ORGANON Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ORGANON, and what generic alternatives to ORGANON drugs are available?
ORGANON has fifty-nine approved drugs.
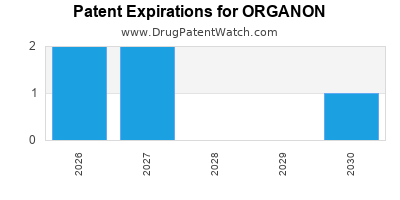
There are six US patents protecting ORGANON drugs. There is one tentative approval on ORGANON drugs.
There are three hundred and thirteen patent family members on ORGANON drugs in forty-four countries and one hundred and thirty-six supplementary protection certificates in fifteen countries.
Drugs and US Patents for ORGANON
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Organon Llc | ASMANEX HFA | mometasone furoate | AEROSOL, METERED;INHALATION | 205641-002 | Apr 25, 2014 | RX | Yes | Yes | ⤷ Sign Up | ⤷ Sign Up | |||||
| Organon Usa Inc | NORCURON | vecuronium bromide | INJECTABLE;INJECTION | 018776-003 | Jan 3, 1992 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | |||||
| Organon | NEXPLANON | etonogestrel | IMPLANT;IMPLANTATION | 021529-002 | May 13, 2011 | RX | Yes | Yes | 8,722,037 | ⤷ Sign Up | Y | ⤷ Sign Up | |||
| Organon Llc | MAXALT | rizatriptan benzoate | TABLET;ORAL | 020864-001 | Jun 29, 1998 | DISCN | Yes | No | ⤷ Sign Up | ⤷ Sign Up | |||||
| Organon Usa Inc | DOCA | desoxycorticosterone acetate | INJECTABLE;INJECTION | 001104-001 | Approved Prior to Jan 1, 1982 | DISCN | No | No | ⤷ Sign Up | ⤷ Sign Up | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ORGANON
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Organon | VYTORIN | ezetimibe; simvastatin | TABLET;ORAL | 021687-003 | Jul 23, 2004 | 4,444,784 | ⤷ Sign Up |
| Organon | CLARINEX | desloratadine | TABLET, ORALLY DISINTEGRATING;ORAL | 021312-002 | Jul 14, 2005 | 4,659,716*PED | ⤷ Sign Up |
| Organon | FOSAMAX | alendronate sodium | TABLET;ORAL | 020560-001 | Sep 29, 1995 | 5,681,590*PED | ⤷ Sign Up |
| Organon Usa Organon | REMERON SOLTAB | mirtazapine | TABLET, ORALLY DISINTEGRATING;ORAL | 021208-001 | Jan 12, 2001 | 5,977,099 | ⤷ Sign Up |
| Organon | MAXALT-MLT | rizatriptan benzoate | TABLET, ORALLY DISINTEGRATING;ORAL | 020865-001 | Jun 29, 1998 | 4,758,598 | ⤷ Sign Up |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for ORGANON drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Tablets | 10 mg | ➤ Subscribe | 2007-02-20 |
| ➤ Subscribe | Tablets | 5 mg | ➤ Subscribe | 2006-06-21 |
| ➤ Subscribe | Orally Disintegrating Tablets | 5 mg and 10 mg | ➤ Subscribe | 2006-02-17 |
| ➤ Subscribe | Tablets | 50 mg/12.5 mg and 100 mg/25 mg | ➤ Subscribe | 2004-05-24 |
| ➤ Subscribe | Tablets | 10 mg/10 mg, 10 mg/20 mg, 10 mg/40 mg and 10 mg/80 mg | ➤ Subscribe | 2009-07-27 |
| ➤ Subscribe | Nasal Spray | 50 mcg/ Spray | ➤ Subscribe | 2009-08-07 |
| ➤ Subscribe | Tablets | 5 mg and 10 mg | ➤ Subscribe | 2004-09-02 |
| ➤ Subscribe | Vaginal Ring | 0.015 mg/24 hour and 0.12 mg/24 hour | ➤ Subscribe | 2013-06-17 |
| ➤ Subscribe | Topical Solution (Lotion) | 0.10% | ➤ Subscribe | 2004-06-10 |
| ➤ Subscribe | Chewable Tablets | 4 mg and 5 mg | ➤ Subscribe | 2006-12-26 |
| ➤ Subscribe | Orally Disintegrating Tablets | 2.5 mg and 5 mg | ➤ Subscribe | 2006-06-21 |
| ➤ Subscribe | Oral Solution | 70 mg/75 mL | ➤ Subscribe | 2007-09-07 |
| ➤ Subscribe | Tablets | 100 mg/12.5 mL | ➤ Subscribe | 2006-04-04 |
| ➤ Subscribe | Tablets | 10 mg | ➤ Subscribe | 2006-10-25 |
| ➤ Subscribe | Tablets | 70 mg/2800 IU and 70 mg/5600 IU | ➤ Subscribe | 2007-11-20 |
| ➤ Subscribe | Injection | 250 mcg/0.5 mL, 1 mL PFS | ➤ Subscribe | 2012-03-30 |
International Patents for ORGANON Drugs
Supplementary Protection Certificates for ORGANON Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0497512 | 99C0011 | Belgium | ⤷ Sign Up | PRODUCT NAME: RIZATRIPTAN BENZOAS EQ. RIZATRIPTAN; REGISTRATION NO/DATE: 922 IS 181 F 3 19981130; FIRST REGISTRATION: NL RVG 21815 19980211 |
| 2435025 | 36/2019 | Austria | ⤷ Sign Up | PRODUCT NAME: GLYCOPYRRONIUMBROMID / FORMOTEROL; REGISTRATION NO/DATE: EU/1/18/1339 (MITTEILUNG) 20181220 |
| 0277829 | 2000C/030 | Belgium | ⤷ Sign Up | PRODUCT NAME: GANIRELIX; REGISTRATION NO/DATE: EU/1/00/130/001 20000518 |
| 1453521 | 300814 | Netherlands | ⤷ Sign Up | PRODUCT NAME: LEVONORGESTREL EN ETHINYLESTRADIOL; NATIONAL REGISTRATION NO/DATE: RVG 117453 20151211; FIRST REGISTRATION: SK 17/0017/15-S 20150211 |
| 0720599 | 122014000088 | Germany | ⤷ Sign Up | PRODUCT NAME: EZETIMIB ODER PHARMAZEUTISCH ANNEHMBARE SALZE DAVON IN KOMBINATION MIT ATORVASTATIN ODER PHARMAZEUTISCH ANNEHMBARE SALZE DAVON, INSBESONDERE ATORVASTATIN ALS ATORVASTATINCALCIUMTRIHYDRAT; NAT. REGISTRATION NO/DATE: 90223.00.00 20141113; FIRST REGISTRATION: FRANKREICH CIS: 6 928 917 6 20140912 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.