Last updated: February 19, 2026
ZAVZPRET (zavegepant) nasal spray, approved by the U.S. Food and Drug Administration (FDA) in July 2023, targets acute treatment of migraine with or without aura in adults. Developed by Biohaven Pharmaceuticals, a subsidiary of Pfizer, the drug's market entry is positioned within a competitive landscape for migraine therapeutics. Its mechanism of action as a calcitonin gene-related peptide (CGRP) receptor antagonist offers a distinct delivery method—nasal spray—which may appeal to patients seeking rapid relief and alternatives to oral or injectable formulations. The financial trajectory of ZAVZPRET will be shaped by its market penetration, physician adoption, patient access, and competitive pressures from existing and emerging migraine treatments.
What is the mechanism of action and therapeutic indication for ZAVZPRET?
ZAVZPRET is a prescription medication indicated for the acute treatment of migraine with or without aura in adults. It operates as a selective CGRP receptor antagonist. CGRP is a neuropeptide implicated in the pathophysiology of migraine. By blocking the CGRP receptor, zavegepant is designed to inhibit the vasodilation and neurogenic inflammation associated with migraine attacks, thereby providing pain relief. The nasal spray formulation aims for rapid absorption, offering a potential advantage for acute symptom management.
Who are the key developers and manufacturers of ZAVZPRET?
Biohaven Pharmaceuticals, a Pfizer subsidiary, developed ZAVZPRET. Pfizer acquired Biohaven in October 2022, integrating ZAVZPRET into its neuroscience portfolio. Manufacturing is handled by Biohaven and its contracted facilities, with Pfizer overseeing commercialization and distribution in the United States.
What is the regulatory status and approval history of ZAVZPRET?
ZAVZPRET received U.S. FDA approval on July 12, 2023. The approval was based on data from two Phase 3 clinical trials (Study 301 and Study 302) that demonstrated the efficacy and safety of ZAVZPRET compared to placebo. These trials evaluated ZAVZPRET 10 mg as an immediate-release nasal spray for the acute treatment of migraine. The FDA approval allows for its marketing and sale in the U.S. for its approved indication.
What is the current market landscape for migraine treatments?
The market for migraine treatments is extensive and continually evolving, driven by the high prevalence of migraine and the unmet needs of many patients. Key segments include:
The market is characterized by a shift towards targeted therapies like CGRP antagonists, offering more specific mechanisms of action. Competition is intense, with multiple drug classes and individual products vying for physician prescription and patient adherence.
How does ZAVZPRET compare to existing acute migraine treatments in terms of efficacy and safety?
ZAVZPRET demonstrated statistically significant superiority over placebo in key efficacy endpoints in its Phase 3 trials. In Study 301, ZAVZPRET 10 mg achieved an 18.5% pain freedom rate at 2 hours post-dose, compared to 12.0% for placebo (p=0.001). In Study 302, the pain freedom rate was 24.2% for ZAVZPRET 10 mg versus 13.8% for placebo (p<0.001) [1].
Key Efficacy Endpoints (2-hour post-dose):
| Endpoint |
ZAVZPRET 10 mg |
Placebo |
Difference |
| Pain Freedom |
18.5% (Study 301) / 24.2% (Study 302) |
12.0% (Study 301) / 13.8% (Study 302) |
6.5 pp / 10.4 pp |
| Most Bothersome Symptom Freedom |
30.4% (Study 301) / 37.2% (Study 302) |
20.6% (Study 301) / 25.7% (Study 302) |
9.8 pp / 11.5 pp |
| Sustained Pain Freedom (24 hours) |
14.5% (Study 301) / 20.2% (Study 302) |
10.5% (Study 301) / 14.2% (Study 302) |
4.0 pp / 6.0 pp |
pp: percentage points.
Safety Profile: The most common adverse events reported in clinical trials included altered taste (dysgeusia) and nasal discomfort. These were generally mild to moderate in severity. Rates of cardiovascular ischemic events were low and comparable between ZAVZPRET and placebo groups. ZAVZPRET has a boxed warning regarding the potential for sphincter of Oddi spasm, a risk shared with other CGRP antagonists.
Comparison with other acute CGRP antagonists (oral gepants):
- Ubrelvy (ubrogepant): Approved for acute migraine treatment. Efficacy rates for pain freedom at 2 hours in Phase 3 trials were around 20-24% for 50mg and 100mg doses, compared to placebo rates of approximately 10-12% [2].
- Nurtec ODT (rimegepant): Approved for both acute treatment and migraine prevention. For acute treatment, pain freedom at 2 hours in Phase 3 trials was approximately 19% for the 75mg dose, compared to 11% for placebo [3].
ZAVZPRET's nasal spray formulation may offer a faster onset of action compared to oral gepants, though direct head-to-head comparisons are limited. Physician preference and patient tolerance to specific formulations will influence adoption.
What are the pricing and reimbursement strategies for ZAVZPRET?
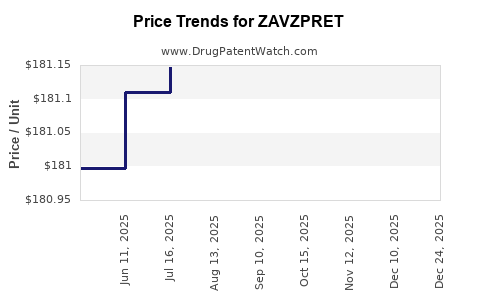
Biohaven established a wholesale acquisition cost (WAC) of $2,875 for a 30-day supply of ZAVZPRET (which contains 8 doses). This pricing positions it competitively within the acute migraine market. Pfizer's market access strategies are expected to focus on securing formulary placement with major pharmacy benefit managers (PBMs) and commercial insurers. Patient support programs, such as co-pay assistance, will likely be implemented to mitigate out-of-pocket costs and enhance affordability, particularly given the cost of newer migraine therapies. The company aims to ensure broad patient access, acknowledging the critical role of insurance coverage for high-value novel therapeutics.
What is the expected market penetration and revenue potential for ZAVZPRET?
Market penetration for ZAVZPRET will be contingent on several factors:
- Physician Adoption: Clinician familiarity with CGRP antagonists and comfort with the nasal spray delivery system will be crucial. Educational initiatives and physician outreach by Pfizer will support adoption.
- Patient Acceptance: Patients seeking rapid relief and those who have not responded well to or tolerated oral or injectable treatments may be drawn to the nasal spray format.
- Payer Coverage: Favorable formulary placement and reasonable co-pays will be essential for patient access.
- Competitive Landscape: The existing market includes established oral gepants and the broad class of triptans. ZAVZPRET must demonstrate clear advantages or address unmet needs.
While specific revenue projections are proprietary, analysts anticipate ZAVZPRET to contribute significantly to Biohaven's and Pfizer's migraine franchise revenue. Given that migraine affects millions of individuals globally and the market for effective treatments is substantial, a successful launch could lead to hundreds of millions in annual sales. The rapid onset of action claimed for the nasal spray format could capture a meaningful share of the acute treatment market, particularly among patients prioritizing speed of relief. Industry estimates for the migraine market size vary but are in the tens of billions of dollars globally, with acute treatments representing a substantial portion.
What are the primary challenges and risks for ZAVZPRET's commercial success?
- Competition: The migraine market is crowded. Established triptans (often generic and less expensive) remain widely used. Oral gepants (Ubrelvy, Nurtec ODT) also compete directly. Preventive therapies, while distinct, reduce the overall number of migraine days, potentially impacting acute treatment demand.
- Reimbursement Hurdles: Despite efforts, payers may impose step-therapy requirements or prior authorization hurdles, limiting immediate patient access.
- Physician Education and Patient Education: Convincing healthcare providers to prescribe a new nasal spray formulation and educating patients on its proper use and benefits requires substantial marketing and educational investment.
- Patient Tolerance/Adherence to Nasal Spray: Some patients may have aversions to nasal sprays due to past experiences with other medications or concerns about nasal irritation.
- Safety Profile Nuances: While generally well-tolerated, any rare but serious adverse events could impact perception and market acceptance. The boxed warning for sphincter of Oddi spasm requires careful physician communication.
- Market Saturation: The acute migraine market is mature, making it challenging for new entrants to gain significant market share without clear differentiation.
What is the long-term financial outlook for ZAVZPRET?
The long-term financial outlook for ZAVZPRET hinges on its ability to establish a sustained market presence. Key determinants include:
- Clinical Differentiation: Demonstrating superior real-world effectiveness or patient-reported outcomes compared to existing therapies.
- Lifecycle Management: Potential development of other formulations or indications, though currently none are disclosed.
- Patent Protection: The duration of patent exclusivity for zavegepant will dictate the period of market exclusivity before generic competition emerges.
- Pfizer's Commercial Strategy: The effectiveness of Pfizer's sales force, marketing campaigns, and patient support programs in driving and maintaining prescriptions.
If ZAVZPRET can successfully carve out a niche for rapid-acting acute migraine relief and achieve broad payer coverage, it could become a significant revenue generator for Pfizer's neuroscience division, potentially achieving blockbuster status (>$1 billion in annual sales) over its lifecycle. However, achieving this will require overcoming substantial competitive and market access challenges.
Key Takeaways
- ZAVZPRET is a newly approved CGRP receptor antagonist nasal spray for acute migraine treatment, developed by Biohaven (Pfizer).
- It competes in a mature and crowded market, facing established triptans, oral gepants, and preventive therapies.
- Clinical trials show ZAVZPRET provides statistically significant pain freedom and MBS freedom compared to placebo, with common side effects including altered taste and nasal discomfort.
- Pricing is competitive, and commercial strategies will focus on payer access and patient support programs.
- Challenges include intense competition, reimbursement hurdles, and the need for physician and patient education on its novel nasal spray formulation.
- Long-term financial success depends on sustained market penetration, clinical differentiation, and effective commercial execution by Pfizer.
Frequently Asked Questions
1. What is the primary advantage of ZAVZPRET's nasal spray formulation?
The primary advantage of ZAVZPRET's nasal spray formulation is its potential for rapid absorption, which may lead to faster onset of pain relief compared to oral medications.
2. How does ZAVZPRET's safety profile compare to oral gepants?
ZAVZPRET shares a similar safety profile to oral gepants, with common side effects like altered taste and nasal discomfort. Both drug classes carry a boxed warning regarding the potential for sphincter of Oddi spasm.
3. What is the typical patient profile that might benefit most from ZAVZPRET?
Patients who experience migraines and require fast-acting relief, or those who have had difficulty with oral medications or prefer non-oral administration routes, may benefit most from ZAVZPRET.
4. Will ZAVZPRET be available in generic form soon?
As a newly approved drug (July 2023), ZAVZPRET is currently protected by patent exclusivity. Generic versions are not expected for many years, contingent on patent expiry and potential challenges.
5. How does ZAVZPRET fit into the broader CGRP antagonist market?
ZAVZPRET expands the CGRP antagonist class by offering a nasal spray option for acute treatment, complementing existing oral gepants and injectable preventive monoclonal antibodies. This provides more delivery method choices within the CGRP-targeting therapeutic space.
Citations
[1] Biohaven Pharmaceuticals. (2023, January 3). Biohaven announces positive results from pivotal Phase 3 trial of zavegepant 10 mg nasal spray for the acute treatment of migraine. GlobeNewswire. https://ir.biohaven.com/news-releases/news-release-details/biohaven-announces-positive-results-pivotal-phase-3-trial
[2] Product Information for Ubrelvy (ubrogepant) tablets. AbbVie Inc. U.S.A. [Date of Access relevant to information cited]
[3] Product Information for Nurtec ODT (rimegepant) orally disintegrating tablets. Biohaven Pharmaceuticals, Inc. U.S.A. [Date of Access relevant to information cited]