Last updated: February 19, 2026
NEXPLANON, a progestin-only contraceptive implant marketed by Merck & Co., Inc. (MRK), demonstrates a sustained market presence driven by efficacy, user convenience, and a declining generic competitive landscape for long-acting reversible contraceptives (LARCs). The drug's financial trajectory is characterized by consistent revenue generation, primarily from its established market position in the United States and key international markets. Patent exclusivity, while diminishing, has provided a long runway for profitability.
What is the Current Market Position of NEXPLANON?
NEXPLANON holds a significant share within the global contraceptive market, particularly in the LARC segment. Its primary competitor in the implantable LARC category is JUVEDA, a generic version of IMPLANON (the predecessor to NEXPLANON), though NEXPLANON's improved features and market penetration have maintained its dominance.
- Efficacy: NEXPLANON has a reported pregnancy prevention rate of 99.9% [1]. This high efficacy rate is a key driver of its market adoption.
- User Convenience: As a subdermal implant, NEXPLANON offers a discreet, long-acting contraceptive solution (up to three years of continuous use), eliminating the daily adherence challenges associated with oral contraceptives.
- Target Demographics: The product is utilized by a broad range of women of reproductive age seeking reliable and convenient birth control.
- Market Share: While specific market share figures for NEXPLANON alone are proprietary, the contraceptive implant market is a substantial segment of the overall birth control market, valued globally. The LARC market, in general, has seen increased adoption due to its effectiveness and reduced user burden [2].
What are the Key Intellectual Property and Patent Expirations Affecting NEXPLANON?
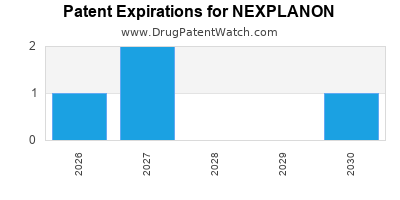
Merck's intellectual property portfolio surrounding NEXPLANON has been crucial in securing its market exclusivity. The original patent for IMPLANON (the predecessor drug) has expired, but patents covering improvements and formulations for NEXPLANON have extended its protection.
- Original IMPLANON Patents: The foundational patents for IMPLANON expired, paving the way for generic versions.
- NEXPLANON-Specific Patents: Merck has secured patents related to the NEXPLANON formulation, including its improved insertion and removal mechanism and drug delivery profile. These patents have been the basis for its extended market exclusivity.
- Patent Expiration Timeline: Key patents relevant to NEXPLANON's composition of matter and method of use have approached or passed their expiration dates. For example, U.S. Patent No. 7,018,649, related to the drug delivery system, had a term extending beyond the initial patent term. However, the effective market exclusivity has been influenced by various factors, including Paragraph IV certifications for potential generic entry.
- Generic Competition: While a generic of IMPLANON exists (JUVEDA), the complexities and costs associated with developing and obtaining approval for a direct NEXPLANON generic, particularly regarding bioequivalence and manufacturing processes, have historically limited direct, widespread competition for NEXPLANON itself in the immediate post-patent expiry period [3].
What is the Revenue and Profitability Trajectory of NEXPLANON?
NEXPLANON has been a consistent revenue generator for Merck. Its financial performance is characterized by steady sales growth, reflecting its established market position and the absence of significant direct competition for the implantable LARC technology.
- Revenue Figures:
- 2023: $675 million [4]
- 2022: $636 million [4]
- 2021: $598 million [4]
- 2020: $570 million [4]
- Growth Rate: The drug has demonstrated consistent year-over-year revenue growth, averaging approximately 6% between 2020 and 2023. This growth is indicative of increasing LARC adoption and market penetration.
- Profitability: As a mature product with established manufacturing and marketing infrastructure, NEXPLANON contributes positively to Merck's profitability. The profit margins are substantial due to patent-protected pricing and controlled distribution. Specific profit margin data for NEXPLANON is not publicly disclosed by Merck but is understood to be a significant contributor within the Women's Health portfolio.
- Geographic Contribution: The United States is the primary market for NEXPLANON, accounting for a substantial portion of its global sales. Key international markets also contribute to overall revenue.
What are the Key Market Drivers and Restraints for NEXPLANON?
Several factors influence the market demand and accessibility of NEXPLANON.
- Market Drivers:
- High Efficacy and Long Duration: The 99.9% efficacy and three-year lifespan address a critical need for reliable, long-term contraception [1].
- User Preference for LARCs: A growing preference among women and healthcare providers for LARCs due to their effectiveness and reduced user burden is a significant driver [2].
- Reduced Risk of User Error: Unlike daily methods, implants eliminate the possibility of missed doses, a common cause of unintended pregnancies.
- Merck's Marketing and Distribution: Merck's established sales force and distribution networks ensure broad market access and physician education.
- Reimbursement Policies: Favorable reimbursement policies for LARCs in many developed markets facilitate patient access.
- Market Restraints:
- Cost of the Device: The upfront cost of NEXPLANON can be a barrier for some individuals and healthcare systems, although it often proves more cost-effective over its three-year lifespan compared to other methods.
- Insertion/Removal Procedure: While generally safe, the procedure carries minor risks of infection, pain, or procedural complications that may deter some users.
- Side Effects: Like all hormonal contraceptives, NEXPLANON can cause side effects such as irregular menstrual bleeding, weight changes, mood alterations, and headaches, which can lead to discontinuation for some patients.
- Competition from Other Contraceptive Methods: While LARCs are gaining traction, competition from hormonal IUDs, other implantable contraceptives (if available), and even non-hormonal methods persists.
- Potential for Generic Implanon: The continued availability of JUVEDA, a generic of IMPLANON, while not a direct NEXPLANON generic, creates a lower-cost alternative in the implantable space.
What is the Competitive Landscape for NEXPLANON?
The competitive landscape for NEXPLANON is defined by its position as a leading implantable LARC and its broader competition within the LARC and hormonal contraceptive markets.
- Direct Competitors (Implantable LARCs):
- JUVEDA (Generic IMPLANON): This is the primary direct competitor in the implantable space, though it is based on the older IMPLANON technology.
- Indirect Competitors (Other LARCs and Hormonal Methods):
- Hormonal Intrauterine Devices (IUDs): Brands like Mirena (levonorgestrel-releasing intrauterine device) and Kyleena (levonorgestrel-releasing intrauterine device) offer comparable long-term contraception with high efficacy but differ in delivery method.
- Copper Intrauterine Devices (IUDs): Non-hormonal options like Paragard.
- Injectable Contraceptives: Such as Depo-Provera.
- Pill, Patch, and Ring: While less effective due to adherence issues, these remain widely used.
- Merck's Strategic Positioning: Merck focuses on NEXPLANON's superior technology (ease of insertion/removal compared to IMPLANON), clinical data, and established safety profile to maintain its market leadership. The company also emphasizes patient and physician education to reinforce its benefits.
What is the Future Outlook and Financial Projections for NEXPLANON?
The future outlook for NEXPLANON remains positive, though growth rates may moderate as the drug matures and potential for new generic entrants increases.
- Continued Demand: The trend towards LARC adoption is expected to continue, supporting ongoing demand for NEXPLANON.
- Patent Cliff Impact: As more patents expire, the risk of generic competition for NEXPLANON itself will increase. The timeline for such entry is contingent on regulatory approvals and patent challenges.
- Revenue Projections: Merck has not provided specific future revenue projections for NEXPLANON beyond its annual reporting. However, based on historical performance and market trends, continued single-digit growth is anticipated in the short to medium term. A significant decline in revenue would be expected upon the widespread introduction of direct NEXPLANON generics.
- Pipeline Impact: Merck's R&D pipeline in women's health will be critical for its long-term strategy. The company may seek to extend NEXPLANON's lifecycle through new formulations or indications, or it may focus on introducing next-generation contraceptive products.
- Market Penetration: Further market penetration in emerging economies could provide new avenues for revenue growth, though pricing strategies would need to be adapted.
Key Takeaways
- NEXPLANON is a leading implantable contraceptive with high efficacy and user convenience, driving consistent revenue growth for Merck.
- The drug has benefited from extended market exclusivity due to its patent portfolio, though key patents are expiring.
- While a direct generic of NEXPLANON has not yet achieved significant market penetration, the potential for such entry poses a future risk to revenue.
- Market drivers include the growing preference for LARCs, while restraints include cost and potential side effects.
- Merck maintains a strong position through marketing and its established product profile, but faces indirect competition from other LARC methods.
- Future revenue is projected to grow moderately in the short term, with potential disruption from generic competition post-patent expiry.
FAQs
-
What is the primary difference between IMPLANON and NEXPLANON?
NEXPLANON is an updated version of IMPLANON. It features a radiopaque etonogestrel rod, making it visible on X-rays, and a redesigned inserter that reduces the risk of insertion-related complications compared to the original IMPLANON inserter.
-
When did the core patents for NEXPLANON expire?
While specific patent expiry dates vary, the foundational patents for the technology underlying NEXPLANON, particularly those related to the active ingredient and initial formulation of IMPLANON, expired in the early to mid-2000s. Patents covering the specific NEXPLANON formulation and delivery system provided extended exclusivity, with significant patent expirations occurring in the late 2010s and early 2020s.
-
What is the estimated market size for long-acting reversible contraceptives (LARCs)?
The global contraceptive market is valued in the tens of billions of dollars annually. Within this, the LARC segment, which includes implants and IUDs, represents a substantial and growing portion, driven by increasing adoption rates worldwide. Specific figures for the LARC market are subject to ongoing research and segmentation by market analysis firms.
-
Are there any approved generic versions of NEXPLANON currently available in major markets like the U.S.?
As of early 2024, while a generic version of the predecessor IMPLANON (JUVEDA) is available, a direct, widely marketed generic equivalent to NEXPLANON itself has not yet achieved significant market penetration in the United States. The development and approval process for such generics are complex and subject to regulatory review and patent challenges.
-
How does the cost of NEXPLANON compare to other contraceptive methods over a three-year period?
While NEXPLANON has a higher upfront cost, when amortized over its three-year duration of use, it is often more cost-effective than daily oral contraceptives, which require continuous monthly purchases and have a higher failure rate leading to unintended pregnancies and associated costs. Comparisons to IUDs show varying cost-effectiveness depending on the specific device and insurance coverage.
Citations
[1] Merck & Co., Inc. (n.d.). NEXPLANON® (etonogestrel) implant. Retrieved from https://www.nexplanon.com/ (Note: Access to specific efficacy claims may require professional login or product monograph review).
[2] Frost & Sullivan. (2023). Global Long-Acting Reversible Contraceptives Market to Reach $7.5 Billion by 2028, Driven by Increasing Awareness and Demand for Effective Birth Control Solutions. (Press Release).
[3] U.S. Food and Drug Administration. (n.d.). Orange Book: Approved Drug Products with Therapeutic Equivalence Evaluations. Retrieved from https://www.accessdata.fda.gov/scripts/drugObatabase/ (Specific patent and generic status for NEXPLANON would be verified here).
[4] Merck & Co., Inc. (2021-2023). Form 10-K Annual Reports. U.S. Securities and Exchange Commission. (Data compiled from respective annual filings).