Pfizer Company Profile
✉ Email this page to a colleague
What is the competitive landscape for PFIZER, and what generic alternatives to PFIZER drugs are available?
PFIZER has one hundred and ninety-five approved drugs.
There are forty-nine US patents protecting PFIZER drugs. There is one tentative approval on PFIZER drugs.
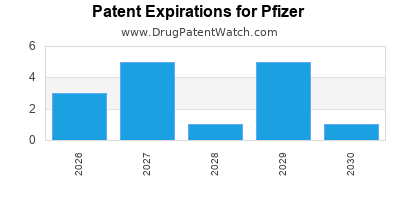
There are one thousand and twenty-four patent family members on PFIZER drugs in seventy countries and two hundred and sixty supplementary protection certificates in nineteen countries.
Summary for Pfizer
| International Patents: | 1024 |
| US Patents: | 49 |
| Tradenames: | 159 |
| Ingredients: | 130 |
| NDAs: | 195 |
| Drug Master File Entries: | 3 |
| Patent Litigation for Pfizer: | See patent lawsuits for Pfizer |
| PTAB Cases with Pfizer as petitioner: | See PTAB cases with Pfizer as petitioner |
Drugs and US Patents for Pfizer
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pfizer | HEPARIN SODIUM | heparin sodium | INJECTABLE;INJECTION | 201370-002 | Jul 21, 2011 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Pfizer | UNASYN | ampicillin sodium; sulbactam sodium | INJECTABLE;INJECTION | 062901-001 | Nov 23, 1988 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Pfizer | TALZENNA | talazoparib tosylate | CAPSULE;ORAL | 211651-006 | Jun 20, 2023 | RX | Yes | No | 8,420,650 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Pfizer | GLUCOTROL | glipizide | TABLET;ORAL | 017783-002 | May 8, 1984 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Pfizer
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Pfizer | TROVAN | trovafloxacin mesylate | TABLET;ORAL | 020759-001 | Dec 18, 1997 | 5,164,402 | ⤷ Try a Trial |
| Pfizer | COVERA-HS | verapamil hydrochloride | TABLET, EXTENDED RELEASE;ORAL | 020552-001 | Feb 26, 1996 | 5,200,196 | ⤷ Try a Trial |
| Pfizer Pharms | ACCURETIC | hydrochlorothiazide; quinapril hydrochloride | TABLET;ORAL | 020125-003 | Dec 28, 1999 | 4,344,949*PED | ⤷ Try a Trial |
| Pfizer | ZAVZPRET | zavegepant hydrochloride | SPRAY, METERED;NASAL | 216386-001 | Mar 9, 2023 | 7,314,883 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for PFIZER drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2016-11-07 |
| ➤ Subscribe | Delayed-release Tablets | 50 mg/0.2 mg | ➤ Subscribe | 2009-06-29 |
| ➤ Subscribe | Extended-release Tablets | 11 mg | ➤ Subscribe | 2016-11-07 |
| ➤ Subscribe | Capsules | 20 mg, 40 mg, 60 mg and 80 mg | ➤ Subscribe | 2005-02-07 |
| ➤ Subscribe | Capsules | 75 mg, 100 mg and 125 mg | ➤ Subscribe | 2019-02-04 |
| ➤ Subscribe | Capsules | 0.125 mg, 0.25 mg, and 0.5 mg | ➤ Subscribe | 2014-05-01 |
| ➤ Subscribe | Tablets | 600 mg | ➤ Subscribe | 2005-12-21 |
| ➤ Subscribe | Injection | 2 mg/mL, 300 mL bag | ➤ Subscribe | 2009-09-01 |
| ➤ Subscribe | Tablets | 1 g | ➤ Subscribe | 2005-08-23 |
| ➤ Subscribe | Delayed-release Tablets | 75 mg/0.2 mg | ➤ Subscribe | 2008-11-28 |
| ➤ Subscribe | Extended-release Tablets | 4 mg and 8 mg | ➤ Subscribe | 2012-10-31 |
| ➤ Subscribe | Injection | 20 mg/mL, 2 mL and 5 mL vials | ➤ Subscribe | 2004-07-26 |
| ➤ Subscribe | Capsules | 5 mg and 10 mg | ➤ Subscribe | 2005-06-21 |
| ➤ Subscribe | For Injection | 500 mg/vial | ➤ Subscribe | 2011-06-17 |
| ➤ Subscribe | Oral Suspension | 100 mg/5 mL | ➤ Subscribe | 2009-08-03 |
| ➤ Subscribe | Injection | 2 mg/mL, 100 mL bag | ➤ Subscribe | 2009-12-29 |
International Patents for Pfizer Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Spain | 2251677 | ⤷ Try a Trial |
| San Marino | T201400019 | ⤷ Try a Trial |
| African Intellectual Property Organization (OAPI) | 12755 | ⤷ Try a Trial |
| Portugal | 2958921 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Pfizer Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2170860 | C20200032 00354 | Estonia | ⤷ Try a Trial | PRODUCT NAME: GLASDEGIIB;REG NO/DATE: EU/1/20/1451 29.06.2020 |
| 1666481 | CA 2017 00035 | Denmark | ⤷ Try a Trial | PRODUCT NAME: TOFACITINIB, EVENTUELT I FORM AF ET FARMACEUTISK ACCEPTABELT SALT, HERUNDER CITRAT-SALTET; REG. NO/DATE: EU/1/17/1178/001-003 20170324 |
| 0328535 | 96C0021 | Belgium | ⤷ Try a Trial | PRODUCT NAME: LANSOPRAZOLE + CLARITHROMYCINE + METRONIDAZOLE; REGISTRATION NO/DATE IN FRANCE: K 27 17033R DU 19960209; REGISTRATION NO/DATE AT EEC: K 27 17033R DU 19960209 |
| 2603514 | CA 2019 00015 | Denmark | ⤷ Try a Trial | PRODUCT NAME: EN KOMBINATION AF VABORBACTAM, OG/ELLER ET SALT OG/ELLER HYDRAT DERAF, OG MEROPENEM, OG/ELLER ET SALT OG/ELLER HYDRAT DERAF, SAERLIGT MEROPENEM TRIHYDRAT; REG. NO/DATE: EU/1/18/1334 20181122 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.