How does generic competition shape pricing and volume?
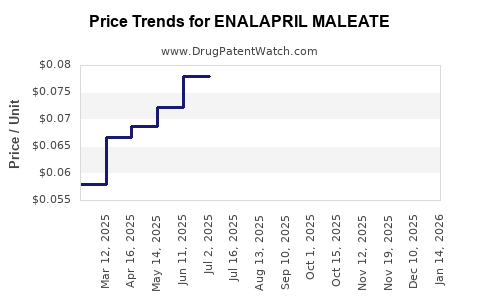
Enalapril maleate’s financial trajectory tracks the generic “race to the bottom,” then stabilizes when consolidation and tender mechanics reduce the number of profitable suppliers.
Typical pricing dynamics in mature generics
- Initial post-launch period: price compression after generic entries and pharmacy substitution.
- Mid-life: tender-driven price steps as public payers negotiate multi-source bids.
- Late-life: stabilization occurs when:
- Fewer manufacturers remain competitive at tender price points
- Some suppliers exit or throttle supply due to margin erosion
- Product presentation (strengths, blister formats) and regional licensing become binding
Competitor set and substitution
- Within-class substitution is common because ACE inhibitors are therapeutically substitutable in many payer formularies.
- Switching drivers:
- Reimbursement differentials
- Hospital formularies and guideline alignment
- Stability of supply and lead times
- Patient adherence factors tied to dosing frequency and tablet formulation
What are the key demand drivers for enalapril maleate?
Demand for enalapril maleate is steady and largely inelastic because it treats chronic conditions that require ongoing therapy.
Demand supports
- Persistent burden of cardiovascular disease (hypertension and heart failure)
- Chronic prescribing patterns (patients remain on therapy for long periods)
- Multiple line-of-therapy roles:
- Hypertension management
- Heart failure management, often as part of combination regimens
Demand headwinds
- Mortality and disease management improvements that alter incidence over time
- Substitution to other ACE inhibitors or ARBs depending on tolerability and formulary rules
- Regulatory changes affecting procurement (tender frequency, price ceilings, reference pricing)
How does the supply chain and manufacturing footprint affect margins?
In an off-patent generic, financial outcome is frequently determined by cost position and manufacturing uptime.
Supply-chain determinants
- Raw material availability and API cost fluctuations
- Finished dose manufacturing efficiency and batch release performance
- Compliance events that restrict supply and force price resets
- Contract scale and procurement terms (especially for wholesalers and tender aggregators)
Margin math in this category
- As price declines, only manufacturers with:
- Lowest landed cost of API and excipients
- Consistent yields and high OEE (overall equipment effectiveness)
- Tight quality systems that reduce batch rejection and recall risk
- Strong procurement leverage
remain profitable.
What does the regulatory status imply for commercial trajectory?
Enalapril maleate is widely marketed as a generic medicine globally. In such categories, “financial trajectory” typically reflects life-cycle effects: entry, consolidation, price stabilization, and periodic tender resets.
Regulatory features that shape business outcomes
- Market access depends on:
- Bioequivalence and quality dossier strength
- Ongoing GMP compliance and site approvals
- Labeling consistency with country-specific requirements
- Switching and substitution rely on:
- Pharmacy substitution rules
- Formulary inclusion and reimbursement categories
- Reference pricing or therapeutic equivalence frameworks
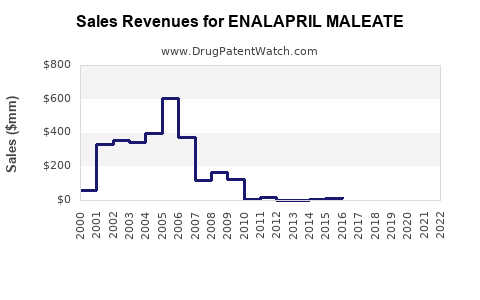
How do market dynamics translate into financial performance patterns?
While exact company-level revenue figures vary by geography and manufacturer, generic cardiovascular products show consistent financial patterns:
1) Revenue stability, margin compression
- Revenue tends to remain relatively stable because demand is chronic.
- Operating margin compresses as:
- New entrants lower prices
- Payers impose reference pricing and price caps
- Tender competition intensifies
2) Volume growth depends on procurement wins
- Volume increases usually come from:
- Winning tenders
- Gaining preferred supplier status
- Expanding strength and pack variants that meet formulary and dispensing preferences
3) Margin recovery is episodic
- Margin can improve when:
- Competitors exit or fail quality thresholds
- Supply shortages temporarily lift pricing
- Distribution contracts shift mix toward higher-margin pack formats or strengths
What are the realistic financial trajectory scenarios for enalapril maleate?
For business planning, generic ACE inhibitors typically fall into one of three trajectory patterns:
Scenario A: Continuous price erosion with stable volume
- Market remains multi-supplier.
- Procurement price floors decline gradually.
- Revenue tracks population and prescribing volume growth slowly.
Scenario B: Consolidation leading to price stabilization
- Fewer suppliers remain at tender economics.
- Buyers still require competition, but fewer bidders win.
- Revenue stabilizes; margin improves relative to worst-case erosion.
Scenario C: Supply disruptions create short-term price spikes
- Batch rejections, GMP actions, API disruptions, or logistics issues reduce supply.
- Pricing temporarily increases before competition returns and resets prices.
What competitive levers drive results for manufacturers of enalapril maleate?
In a generic class, differentiation is mostly operational, not clinical.
Manufacturers’ primary levers
- Cost-down roadmap on API and finished goods
- Tender strategy tied to:
- regional distribution strength
- lead time reliability
- pack format alignment with procurement specs
- Portfolio approach:
- multi-strength coverage
- bundling with other cardiovascular generics to sell through accounts
- Quality and compliance track record to prevent supply interruptions
Where are the economic pressure points likely to appear?
Even in mature generic markets, specific pressure points repeatedly affect P&L:
Price and reimbursement pressure
- Reference pricing revisions
- Annual tender renegotiations
- Changes in list pricing and reimbursement caps
- Increased scrutiny on “lowest priced equivalent” procurement
Regulatory and supply risk
- GMP enforcement actions at manufacturing sites
- API supply concentration and upstream constraints
- Shelf-life management and distribution losses
What does the product lifecycle look like for enalapril maleate?
Enalapril maleate fits the mature stage where:
- New patent-driven entrants are not the core story.
- Commercial focus shifts to manufacturing scale, compliance, tender participation, and cost leadership.
- Financial trajectory is cyclical and contract-driven rather than innovation-driven.
How should investors and operators read the market signal?
For underwriting or capacity planning, the market signal for enalapril maleate is:
- Demand is durable and predictable.
- Pricing is volatile mainly around tender and competitive entry cycles.
- Profitability is tied to supply reliability and unit cost discipline.
Key Takeaways
- Enalapril maleate is a mature generic ACE inhibitor where commercial value is determined by tender mechanics, generic substitution, and manufacturing cost position.
- Financial trajectory typically shows stable-to-slow revenue growth with persistent margin compression, punctuated by episodic margin improvement during competitor exit or supply constraints.
- Competitive advantage is operational: lowest landed cost, highest quality reliability, and procurement alignment across strengths and pack formats.
- Market outcomes are geography-specific because reference pricing and tender rules vary by country and procurement body.
FAQs
1) Is enalapril maleate a branded market product or a generic market product?
It is predominantly a generic market product with multiple manufacturers and pharmacy substitution dynamics.
2) What most influences pricing for enalapril maleate?
Tender outcomes, reference pricing, and generic entry competition determine pricing far more than brand-level factors.
3) What determines profitability for manufacturers?
Unit cost (API and finished goods), manufacturing uptime and yield, batch release reliability, and ability to win procurement contracts.
4) Does demand decline over time?
Demand tends to stay resilient because indications are chronic; the main risk is substitution to other ACE inhibitors or ARBs and reimbursement tightening rather than loss of disease need.
5) What is the most likely pattern investors should expect?
Stable volume with margin pressure, plus short-term financial spikes only when supply tightness or competitor withdrawals reduce bidding intensity.
References
[1] FDA Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. U.S. Food and Drug Administration. (Accessed via FDA Orange Book database).
[2] MedlinePlus: Enalapril. U.S. National Library of Medicine.
[3] EMA: Enalapril-containing medicines (product-related public information). European Medicines Agency.
[4] WHO Model List of Essential Medicines: ACE inhibitors for hypertension and heart failure (class positioning). World Health Organization.