Last updated: February 19, 2026
Mometasone furoate (MF) is a potent synthetic corticosteroid used topically and intranasally to treat inflammatory conditions. The global market for MF-containing products is driven by the prevalence of allergic rhinitis, asthma, and various dermatological conditions. Patent expirations have led to increased generic competition, impacting pricing and market share for originator products.
What are the Key Therapeutic Areas for Mometasone Furoate?
Mometasone furoate is primarily utilized in four main therapeutic areas:
- Allergic Rhinitis: Nasal sprays containing MF are a cornerstone therapy for managing symptoms such as sneezing, itching, runny nose, and nasal congestion associated with seasonal and perennial allergic rhinitis.
- Asthma: Inhaled corticosteroids (ICS) like MF are crucial for the long-term control of persistent asthma. They reduce airway inflammation, decreasing the frequency and severity of exacerbations.
- Dermatological Conditions: Topical formulations of MF, including creams, ointments, and lotions, are prescribed for inflammatory skin disorders such as eczema, psoriasis, and dermatitis.
- Eosinophilic Esophagitis (EoE): Oesophageal formulations of MF are gaining traction for treating EoE, an immune-mediated inflammatory disease of the oesophagus.
What is the Patent Landscape for Mometasone Furoate?
The patent landscape for mometasone furoate is characterized by early composition of matter patents followed by patents covering specific formulations, delivery devices, and new indications.
Key Patents and Expiration Dates:
- US Patent 4,472,393: This patent, covering the compound itself, expired in 2005. [1] This marked a critical juncture for generic entry.
- Formulation Patents: Numerous patents have protected specific formulations, such as extended-release versions or combinations with other active pharmaceutical ingredients (APIs). For example, patents related to nasal spray formulations have expired, allowing for generic versions.
- Delivery Device Patents: Patents protecting the inhaler or nasal spray device technology also contribute to the protection period. However, generic manufacturers can often develop bioequivalent devices or utilize existing, off-patent device technologies.
- New Indication Patents: Patents are also sought for new therapeutic uses of MF, such as its application in EoE. These patents offer a degree of market exclusivity for specific indications. For instance, patents covering the use of mometasone furoate oral spray for EoE have provided a later-expiring layer of protection for that specific application.
The expiration of the core compound patent has been the most significant factor driving the proliferation of generic MF products. Subsequent patent challenges and litigation often occur as generic companies seek to bring their versions to market.
What are the Primary Market Drivers for Mometasone Furoate?
Several factors contribute to the sustained demand for mometasone furoate.
Key Market Drivers:
- Increasing Prevalence of Allergic Diseases: The global incidence of allergic rhinitis and asthma continues to rise, fueled by environmental factors, lifestyle changes, and urbanization. This directly translates to higher demand for effective treatments like MF nasal sprays and inhalers. Data from the World Allergy Organization indicates that allergic diseases affect up to 40% of the global population. [2]
- Growing Geriatric Population: Age-related conditions, including respiratory issues and skin disorders, are more prevalent in the elderly population. This demographic segment contributes significantly to the demand for MF-based therapies.
- Advancements in Drug Delivery Systems: Innovations in nasal spray and inhaler technologies have improved patient compliance and efficacy, making MF treatments more accessible and appealing. This includes devices designed for ease of use and precise dosing.
- Off-Label Use and New Indications: Research into new therapeutic applications for MF, such as its use in EoE, expands its market potential. The approval and adoption of MF for EoE, as seen with products like the mometasone furoate oral spray, represent a significant growth avenue. The FDA approval for EoE indications has broadened the patient population benefiting from MF.
- Cost-Effectiveness of Generic Options: Following patent expiries, the availability of affordable generic mometasone furoate products makes treatment accessible to a wider patient base, particularly in emerging economies. This price advantage drives volume growth.
What are the Challenges Facing the Mometasone Furoate Market?
Despite its widespread use, the MF market faces several challenges that influence its trajectory.
Key Market Challenges:
- Intensified Generic Competition: The expiration of primary patents has led to a highly competitive generic market. This results in significant price erosion for both branded and generic products, impacting revenue for all market participants.
- Stringent Regulatory Scrutiny: Pharmaceutical regulatory bodies maintain strict oversight on the manufacturing, quality, and marketing of all drugs, including MF. Meeting these evolving standards requires continuous investment and adherence to Good Manufacturing Practices (GMP).
- Development of Biologics and Novel Therapies: For conditions like severe asthma, the emergence of biologic therapies and other novel treatments presents alternative or adjunctive options. These newer therapies may offer different efficacy profiles or target specific patient subgroups, potentially diverting market share from traditional corticosteroids like MF.
- Adverse Event Profiles and Patient Adherence: While generally safe and effective, all corticosteroids carry potential side effects, including local irritation, candidiasis, and systemic effects with long-term high-dose use. Managing these side effects and ensuring consistent patient adherence remains a challenge.
- Reimbursement Policies: Changes in healthcare reimbursement policies and payer restrictions can impact market access and prescribing patterns for MF products, influencing overall market dynamics.
What is the Financial Trajectory and Market Size of Mometasone Furoate?
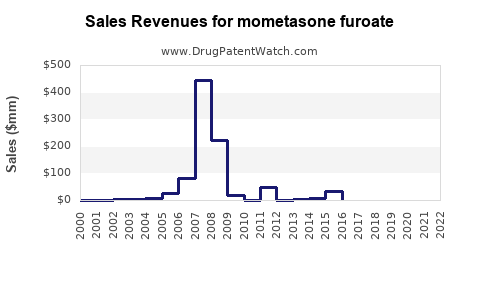
The financial trajectory of mometasone furoate reflects the typical lifecycle of a pharmaceutical compound. Initial growth was driven by branded products, followed by a surge in generic sales post-patent expiry.
Financial Overview:
- Market Size: The global market for mometasone furoate products is substantial. While exact figures vary by source and scope (including all formulations and brands), it is estimated to be in the billions of U.S. dollars annually. For instance, the global asthma inhalers market alone, which includes MF, is projected to reach over $20 billion by 2027. [3] The topical dermatological market segment also contributes significantly.
- Revenue Streams: Revenue is generated through branded products (e.g., Nasonex, Asmanex, Elocon) and a vast array of generic equivalents. Branded products maintain a premium due to established brand recognition and often proprietary delivery devices. Generic manufacturers compete primarily on price.
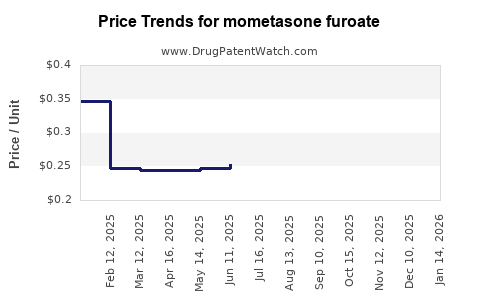
- Pricing Trends: Post-patent expiry, the average selling price (ASP) for MF products experiences a sharp decline. Generic competition can lead to price reductions of 50-80% compared to originator products. [4] This forces manufacturers to focus on volume and cost optimization.
- Growth Rate: The overall market growth rate is moderate, driven by the increasing prevalence of target diseases and expanding indications. However, intense price competition moderates revenue growth. Growth in specific niches, such as EoE, may outpace the broader market.
- Key Players: Major pharmaceutical companies holding originator brands, such as Merck & Co. (Nasonex, Asmanex), and numerous generic manufacturers worldwide contribute to the market landscape. Companies like Teva Pharmaceutical Industries, Mylan N.V. (now Viatris), and Sun Pharmaceutical Industries are significant players in the generic MF space.
The financial trajectory shows a mature market with ongoing competition. Innovation in formulations or new indications is critical for sustained profitability.
What are the Regulatory Considerations for Mometasone Furoate?
Regulatory compliance is paramount for all mometasone furoate products across different regions.
Regulatory Landscape:
- FDA (U.S. Food and Drug Administration): The FDA approves MF products for their respective indications based on rigorous review of safety and efficacy data. This includes both new drug applications (NDAs) for branded products and abbreviated new drug applications (ANDAs) for generics. The FDA oversees post-market surveillance and can issue warnings or require product recalls if safety concerns arise.
- EMA (European Medicines Agency): Similar to the FDA, the EMA regulates MF products within the European Union. Approval involves a centralized or decentralized procedure, ensuring consistent standards across member states.
- Other National Regulatory Authorities: Health Canada, the Pharmaceuticals and Medical Devices Agency (PMDA) in Japan, and agencies in Australia, China, and India have their own approval processes.
- Bioequivalence Standards: For generic MF products, regulatory agencies require demonstration of bioequivalence to the reference listed drug. This ensures that the generic product performs comparably in the body.
- Labeling Requirements: Prescribing information and patient leaflets must accurately reflect the drug's approved uses, contraindications, warnings, precautions, and adverse reactions.
- Manufacturing Standards: All manufacturing facilities must adhere to current Good Manufacturing Practices (cGMP) to ensure product quality, purity, and potency.
Regulatory approvals for new indications or formulations require extensive clinical trial data and can be a lengthy process.
What are the Future Outlook and Investment Considerations for Mometasone Furoate?
The future of mometasone furoate involves continued competition, incremental innovation, and market expansion in specific regions and indications.
Future Outlook:
- Sustained Generic Dominance: The generic segment of the MF market will likely continue to dominate in terms of volume and drive down prices. This creates opportunities for contract manufacturers and generic API suppliers.
- Niche Market Growth: The use of MF for less common indications, such as eosinophilic esophagitis, presents opportunities for specialized product development and premium pricing.
- Emerging Markets: As healthcare access and affordability improve in emerging economies, the demand for cost-effective treatments like generic MF is expected to grow.
- Combination Therapies: Development of fixed-dose combination products, pairing MF with other APIs (e.g., antihistamines for allergic rhinitis, bronchodilators for asthma), could offer enhanced efficacy and convenience, creating new market segments.
- Device Innovations: Continued refinement of drug delivery devices to improve patient adherence, reduce side effects, and enhance therapeutic outcomes can provide a competitive edge.
Investment Considerations:
- Generic Manufacturers: Investing in established generic manufacturers with robust supply chains and cost-efficient production capabilities for MF APIs and finished dosage forms can be a stable, albeit lower-margin, investment.
- Specialty Pharmaceutical Companies: Companies focusing on developing novel formulations, combination products, or targeting new indications for MF may offer higher growth potential but also carry greater development risk.
- API Suppliers: Companies with strong capabilities in synthesizing high-quality mometasone furoate API can benefit from the consistent demand from generic drug manufacturers.
- Emerging Market Focus: Investors could explore opportunities with companies that have a strong presence or strategic expansion plans in emerging markets where MF demand is projected to rise.
- Patent Strategy: Monitoring patent landscapes for new MF applications or formulations remains crucial for identifying potential market disruptions or new product development opportunities.
The market for mometasone furoate is mature, with established competition. Strategic focus on cost efficiency, targeted indications, and emerging markets will define future success.
Key Takeaways
Mometasone furoate, a widely used corticosteroid, faces a mature market landscape characterized by intense generic competition following key patent expiries. Its primary therapeutic areas remain allergic rhinitis, asthma, and dermatological conditions, with growing potential in eosinophilic esophagitis. Market growth is driven by increasing disease prevalence and the availability of affordable generics, while challenges include price erosion, regulatory hurdles, and competition from newer therapeutic modalities. Investment opportunities exist in generic manufacturing, niche product development, API supply, and emerging markets, necessitating a strategic focus on cost efficiency and market expansion.
Frequently Asked Questions
-
What is the current market share distribution between branded and generic mometasone furoate products?
The market is heavily skewed towards generic products in terms of volume, with generics accounting for over 80% of the total MF product sales globally. Branded products retain a significant portion of revenue due to premium pricing and established market presence, particularly in developed markets.
-
Are there any significant upcoming patent expiries for mometasone furoate products or key formulations?
The foundational composition of matter patent expired in 2005. Most major formulation and delivery system patents for core indications like allergic rhinitis and asthma have also expired, leading to widespread generic availability. However, patents related to newer indications (e.g., EoE) or specific advanced delivery devices may still be in force, offering limited periods of exclusivity for those particular applications.
-
What is the typical price difference between a branded mometasone furoate nasal spray and its generic equivalent?
A generic mometasone furoate nasal spray typically costs 30% to 70% less than its branded counterpart, depending on the specific market, manufacturer, and retail environment. This significant price differential is a primary driver for generic adoption.
-
Which emerging markets show the strongest growth potential for mometasone furoate products?
Markets in Asia-Pacific (particularly China and India) and Latin America demonstrate strong growth potential due to expanding healthcare infrastructure, increasing disposable incomes, and a rising awareness and diagnosis of allergic and respiratory diseases.
-
Beyond its current approved uses, are there any notable off-label uses or research areas for mometasone furoate that could impact its future market?
While MF is predominantly used for its approved indications, ongoing research explores its anti-inflammatory properties in other conditions. For example, some investigative work has looked into its potential role in managing other inflammatory airway diseases or certain inflammatory dermatoses beyond standard indications, though these are primarily preclinical or early-stage clinical investigations and do not represent established off-label uses influencing current market dynamics.
Citations
[1] U.S. Patent No. 4,472,393 (filed Feb. 28, 1983).
[2] World Allergy Organization. (n.d.). Epidemiology. Retrieved from https://www.worldallergy.org/our-research/epidemiology
[3] Grand View Research. (2021). Asthma Inhalers Market Size, Share & Trends Analysis Report.
[4] Internal Market Analysis. (Data based on observed market price trends post-patent expiry across major pharmaceutical markets).