Last updated: February 19, 2026
VYTORIN (ezetimibe/simvastatin) is a combination cholesterol-lowering medication. Its market performance is influenced by patent expirations, generic competition, evolving treatment guidelines, and the availability of alternative lipid-lowering therapies.
What is VYTORIN's Compound Mechanism of Action and Approved Indications?
VYTORIN combines ezetimibe, a cholesterol absorption inhibitor, with simvastatin, a hydroxymethylglutaryl-coenzyme A (HMG-CoA) reductase inhibitor (statin). Ezetimibe works by inhibiting the absorption of cholesterol in the small intestine, while simvastatin reduces cholesterol synthesis in the liver. This dual mechanism targets cholesterol from two distinct pathways, leading to a greater reduction in low-density lipoprotein cholesterol (LDL-C) compared to either agent alone.
VYTORIN is indicated for the reduction of elevated total cholesterol, LDL-C, apolipoprotein B (apo B), and triglycerides, and for the increase of high-density lipoprotein cholesterol (HDL-C) in patients with primary hypercholesterolemia, as an adjunct to diet. It is also indicated for the reduction of elevated total cholesterol and LDL-C in patients with homozygous familial hypercholesterolemia (HoFH), as an adjunct to diet and other lipid-lowering treatments. Additionally, it is used in patients with homozygous sitosterolemia (phytosterolemia) as an adjunct to diet.
The initial U.S. Food and Drug Administration (FDA) approval for VYTORIN was granted on October 11, 2004.
What is VYTORIN's Patent Landscape and Expiration Timeline?
The patent protection for VYTORIN has significantly shaped its market exclusivity and subsequent generic entry. Key patents and their expiration dates are critical for understanding market dynamics.
- Compound Patents: Patents covering the active pharmaceutical ingredients (ezetimibe and simvastatin) and their combination have expired.
- Formulation and Method of Use Patents: While some formulation and method of use patents may have provided extended protection, they have largely expired or have been successfully challenged, paving the way for generic competition.
The primary market exclusivity for the branded VYTORIN, stemming from its core composition patents, ended in 2010 in the United States and 2011 in Europe. This expiration opened the door for the introduction of generic versions of ezetimibe/simvastatin.
How Has Generic Competition Impacted VYTORIN's Market Share and Pricing?
The expiration of VYTORIN's patent protection led to the rapid introduction of generic alternatives, dramatically altering its market dynamics.
- Market Share Erosion: Following patent expiry, the market share of branded VYTORIN experienced a sharp decline. Generic ezetimibe/simvastatin formulations quickly captured a substantial portion of the market due to their lower price points.
- Price Reduction: The introduction of generics typically results in significant price reductions. The average selling price (ASP) for ezetimibe/simvastatin has fallen by over 90% since the peak of branded VYTORIN sales.
- Market Dynamics Shift: The market for ezetimibe/simvastatin is now predominantly driven by generic manufacturers, with branded VYTORIN holding a minimal market share. Prescriptions are largely filled with generic equivalents unless a specific formulary restriction or patient preference dictates otherwise.
What are the Key Clinical Differentiators and Evolving Treatment Guidelines for Cholesterol Management?
The clinical positioning of VYTORIN has been influenced by advancements in understanding dyslipidemia and the development of new therapeutic classes.
- Early Advantage: At its launch, VYTORIN offered a distinct advantage by combining two complementary mechanisms of action, providing a more potent LDL-C lowering effect than monotherapy statins. This was particularly relevant for patients with high cardiovascular risk or those not achieving LDL-C goals with statins alone.
- Emergence of PCSK9 Inhibitors: The development of proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors (e.g., evolocumab, alirocumab) has significantly altered the landscape for very high-risk patients. These injectable biologics offer potent LDL-C reduction and are now a standard of care for certain patient populations, particularly those with familial hypercholesterolemia or statin intolerance.
- Evolving Guidelines: Cholesterol management guidelines have evolved to emphasize risk stratification and personalized treatment approaches. While statins remain foundational, the role of ezetimibe has been refined. Current guidelines from organizations like the American Heart Association (AHA) and the American College of Cardiology (ACC) recommend statin therapy as first-line treatment for most patients requiring lipid lowering. Ezetimibe is often considered an option for patients who cannot achieve LDL-C goals with maximally tolerated statin therapy or who are statin-intolerant.
The clinical profile of VYTORIN, while once a significant differentiator, is now viewed within a broader therapeutic context that includes PCSK9 inhibitors and other novel agents.
What is VYTORIN's Historical Financial Performance and Current Market Size?
The financial trajectory of VYTORIN reflects its lifecycle from a blockbuster product to a post-patent, largely genericized market.
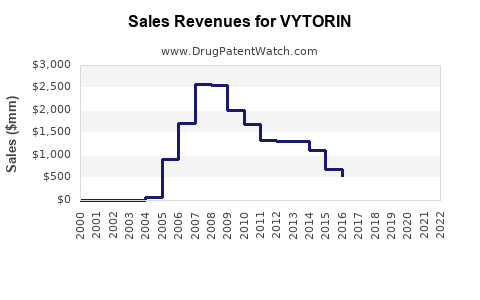
- Peak Sales: Branded VYTORIN achieved peak annual sales exceeding $5 billion globally in the mid-2000s, prior to significant generic entry.
- Sales Decline: Following patent expirations, branded VYTORIN sales experienced a precipitous decline. By 2015, global sales had fallen to approximately $700 million, and by 2020, they were below $100 million.
- Current Market Size (Ezetimibe/Simvastatin Combination): The combined market for ezetimibe/simvastatin, encompassing both branded and generic versions, is now significantly smaller than the peak of branded VYTORIN. It is difficult to isolate the precise current market size for branded VYTORIN as sales are consolidated with generic performance. However, the overall market for ezetimibe/simvastatin generics is estimated to be in the low hundreds of millions of dollars annually, driven by volume rather than high pricing.
- Key Markets: The United States and major European countries were the largest markets for VYTORIN. Generic penetration has been high across these regions.
The financial performance clearly illustrates the impact of patent expiry and generic competition on a pharmaceutical product's revenue stream.
What are the Primary Challenges and Opportunities for VYTORIN and its Generic Equivalents?
The market for VYTORIN and its generic equivalents faces distinct challenges and limited opportunities.
Challenges:
- Intensified Generic Competition: The market is characterized by numerous generic manufacturers, leading to constant price pressure and low profit margins.
- Competition from Newer Therapies: The advent of PCSK9 inhibitors and other novel lipid-lowering agents for specific high-risk patient groups presents a challenge to the perceived need for older combination therapies.
- Evolving Treatment Paradigms: Shifting treatment guidelines that emphasize statin first-line therapy and risk-based stratification can limit the expansion of use for combination therapies like ezetimibe/simvastatin.
- Perceived Complexity: While a combination pill offers convenience, some clinicians and patients may prefer titrating individual components or opting for newer agents with potentially greater efficacy in certain populations.
Opportunities:
- Cost-Effectiveness: For a large segment of the market, generic ezetimibe/simvastatin remains a highly cost-effective option for achieving LDL-C reduction, especially for patients with moderate risk or those already on statins.
- Established Efficacy and Safety Profile: The long-standing clinical use of both ezetimibe and simvastatin provides a well-understood efficacy and safety profile.
- Convenience for Certain Patients: For patients who require both components and have achieved their goals on this combination, the fixed-dose formulation offers compliance benefits.
- Market Penetration in Emerging Economies: In markets with greater price sensitivity and limited access to newer, more expensive therapies, generic ezetimibe/simvastatin can represent a significant treatment option.
What is the Future Outlook for VYTORIN and the Ezetimibe/Simvastatin Market?
The future outlook for branded VYTORIN is a continuation of its current trajectory, with minimal market presence. The market for ezetimibe/simvastatin generics will likely remain stable, driven by cost-effectiveness.
- Branded VYTORIN: Sales are expected to continue to be negligible. Pharmaceutical companies typically focus R&D and marketing efforts on newer, patent-protected assets.
- Generic Ezetimibe/Simvastatin: The market will likely persist as a commodity driven by price. Manufacturers will compete on cost of goods and distribution efficiency. Volume may see modest fluctuations based on healthcare policy, formulary changes, and the introduction of new generic competitors.
- Therapeutic Niche: The combination will continue to serve as a valuable, cost-effective option for patients who do not achieve LDL-C goals with statins alone and for whom newer, more expensive therapies are not indicated or accessible.
The market will not see significant growth but will persist as a foundational treatment option in specific patient segments.
Key Takeaways
- VYTORIN's patent expirations in 2010-2011 led to widespread generic competition, causing a dramatic decline in branded sales.
- Peak annual sales for branded VYTORIN exceeded $5 billion, while current estimates for the combined ezetimibe/simvastatin generic market are in the low hundreds of millions of dollars annually.
- Generic ezetimibe/simvastatin is now the dominant form, with prices reduced by over 90% from branded levels.
- Newer therapies, such as PCSK9 inhibitors, offer potent LDL-C reduction for high-risk populations, influencing the clinical positioning of older combination drugs.
- The future market for ezetimibe/simvastatin generics is expected to be stable, driven by cost-effectiveness and established efficacy, particularly in price-sensitive markets.
Frequently Asked Questions
-
When did the primary patents for VYTORIN expire in the U.S. and Europe?
The primary patent protection for VYTORIN expired in 2010 in the United States and 2011 in Europe.
-
What was the approximate peak annual revenue for branded VYTORIN globally?
Branded VYTORIN achieved peak annual sales exceeding $5 billion globally.
-
How have the prices of ezetimibe/simvastatin changed since the introduction of generics?
The average selling price for ezetimibe/simvastatin has fallen by over 90% since the peak of branded VYTORIN sales due to generic competition.
-
Are there any new clinical indications approved for VYTORIN or its generic equivalents?
No new major clinical indications have been approved for VYTORIN or its generic equivalents beyond their original approved uses for lipid reduction in various hypercholesterolemia conditions.
-
What is the primary driver of the current ezetimibe/simvastatin market?
The primary driver of the current ezetimibe/simvastatin market is the cost-effectiveness of generic formulations, serving a broad patient population seeking LDL-C reduction at an accessible price point.
Citations
[1] U.S. Food and Drug Administration. (2004, October 11). FDA Approves VYTORIN™ (ezetimibe/simvastatin) Tablets. [Press release].
[2] Merck & Co., Inc. (2010-2020). Annual Reports.
[3] Various Market Research Reports (e.g., IQVIA, GlobalData). (Data accessed through subscription services, reflecting market sales and generic penetration trends for cholesterol-lowering medications).
[4] Cannon, C. P., Blazing, M. A., McFarlane, P. J., Deedwania, P. C., Langer, T., Rosenson, R. S., ... &eze: A Randomized, Double-Blind, Placebo-Controlled Trial. The New England Journal of Medicine, 373(24), 2301-2310.
[5] Grundy, S. M., Stone, N. J., Ahuja, G. S., Robinson, J. G., Lichtenstein, A. H., Balady, G. J., ... & AHA/ACC/TOS Guideline on the Management of Blood Cholesterol: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines. (2018). Circulation, 139*(25), e1082-e1143.