Bausch And Lomb Inc Company Profile
✉ Email this page to a colleague
What is the competitive landscape for BAUSCH AND LOMB INC, and what generic alternatives to BAUSCH AND LOMB INC drugs are available?
BAUSCH AND LOMB INC has nineteen approved drugs.
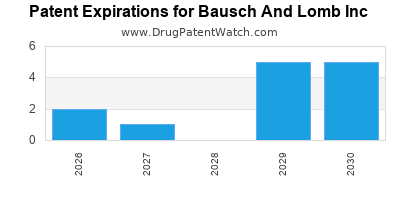
There are thirty-two US patents protecting BAUSCH AND LOMB INC drugs.
There are three hundred and seventy-four patent family members on BAUSCH AND LOMB INC drugs in thirty-one countries and forty-one supplementary protection certificates in fourteen countries.
Summary for Bausch And Lomb Inc
| International Patents: | 374 |
| US Patents: | 32 |
| Tradenames: | 19 |
| Ingredients: | 15 |
| NDAs: | 19 |
| Drug Master File Entries: | 1 |
Drugs and US Patents for Bausch And Lomb Inc
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Bausch And Lomb Inc | METHAZOLAMIDE | methazolamide | TABLET;ORAL | 207438-001 | Oct 5, 2018 | AB | RX | No | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bausch And Lomb Inc | OLOPATADINE HYDROCHLORIDE | olopatadine hydrochloride | SOLUTION/DROPS;OPHTHALMIC | 206046-001 | Jul 26, 2017 | DISCN | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bausch And Lomb Inc | MIEBO | perfluorohexyloctane | SOLUTION/DROPS;OPHTHALMIC | 216675-001 | May 18, 2023 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bausch And Lomb Inc | ALAWAY | ketotifen fumarate | SOLUTION/DROPS;OPHTHALMIC | 208158-001 | Sep 24, 2020 | OTC | No | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Bausch And Lomb Inc | TIMOPTIC-XE | timolol maleate | SOLUTION, GEL FORMING/DROPS;OPHTHALMIC | 020330-001 | Nov 4, 1993 | AB | RX | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Bausch And Lomb Inc | XIPERE | triamcinolone acetonide | SUSPENSION;INJECTION | 211950-001 | Oct 22, 2021 | RX | Yes | Yes | 9,937,075 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Bausch And Lomb Inc | MACUGEN | pegaptanib sodium | INJECTABLE;INTRAVITREAL | 021756-001 | Dec 17, 2004 | DISCN | Yes | No | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Bausch And Lomb Inc
Paragraph IV (Patent) Challenges for BAUSCH AND LOMB INC drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Ophthalmic Solution | 1.5% | ➤ Subscribe | 2013-09-09 |
International Patents for Bausch And Lomb Inc Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| New Zealand | 623752 | ⤷ Try a Trial |
| Japan | 2018531233 | ⤷ Try a Trial |
| Portugal | 2934510 | ⤷ Try a Trial |
| South Africa | 201400616 | ⤷ Try a Trial |
| Australia | 2013361579 | ⤷ Try a Trial |
| Australia | 2013295706 | ⤷ Try a Trial |
| South Korea | 102467714 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Bausch And Lomb Inc Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 0957929 | C 2007 038 | Romania | ⤷ Try a Trial | PRODUCT NAME: 5'-ESTERUL((2'-DEOXI-2'-FLUORO)C-GM-GM-A-A-(2'-DEOXI-2'-FLUORO)U-(2'-DEOXI-2'-FLUORO)C-AM-GM-(2'-DEOXI-2'-FLUORO)U-GM-AM-AM-(2'-DEOXI-2'-FLUORO)U-GM-(2'-DEOXI-2'-FLUORO)C-(2'-DEOXI-2'-FLUORO)U-(2'-DEOXI-2'-FLUORO)U-AM-(2'-DEOXI-2'-FLUORO)U-AM-(2'-DEOXI-2'-FLUORO)C-AM-(2'-DEOXI-2'-FLUORO)U-(2'-DEOXI-2'-FLUORO)C-(2'-DEOXI-2'-FLUORO)C-GM-(3'-3')-DT)CUALFA,ALFA'-[(1S)-1-[(5-(FOSFOONOXI)PENTIL]CARBAMOIL]PENTAN-1,5-DIIL]BIS(IMINOCARBONIL)]BIS[OMEGA-METOXIPOLI(OXIETAN-1,2-DIIL)], DE PREFERINTA SUB FORMA SARII DE SODIU - PEGAPTANIB, DE PREFERINTA SUB FORMA SARII DESODIU; NATIONAL AUTHORISATION NUMBER: EU/1/05/325/001; DATE OF NATIONAL AUTHORISATION: 20060131; NUMBER OF FIRST AUT [...] |
| 0957929 | 20/2006 | Austria | ⤷ Try a Trial | PRODUCT NAME: PEGAPTANIB UND SALZE DAVON; REGISTRATION NO/DATE: EU/1/05/325/001 20060131 |
| 1586316 | C300494 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: BROMFENAC, DESGEWENST IN DE VORM VAN EEN FARMACEUTISCH AANVAARDBAAR ZOUT OF HYDRAAT; REGISTRATION NO/DATE: EU/1/11/692/001 20110518 |
| 1631293 | C300683 | Netherlands | ⤷ Try a Trial | PRODUCT NAME: BRIMONIDINE EN FARMACEUTISCH AANVAARDBARE ZOUTEN DAARVAN VOOR TOEPASSING ALS GENEESMIDDEL VOOR DE BEHANDELING VAN DOOR ROSACEA GEINDUCEERDE ROODHEID; REGISTRATION NO/DATE: EU/1/13/904 20140225 |
| 0957929 | 06C0021 | France | ⤷ Try a Trial | PRODUCT NAME: PEGAPTANIB SODIUM; REGISTRATION NO/DATE: EU/1/05/325/001 20060131 |
| 1631293 | 2014C/042 | Belgium | ⤷ Try a Trial | PRODUCT NAME: MIRVASO (BRIMONIDINE) EN FARMACEUTISCHE ZOUTEN DAARVAN VOOR GEBRUIK ALS MEDICIJN VOOR HET BEHANDELEN VAN ROSACEA GEINDUCEERDE ROODHEID; AUTHORISATION NUMBER AND DATE: EU/1/13/904 20140221 |
| 2826776 | 13/2021 | Austria | ⤷ Try a Trial | PRODUCT NAME: KOMBINATION VON (A) (R)-1-(2,2-DIFLUORBENZO(D)(1,3)DIOXOL-5-YL)-N-(1-(2,3-DIHYDROXYPROPYL)-6-FLUOR-2-(1-HYDROXY-2-METHYLPROPAN-2-YL)-1H-INDOL-5-YL) CYCLOPROPANCARBOXAMID UND (B) N-(5-HYDROXY-2,4-DI-TERT-BUTYLPHENYL)-4-OXO-1H-CHINOLIN-3-CARBOXAMID; REGISTRATION NO/DATE: EU/1/18/1306 (MITTEILUNG) 20181106 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.