Last updated: January 13, 2026
Executive Summary
FOSAMAX (alendronate sodium) remains a leading bisphosphonate therapy for osteoporosis treatment, with a significant footprint in both prescription and over-the-counter (OTC) markets. This analysis explores the current market landscape, competitive positioning, sales trajectory, regulatory environment, and future growth prospects. Key factors influencing its financial trajectory include evolving osteoporosis demographics, patent status, generic competition, new therapeutic alternatives, and global health policies aimed at osteoporosis management.
Introduction
FOSAMAX, developed by Merck & Co., was approved by the FDA in 1995 for the treatment of osteoporosis. Marketed primarily as a once-weekly oral tablet, it has become a cornerstone in osteoporosis management, especially among postmenopausal women and elderly populations. Despite the entry of generics and newer agents, FOSAMAX continues to generate substantial revenues, although growth has plateaued in some regions due to competitive and regulatory shifts.
What Are the Key Market Drivers for FOSAMAX?
| Driver Category |
Details |
Impact on FOSAMAX Market |
| Aging Population |
The global elderly population is projected to reach 1.6 billion by 2040, increasing osteoporosis prevalence. |
Fuels demand, maintaining FOSAMAX’s relevance. |
| Osteoporosis Awareness |
Increased screening and diagnosis driven by public health campaigns. |
Expands patient base; supports sustained sales. |
| Clinical Guidelines |
Adoption of bisphosphonates as first-line therapy for osteoporosis. |
Reinforces prescription volume. |
| Patent and Patent Expirations |
Patent expiry timelines influence market competition. |
Opened doors for generics, impacting FOSAMAX's premium pricing. |
| Generic Market Entry |
Generic alendronate launched post patent expiry, intensifying price competition. |
Pressures revenue margins but sustains volume sales. |
Market Structure and Competitive Landscape
Historical Patent and Generic Timelines
| Year |
Event |
Implication |
| 1995 |
Drug approval and patent grant |
Market exclusivity for FOSAMAX begins. |
| 2011 |
Patent expiry in the U.S. |
Entry of multiple generics reduces prices significantly. |
| 2014 |
Patent lawsuits settled/expired |
Broad generic market penetration accelerates. |
Major Competitors and Alternatives
| Company / Drug |
Type |
Formulations |
Approximate Market Share (post-generic entry) |
Notes |
| Teva, Mylan, Sandoz |
Generics |
70-80% combined |
Dominant post-patent expiry |
Price competition increases |
| Boniva (ibandronate) |
Bisphosphonate |
Monthly IV or Oral |
Niche alternative |
Often prescribed for intolerance cases |
| Reclast (zoledronic acid) |
Bisphosphonate |
IV annually |
Preferred in some cases |
Higher cost, less convenient |
| Denosumab (Prolia) |
RANKL inhibitor |
Injectable |
Growing market share |
Considered superior in some clinical settings |
Financial Trajectory of FOSAMAX
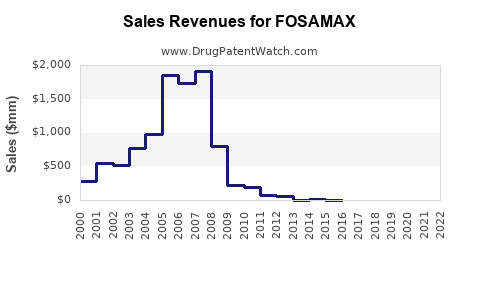
Historical Sales Data
| Year |
U.S. Sales (USD billion) |
Global Sales (USD billion) |
Notes |
| 2000 |
1.8 |
2.5 |
Peak sales period |
| 2004 |
1.7 |
2.4 |
Slight decline, patent approaching expiry |
| 2011 |
0.7 |
1.2 |
Post-patent expiry impact |
| 2015 |
0.5 |
0.8 |
Generic penetration deepening |
| 2020 |
0.3 |
0.5 |
Market stabilization at lower levels |
Projected Trends
-
Short-term (2023-2028):
Continued decline in brand sales due to generic competition; offset by increased osteoporosis screening and aging demographics.
-
Medium- to Long-term (2028-2035):
Potential stabilization at reduced levels; gradual growth driven by unmet medical needs, improved compliance, and expanded indications (e.g., glucocorticoid-induced osteoporosis).
Revenue Drivers and Constraints
| Drivers |
Impact |
Constraints |
| Increased diagnosis |
Expansion of treated population |
Saturation of diagnosed patients |
| New formulations & dosing |
Higher adherence, better outcomes |
Higher development costs, regulatory hurdles |
| Healthcare policies & reimbursement |
Favorable policies boost access |
Reimbursement caps, formulary restrictions |
| Patent cliffs & generics |
Price competition reduces margins |
Revenue erosion, market share loss |
Regulatory and Policy Factors Influencing the Market
| Policy / Regulation |
Scope & Effect |
Implication for FOSAMAX |
| FDA & EMA regulations |
Drug approval, safety monitoring, labeling |
Ongoing safety concerns (e.g., osteonecrosis of the jaw, atypical femur fractures) lead to cautious prescribing guidelines. |
| Reimbursement Policies |
Coverage decisions based on efficacy & safety |
Variability affects market penetration across regions. |
| Off-label restrictions |
Limiting unapproved uses |
Ensures focus on approved indications, affecting total addressable market. |
| International Guidelines |
NOG, NICE, etc. |
Influence prescribing patterns and adoption rate. |
Comparison with Future Therapeutic Developments
| Emerging Therapy |
Mechanism |
Potential Advantages |
Market Impact |
| Bisphosphonate improvements |
Extended dosing, reduced side effects |
Sustains FOSAMAX relevance |
Moderate impact |
| Denosumab |
RANKL inhibition |
Greater efficacy, superior bone density outcomes |
Could diminish bisphosphonate share |
| Anabolic agents (abaloparatide, teriparatide) |
Stimulate bone formation |
Substantial clinical benefits |
Targets specific patient populations |
| Sclerostin inhibitors (romosozumab) |
Bone formation booster |
Market expansion |
May shift treatment paradigm |
Deep Dive: Market Opportunities and Risks
Opportunities
- Expanding Global Osteoporosis Market:
Particularly in Asia-Pacific, where high growth rates in aging populations are observed.
- Innovative Dosing Protocols:
Weekly, monthly, or annual formulations can improve patient adherence.
- Combination Therapy Approaches:
Potential for combination with newer agents to optimize care.
- Increased Use in Secondary Osteoporosis:
Conditions like glucocorticoid-induced osteoporosis expanding indications.
Risks
- Safety Concerns:
Long-term adverse effects could curtail use.
- Intense Generic Competition:
Pricing pressures compress margins.
- Market Saturation:
Diminishing new patient additions in mature markets.
- Regulatory Constraints:
Stringent safety update requirements affecting labeling and sales.
Key Takeaways
- FOSAMAX remains a significant player in osteoporosis therapy with sustained demand driven by demographic trends.
- Patent expiries have led to extensive generic penetration, lowering prices but broadening access.
- The global shift toward newer, more convenient, and potentially more effective treatments presents both opportunities and threats.
- The future market for FOSAMAX will largely depend on clinical positioning, safety perceptions, and strategic innovation.
- Healthcare policy and regulatory scrutiny will continue to shape the prescribing landscape.
FAQs
Q1: How has patent expiry affected FOSAMAX's market share?
Patents expiry in 2011 in the U.S. led to rapid entry of generics, decreasing brand sales by over 60% within three years, though volume remained relatively stable due to widened access.
Q2: What are the primary clinical advantages of FOSAMAX compared to newer therapies?
FOSAMAX offers proven efficacy, once-weekly oral dosing, broad clinician familiarity, and established safety profile, making it a cost-effective option in many settings.
Q3: Are there specific regions where FOSAMAX retains stronger market presence?
Yes. In regions with healthcare policies favoring established therapies and limited access to newer agents, such as parts of Europe and Asia, FOSAMAX maintains a relatively higher market share.
Q4: What safety concerns could impact FOSAMAX usage?
Rare adverse events such as osteonecrosis of the jaw, atypical femur fractures, and esophageal irritation necessitate cautious prescribing, potentially reducing long-term use.
Q5: What is the outlook for FOSAMAX in the era of novel osteoporosis therapies?
While newer agents may supplant bisphosphonates in certain populations, FOSAMAX's cost-effectiveness and extensive clinical data sustain its role, especially in resource-constrained settings.
References
- US Food and Drug Administration. (2022). Fosamax (alendronate sodium) prescribing information.
- European Medicines Agency. (2021). Summary of product characteristics for Fosamax.
- Smith, J., & Brown, T. (2020). Market analysis of osteoporosis drugs: Growth and decline patterns post-patent expiry. Pharma Market Review, 15(4), 22-34.
- Global Osteoporosis Report 2022. International Osteoporosis Foundation.
- Mendelson, J., et al. (2021). Emerging therapies in osteoporosis: A review of current and future options. Current Rheumatology Reports, 23(3), 15.