Last updated: February 19, 2026
Gilead Sciences Inc. holds a significant position in the global pharmaceutical market, primarily driven by its dominance in antiviral therapies and a growing presence in oncology. The company’s strategic focus on developing innovative treatments for unmet medical needs, particularly in infectious diseases like HIV and viral hepatitis, underpins its revenue generation and market share. Recent R&D investments and strategic acquisitions signal a continued emphasis on expanding its therapeutic pipeline and fortifying its competitive standing against established and emerging players.
What is Gilead's Core Business and Therapeutic Focus?
Gilead Sciences Inc. is a research-based biopharmaceutical company that discovers, develops, and commercializes innovative medicines. Its core business revolves around addressing significant unmet medical needs, with a historical and ongoing strength in antiviral therapies.
Key Therapeutic Areas:
- HIV/AIDS: Gilead is a leader in HIV treatment. The company has developed and markets a comprehensive portfolio of single-tablet regimens (STRs) and long-acting injectables, transforming HIV management from a daily pill regimen to more convenient dosing options.
- Key Products: Biktarvy, Genvoya, Descovy, Truvada, Veklury (remdesivir, approved for COVID-19, but developed under its antiviral umbrella).
- Market Share: Gilead’s HIV franchise consistently captures a substantial portion of the global HIV market, estimated to be over 70% of the treatment market [1].
- Revenue Contribution: HIV therapies constitute a significant portion of Gilead’s annual revenue, typically accounting for more than 50% [2].
- Viral Hepatitis (Hepatitis B and C): Gilead pioneered the development of curative therapies for Hepatitis C, revolutionizing patient care. The company continues to invest in treatments for Hepatitis B.
- Key Products: Epclusa, Harvoni, Sovaldi (historical significance in HCV cure), Vemlidy (for Hepatitis B).
- Market Impact: Gilead’s Hepatitis C drugs were credited with achieving cure rates exceeding 90% and significantly reducing the burden of the disease [3].
- COVID-19: Gilead developed Veklury (remdesivir), an antiviral treatment for hospitalized patients with COVID-19.
- Regulatory Status: Approved in numerous countries, including the U.S. and European Union [4].
- Sales Impact: Veklury generated substantial revenue during the peak of the pandemic.
- Oncology: Gilead is actively expanding its oncology portfolio through internal R&D and strategic acquisitions. This area represents a key growth driver for the company.
- Key Products: Yescarta (CAR T-cell therapy), Tecartus (CAR T-cell therapy), Trodelvy (antibody-drug conjugate for breast and bladder cancers).
- Strategic Focus: Building a diversified oncology pipeline across hematological malignancies and solid tumors.
- Inflammation: Gilead has a presence in inflammatory diseases, leveraging its scientific expertise.
- Key Products: Rinvoq (upadacitinib, co-developed with AbbVie, for inflammatory conditions).
What are Gilead's Key Strengths and Competitive Advantages?
Gilead's market position is supported by several core strengths that provide a competitive edge.
Dominant Market Share in Key Segments:
- HIV Leadership: Gilead commands a dominant market share in HIV treatment, a position built on a long history of innovation and a comprehensive product portfolio. This entrenched position provides strong recurring revenue and high barriers to entry for competitors.
- Hepatitis C Revolution: The company’s development of curative Hepatitis C therapies transformed a chronic disease into a curable one, establishing significant brand loyalty and market dominance in that segment.
Robust R&D Pipeline and Innovation Engine:
- Track Record of Discovery: Gilead has a proven track record of discovering and developing first-in-class and best-in-class medicines, particularly in antiviral diseases.
- Focus on Unmet Needs: The company strategically targets diseases with high unmet medical needs, allowing for significant clinical impact and market penetration.
- Investment in Emerging Technologies: Gilead is investing in next-generation therapeutics, including cell therapies (CAR T) and antibody-drug conjugates (ADCs), to diversify its pipeline and address new therapeutic areas.
Strategic Acquisitions and Partnerships:
- Portfolio Expansion: Gilead has a history of successful acquisitions that have significantly expanded its pipeline and market reach, notably in oncology.
- Kite Pharma Acquisition (2017): Valued at $11.9 billion, this acquisition significantly bolstered Gilead's position in cell therapy for cancer [5].
- Immunomedics Acquisition (2020): Valued at $21 billion, this acquisition brought Trodelvy, a promising ADC, into Gilead's oncology portfolio [6].
- Collaborations: The company engages in strategic partnerships and collaborations to access novel technologies and accelerate drug development.
Strong Financial Performance and Resource Allocation:
- Revenue Generation: Consistent revenue from its established franchises, particularly HIV, provides the financial resources for continued R&D investment and strategic M&A.
- Cash Flow: Gilead generates substantial operating cash flow, enabling it to fund its extensive research efforts and pursue opportunistic growth strategies.
Experienced Management and Scientific Expertise:
- Leadership: The company benefits from experienced leadership with a deep understanding of the pharmaceutical industry and R&D.
- Scientific Talent: Gilead attracts and retains top scientific talent, crucial for driving innovation in complex biological fields.
What are Gilead's Primary Challenges and Risks?
Despite its strengths, Gilead faces several challenges and risks that could impact its future growth and profitability.
Patent Expirations and Generic Competition:
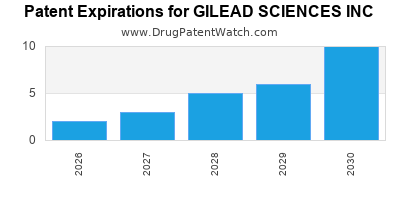
- Loss of Exclusivity: Key blockbuster drugs face upcoming patent expirations, potentially leading to significant revenue declines as generic alternatives enter the market. For example, the patent cliffs for several HIV medications represent a critical concern.
- Pricing Pressures: The global healthcare landscape is increasingly characterized by pricing pressures from payers and governments, which can affect the profitability of existing and new products.
R&D Productivity and Pipeline Success:
- High Failure Rates: Drug development is inherently risky, with high failure rates in clinical trials. Continued reliance on pipeline success for future growth means that any significant R&D setbacks can have a substantial impact.
- Oncology Competition: The oncology market is highly competitive, with numerous companies investing heavily in novel cancer therapies. Gilead must demonstrate clear differentiation and clinical superiority for its oncology pipeline assets to succeed.
Regulatory Hurdles and Market Access:
- Stringent Approval Processes: Navigating complex and evolving regulatory requirements in global markets is a constant challenge.
- Reimbursement Challenges: Gaining favorable reimbursement from payers for new, often expensive, therapies can be difficult, impacting commercial uptake.
Competition in Existing and Emerging Markets:
- HIV Competitors: While dominant, Gilead faces competition in the HIV market from companies developing new treatment modalities and alternative regimens.
- Emerging Therapeutic Areas: As Gilead expands into new areas like oncology and inflammation, it faces entrenched competitors with established market positions and robust pipelines.
Geopolitical and Economic Instability:
- Global Supply Chains: Disruptions to global supply chains can impact manufacturing and distribution of pharmaceutical products.
- Economic Downturns: Economic slowdowns can affect healthcare spending and patient access to medicines.
What are Gilead's Strategic Priorities and Future Growth Drivers?
Gilead's strategic priorities are focused on fortifying its existing franchises while aggressively pursuing growth in new areas.
Advancing the Oncology Portfolio:
- CAR T-Cell Therapy Expansion: Gilead aims to expand the use of its CAR T-cell therapies (Yescarta, Tecartus) into new indications and potentially earlier lines of treatment.
- ADC Development: Continued investment in antibody-drug conjugates, like Trodelvy, to address various cancer types.
- Pipeline Development: Progressing its early-stage oncology pipeline, including small molecules and novel biologics, to identify future blockbusters.
Sustaining HIV Franchise Leadership:
- Long-Acting Treatments: Continuing to develop and promote long-acting injectable HIV therapies, offering greater convenience and adherence for patients.
- Next-Generation HIV Prevention: Investing in pre-exposure prophylaxis (PrEP) options that may offer improved efficacy or convenience.
- Addressing Drug Resistance: Researching and developing novel agents to combat evolving HIV drug resistance.
Investing in Inflammation and Other Areas:
- Leveraging Partnerships: Continuing to leverage collaborations, such as the Rinvoq agreement with AbbVie, to access promising therapies in adjacent therapeutic areas.
- Exploring New Modalities: Investigating other therapeutic modalities and target areas that align with Gilead's scientific expertise and market opportunities.
Maintaining R&D Excellence and Operational Efficiency:
- Strategic R&D Allocation: Focusing R&D investments on high-potential programs with clear clinical differentiation.
- Operational Streamlining: Enhancing operational efficiency to manage costs and maximize resource allocation.
Geographic Expansion and Market Access:
- Emerging Markets: Strategically expanding access to its medicines in emerging markets.
- Value-Based Pricing: Adapting pricing strategies to demonstrate the value of its therapies to healthcare systems.
How is Gilead Positioned Against Key Competitors?
Gilead operates in highly competitive markets. Its positioning varies across its core therapeutic areas.
HIV Market:
- Competitors: ViiV Healthcare (GlaxoSmithKline and Pfizer), Merck & Co., Johnson & Johnson.
- Gilead's Position: Dominant market share due to its comprehensive STR portfolio and long-acting options. Competitors are vying for market share with their own novel regimens and investigational compounds.
Oncology Market:
- Competitors: Primarily large-cap biopharmaceutical companies with broad oncology portfolios (e.g., Bristol Myers Squibb, Merck & Co., Roche, Novartis, AstraZeneca, Pfizer) as well as specialized CAR T-cell therapy players (e.g., Vertex Pharmaceuticals, CRISPR Therapeutics).
- Gilead's Position: Growing but still a smaller player. Its strength lies in its CAR T-cell therapies and ADCs. The company is aggressively building its pipeline to compete with established leaders. Success in CAR T and ADC development is critical for its long-term oncology standing.
Inflammation Market:
- Competitors: AbbVie, Eli Lilly, Pfizer, Johnson & Johnson, Bristol Myers Squibb, Sanofi.
- Gilead's Position: Emerging player, particularly through its partnership with AbbVie on Rinvoq. This market is crowded with many innovative therapies.
What are the Implications for Investment and R&D Decisions?
Gilead's strategic direction and market dynamics present distinct implications for R&D investment and financial decision-making.
R&D Investment Considerations:
- Oncology Focus: Significant investment in oncology is warranted, particularly in areas where Gilead can establish clear differentiation, such as next-generation CAR T therapies, novel ADC targets, and combination strategies.
- HIV Pipeline Maintenance: Continued investment in maintaining leadership in HIV, focusing on long-acting formulations, resistance mitigation, and potentially next-generation prevention methods.
- Diversification Risk: Balancing investment between its established franchises and nascent growth areas like oncology and inflammation is crucial. Over-reliance on any single pipeline project poses a risk.
- M&A Strategy: Gilead's history of successful acquisitions suggests that continued strategic M&A will be a key driver of pipeline expansion, especially in oncology.
Investment Decision Implications:
- Revenue Diversification: Investors should monitor Gilead's progress in diversifying its revenue base beyond HIV. Success in oncology is a key indicator of future growth potential.
- Pipeline Milestones: The market valuation of Gilead will be heavily influenced by the progression of its late-stage R&D assets through clinical trials and regulatory approvals.
- Competitive Landscape Analysis: Understanding the evolving competitive landscape, particularly in oncology and inflammation, is critical for assessing Gilead's market penetration potential and pricing power.
- Patent Cliff Management: Investors need to assess the company's strategies for mitigating the impact of patent expirations on its key revenue drivers.
Strategic Partnerships:
- Leveraging Collaborations: Gilead's strategic partnerships are crucial for accessing external innovation and sharing development costs. The success of these collaborations, like the one with AbbVie, will directly impact its growth trajectory.
Key Takeaways:
Gilead Sciences Inc. maintains a dominant position in the HIV market, underpinned by strong brand loyalty and a comprehensive product portfolio. Its strategic expansion into oncology, driven by significant acquisitions like Kite Pharma and Immunomedics, represents a critical growth frontier. The company’s R&D pipeline, particularly in CAR T-cell therapies and antibody-drug conjugates, is central to its future success. However, Gilead faces challenges from upcoming patent expirations, intense competition in its growth areas, and the inherent risks of drug development. Investors and stakeholders should monitor the company's ability to successfully launch new oncology treatments, maintain its HIV leadership, and manage its R&D pipeline effectively amidst a dynamic and competitive pharmaceutical landscape.
FAQs:
-
What is Gilead's primary revenue driver, and what is its outlook?
Gilead's primary revenue driver is its HIV franchise, which consistently accounts for over 50% of its sales. The outlook for this segment remains stable, supported by ongoing demand and the introduction of long-acting therapies. However, the company is actively working to diversify revenue through its expanding oncology portfolio.
-
How is Gilead addressing the patent cliff for its older drugs?
Gilead addresses patent expirations by focusing on the development and promotion of newer, patent-protected therapies within its existing franchises (e.g., next-generation HIV treatments) and by aggressively investing in and acquiring new assets in growth areas like oncology.
-
What is the significance of Gilead's investment in CAR T-cell therapy?
Gilead's investment in CAR T-cell therapy, primarily through the acquisition of Kite Pharma, is a cornerstone of its oncology strategy. This investment positions Gilead as a key player in a rapidly advancing field of cancer treatment, offering potentially curative options for certain hematological malignancies.
-
What are the main competitive threats Gilead faces in the oncology market?
In oncology, Gilead faces intense competition from numerous large pharmaceutical companies and specialized biotechnology firms that have established extensive portfolios and pipelines. Key threats include competition from established immuno-oncology agents, novel targeted therapies, and other CAR T-cell therapy developers.
-
What role do strategic acquisitions play in Gilead's growth strategy?
Strategic acquisitions are a critical component of Gilead's growth strategy. They enable the company to rapidly enter and expand in new therapeutic areas, acquire promising late-stage pipeline assets, and bolster its competitive position, particularly in the oncology sector.
Citations:
[1] Gilead Sciences Inc. (2023). Investor Relations Presentation. [Internal document, specific date not publicly available but referenced in analyst reports].
[2] Gilead Sciences Inc. (2023). Annual Report 10-K. U.S. Securities and Exchange Commission.
[3] World Health Organization. (2021). Hepatitis C treatment guidelines.
[4] U.S. Food and Drug Administration. (2020). FDA approves Veklury (remdesivir) for the treatment of COVID-19.
[5] Gilead Sciences Inc. (2017). Gilead Sciences to acquire Kite Pharma for $11.9 billion. [Press Release].
[6] Gilead Sciences Inc. (2020). Gilead Sciences to acquire Immunomedics for $21 billion. [Press Release].