ALNYLAM PHARMS INC Company Profile
✉ Email this page to a colleague
What is the competitive landscape for ALNYLAM PHARMS INC, and when can generic versions of ALNYLAM PHARMS INC drugs launch?
ALNYLAM PHARMS INC has four approved drugs.
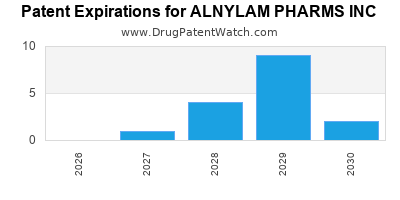
There are forty-four US patents protecting ALNYLAM PHARMS INC drugs.
There are seven hundred and seventy patent family members on ALNYLAM PHARMS INC drugs in fifty-four countries and seventy-five supplementary protection certificates in seventeen countries.
Summary for ALNYLAM PHARMS INC
| International Patents: | 770 |
| US Patents: | 44 |
| Tradenames: | 4 |
| Ingredients: | 4 |
| NDAs: | 4 |
Drugs and US Patents for ALNYLAM PHARMS INC
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Alnylam Pharms Inc | OXLUMO | lumasiran sodium | SOLUTION;SUBCUTANEOUS | 214103-001 | Nov 23, 2020 | RX | Yes | Yes | 10,131,907 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Alnylam Pharms Inc | AMVUTTRA | vutrisiran sodium | SOLUTION;SUBCUTANEOUS | 215515-001 | Jun 13, 2022 | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | |||||
| Alnylam Pharms Inc | OXLUMO | lumasiran sodium | SOLUTION;SUBCUTANEOUS | 214103-001 | Nov 23, 2020 | RX | Yes | Yes | 10,612,024 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Alnylam Pharms Inc | GIVLAARI | givosiran sodium | SOLUTION;SUBCUTANEOUS | 212194-001 | Nov 20, 2019 | RX | Yes | Yes | 9,708,615 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Alnylam Pharms Inc | OXLUMO | lumasiran sodium | SOLUTION;SUBCUTANEOUS | 214103-001 | Nov 23, 2020 | RX | Yes | Yes | 11,060,093 | ⤷ Try a Trial | Y | Y | ⤷ Try a Trial | ||
| Alnylam Pharms Inc | GIVLAARI | givosiran sodium | SOLUTION;SUBCUTANEOUS | 212194-001 | Nov 20, 2019 | RX | Yes | Yes | 10,273,477 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for ALNYLAM PHARMS INC
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Alnylam Pharms Inc | ONPATTRO | patisiran sodium | SOLUTION;INTRAVENOUS | 210922-001 | Aug 10, 2018 | 9,943,538 | ⤷ Try a Trial |
| Alnylam Pharms Inc | ONPATTRO | patisiran sodium | SOLUTION;INTRAVENOUS | 210922-001 | Aug 10, 2018 | 9,193,753 | ⤷ Try a Trial |
| Alnylam Pharms Inc | GIVLAARI | givosiran sodium | SOLUTION;SUBCUTANEOUS | 212194-001 | Nov 20, 2019 | 9,708,610 | ⤷ Try a Trial |
| Alnylam Pharms Inc | ONPATTRO | patisiran sodium | SOLUTION;INTRAVENOUS | 210922-001 | Aug 10, 2018 | 8,552,171 | ⤷ Try a Trial |
| Alnylam Pharms Inc | GIVLAARI | givosiran sodium | SOLUTION;SUBCUTANEOUS | 212194-001 | Nov 20, 2019 | 8,546,143 | ⤷ Try a Trial |
| Alnylam Pharms Inc | GIVLAARI | givosiran sodium | SOLUTION;SUBCUTANEOUS | 212194-001 | Nov 20, 2019 | 9,708,615 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
International Patents for ALNYLAM PHARMS INC Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 6132321 | ⤷ Try a Trial |
| European Patent Office | 3087184 | ⤷ Try a Trial |
| Hungary | E059857 | ⤷ Try a Trial |
| Peru | 20142362 | ⤷ Try a Trial |
| New Zealand | 592867 | ⤷ Try a Trial |
| Singapore | 11201602631X | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for ALNYLAM PHARMS INC Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 3329002 | LUC00297 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: VUTRISIRAN SOUS TOUTES LES FORMES PROTEGEES PAR LE BREVET DE BASE; AUTHORISATION NUMBER AND DATE: EU/1/22/1681 20220916 |
| 3204015 | LUC00252 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: LUMASIRAN SOUS TOUTES SES FORMES PROTEGEES PAR LE BREVET DE BASE; AUTHORISATION NUMBER AND DATE: EU/1/20/1496 20201123 |
| 3581654 | 2021038 | Norway | ⤷ Try a Trial | PRODUCT NAME: LUMASIRAN; REG. NO/DATE: EU/1/20/1496 20201124 |
| 3052628 | 2020/038 | Ireland | ⤷ Try a Trial | PRODUCT NAME: GIVOSIRAN OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF; NAT REGISTRATION NO/DATE: EU/1/20/1428 20200304; FIRST REGISTRATION NO/DATE: EU/1/20/1428 04/03/2020 |
| 3052628 | C20200026 00361 | Estonia | ⤷ Try a Trial | PRODUCT NAME: GIVOSIRAAN;REG NO/DATE: EU/1/20/1428 04.03.2020 |
| 3329002 | C03329002/01 | Switzerland | ⤷ Try a Trial | PRODUCT NAME: VUTRISIRAN; REGISTRATION NO/DATE: SWISSMEDIC-ZULASSUNG 69074 23.06.2023 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.