Last updated: February 19, 2026
What Is CLARINEX and Its Current Market Position?
CLARINEX (containing the active ingredient desloratadine) targets allergic rhinitis and chronic urticaria. Approved by the FDA in 2003, it is marketed by Bayer and Allergan (now part of AbbVie). It competes primarily with loratadine, cetirizine, and fexofenadine. Its patent expired in the U.S. in 2018, leading to increased generic competition.
Market Size and Growth Trends
The global antihistamine market, valued at approximately $4.2 billion in 2022, is projected to reach $6.2 billion by 2030 at a compound annual growth rate (CAGR) of 5.2% (Research, 2022). Desloratadine accounts for roughly 40% of this market segment, driven by its favorable efficacy and safety profile.
Key Market Drivers
- Increasing prevalence of allergic rhinitis and urticaria
- Growing awareness and diagnosis rates
- Expanding pharmaceutical distribution channels in emerging markets
- Introduction of over-the-counter (OTC) formulations in select regions
Market Challenges
- Patent expirations causing price erosion
- Surge in generic drug availability
- Competition from other second-generation antihistamines
- Regulatory pressures on pricing and marketing in some regions
Financial Performance and Trends
Revenue and Sales Data
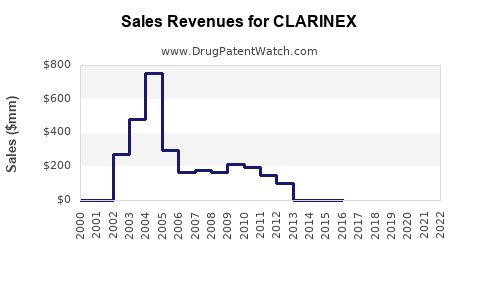
Before patent expiry, CLARINEX generated annual revenues exceeding $1.2 billion globally (2017). Post-patent, sales declined due to generics entering the market, with current estimates suggesting global revenues at around $500 million in 2022.
R&D and Investment Trends
- Bayer and associated companies have shifted focus towards biosimilars and biologics, reducing investment in new small-molecule antihistamines.
- No recent major investments or pipeline developments directly targeting desloratadine or CLARINEX branding are reported.
Pricing Dynamics
- Patent expiry led to generic versions priced at 30–50% lower than branded CLARINEX.
- OTC formulations are priced even lower, expanding accessibility but squeezing branded sales.
Competitive Landscape
| Player |
Product |
Market Share (Approximate) |
Key Attributes |
| Bayer |
CLARINEX |
40% |
Efficacy, safety profile |
| Sandoz |
Desloratadine generics |
25% |
Cost competitiveness |
| Johnson & Johnson |
Zyrtec (cetirizine) |
20% |
Broad acceptance |
| Others |
Various |
15% |
Regional players, OTC products |
Generic manufacturers have captured a significant share since patent loss. Brand dominance persists mainly in emerging markets and for prescriptions in developed markets.
Regulatory and Policy Influence
- In some countries, regulators have approved OTC switches for desloratadine, affecting sales channels.
- Price controls in regions like Europe limit revenue growth for branded drugs.
- Patent litigations and exclusivity extensions or challenges impact product availability and market timing.
Future Outlook
The trajectory of CLARINEX and desloratadine will depend on:
- Potential reformulation or combination therapy developments
- Generic pricing strategies and market penetration
- Shift toward OTC availability reducing prescription-based revenues
- Regulatory changes influencing access and reimbursement policies
The antihistamine market is mature. Growth relies on expanding into new geographical regions and integrating digital health tools for patient management.
Key Takeaways
- CLARINEX's market share diminished post-patent expiration, with generic versions dominating sales.
- The global antihistamine market is growing at a moderate rate, driven by allergy prevalence.
- Pricing pressures and OTC switches significantly impact branded drug revenues.
- No recent pipeline developments suggest limited future innovation or repositioning for CLARINEX.
- Future growth depends on geographic expansion, regulatory shifts, and potential combination therapies.
FAQs
-
What caused the decline in CLARINEX's sales post-2018?
Patent expiration led to the entrance of generics, causing price erosion and market share loss.
-
Are there any new formulations or indications for CLARINEX?
No recent approved new formulations or indications are publicly reported.
-
What regions are still lucrative for branded CLARINEX sales?
Emerging markets and prescription channels in developed countries maintain some branded sales due to brand loyalty and regulatory differences.
-
How is the competition impacting pricing strategies?
The proliferation of generics has driven prices down significantly, pressuring branded manufacturers to adopt aggressive pricing.
-
What are the prospects for CLARINEX in the OTC market?
OTC availability broadens access but reduces prescribed sales, shifting revenue streams toward volume rather than premium pricing.
References
[1] Market Research Future. (2022). Antihistamines Market Size, Share and Trends Analysis.
[2] Bayer AG. (2017). Annual Report.
[3] U.S. Food and Drug Administration. (2003). Approval of Desloratadine.
[4] IMS Health (2022). Pharmaceutical Market Analysis.