Horizon Company Profile
✉ Email this page to a colleague
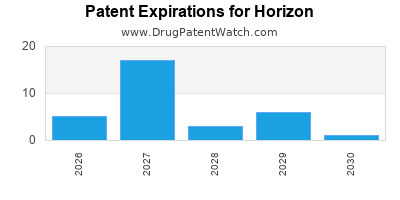
What is the competitive landscape for HORIZON, and when can generic versions of HORIZON drugs launch?
HORIZON has nine approved drugs.
There are fifty-seven US patents protecting HORIZON drugs.
There are two hundred and thirty-one patent family members on HORIZON drugs in forty-four countries and one hundred and one supplementary protection certificates in eighteen countries.
Summary for Horizon
| International Patents: | 231 |
| US Patents: | 57 |
| Tradenames: | 7 |
| Ingredients: | 7 |
| NDAs: | 9 |
| Patent Litigation for Horizon: | See patent lawsuits for Horizon |
| PTAB Cases with Horizon as patent owner: | See PTAB cases with Horizon as patent owner |
Drugs and US Patents for Horizon
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Horizon | PROCYSBI | cysteamine bitartrate | GRANULE, DELAYED RELEASE;ORAL | 213491-002 | Feb 14, 2020 | RX | Yes | Yes | 9,925,156*PED | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Horizon | DUEXIS | famotidine; ibuprofen | TABLET;ORAL | 022519-001 | Apr 23, 2011 | DISCN | Yes | No | 8,449,910 | ⤷ Try a Trial | Y | ⤷ Try a Trial | |||
| Horizon | PENNSAID | diclofenac sodium | SOLUTION;TOPICAL | 204623-001 | Jan 16, 2014 | AB | RX | Yes | Yes | 8,741,956 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Horizon Therap Us | BUPHENYL | sodium phenylbutyrate | TABLET;ORAL | 020572-001 | May 13, 1996 | AB | RX | Yes | Yes | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| Horizon | PENNSAID | diclofenac sodium | SOLUTION;TOPICAL | 204623-001 | Jan 16, 2014 | AB | RX | Yes | Yes | 9,375,412 | ⤷ Try a Trial | ⤷ Try a Trial | |||
| Horizon Therap Us | RAVICTI | glycerol phenylbutyrate | LIQUID;ORAL | 203284-001 | Feb 1, 2013 | RX | Yes | Yes | 10,183,005 | ⤷ Try a Trial | ⤷ Try a Trial | ||||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
Expired US Patents for Horizon
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | Patent No. | Patent Expiration |
|---|---|---|---|---|---|---|---|
| Horizon | RAYOS | prednisone | TABLET, DELAYED RELEASE;ORAL | 202020-001 | Jul 26, 2012 | 9,040,085 | ⤷ Try a Trial |
| Horizon | VIMOVO | esomeprazole magnesium; naproxen | TABLET, DELAYED RELEASE;ORAL | 022511-002 | Apr 30, 2010 | 9,161,920 | ⤷ Try a Trial |
| Horizon | RAYOS | prednisone | TABLET, DELAYED RELEASE;ORAL | 202020-001 | Jul 26, 2012 | 8,309,124 | ⤷ Try a Trial |
| Horizon | VIMOVO | esomeprazole magnesium; naproxen | TABLET, DELAYED RELEASE;ORAL | 022511-001 | Apr 30, 2010 | 8,557,285 | ⤷ Try a Trial |
| Horizon | VIMOVO | esomeprazole magnesium; naproxen | TABLET, DELAYED RELEASE;ORAL | 022511-002 | Apr 30, 2010 | 8,858,996 | ⤷ Try a Trial |
| Horizon | VIMOVO | esomeprazole magnesium; naproxen | TABLET, DELAYED RELEASE;ORAL | 022511-001 | Apr 30, 2010 | 8,858,996 | ⤷ Try a Trial |
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >Patent No. | >Patent Expiration |
Paragraph IV (Patent) Challenges for HORIZON drugs
| Drugname | Dosage | Strength | Tradename | Submissiondate |
|---|---|---|---|---|
| ➤ Subscribe | Topical Solution | 2.0% | ➤ Subscribe | 2014-06-03 |
| ➤ Subscribe | Delayed-release Tablet | 375 mg/20 mg and 500 mg/20 mg | ➤ Subscribe | 2010-11-05 |
| ➤ Subscribe | Oral Liquid | 1.1 g/mL | ➤ Subscribe | 2013-11-19 |
| ➤ Subscribe | Topical Solution | 1.5% | ➤ Subscribe | 2012-07-11 |
| ➤ Subscribe | Delayed-release Tablets | 1 mg, 2 mg, and 5 mg | ➤ Subscribe | 2012-11-26 |
| ➤ Subscribe | Tablets | 800 mg/26.6 mg | ➤ Subscribe | 2011-12-06 |
International Patents for Horizon Drugs
| Country | Patent Number | Estimated Expiration |
|---|---|---|
| Japan | 6468661 | ⤷ Try a Trial |
| China | 110664780 | ⤷ Try a Trial |
| European Patent Office | 2438919 | ⤷ Try a Trial |
| Brazil | 112015031417 | ⤷ Try a Trial |
| European Patent Office | 2330892 | ⤷ Try a Trial |
| Lithuania | 2535044 | ⤷ Try a Trial |
| >Country | >Patent Number | >Estimated Expiration |
Supplementary Protection Certificates for Horizon Drugs
| Patent Number | Supplementary Protection Certificate | SPC Country | SPC Expiration | SPC Description |
|---|---|---|---|---|
| 2330892 | 16C1020 | France | ⤷ Try a Trial | PRODUCT NAME: PHENYLBUTYRATE DE GLYCEROL; REGISTRATION NO/DATE: EU/1/15/1062 20151201 |
| 2465580 | SPC/GB21/030 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: CABOTEGRAVIR OR A PHARMACEUTICALLY ACCEPTABLE SALT OR SOLVATE THEREOF, INCLUDING CABOTEGRAVIR SODIUM.; REGISTERED: UK EU/1/20/1481 (NI) 20201221; UK PLGB 35728/0055-57 20201221 |
| 1758590 | LUC00029 | Luxembourg | ⤷ Try a Trial | PRODUCT NAME: SEL DE SODIUM D'ACIDE DEOXYCHOLIQUE; AUTHORISATION NUMBER AND DATE: IS/1/16/071/01 20170401 |
| 2822954 | 18C1035 | France | ⤷ Try a Trial | PRODUCT NAME: BICTEGRAVIR OU UN SEL PHARMACEUTIQUEMENT ACCEPTABLE,EN PARTICULIER BICTEGRAVIR DE SODIUM; REGISTRATION NO/DATE: EU/1/18/1289 20180625 |
| 2666774 | CA 2020 00037 | Denmark | ⤷ Try a Trial | PRODUCT NAME: RELEBACTAM, OPTIONALLY IN THE FORM OF THE MONOHYDRATE, IMIPENEM AND CILASTATIN, OPTIONALLY IN THE FORM OF THE SODIUM SALT; REG. NO/DATE: EU/1/19/1420 20200217 |
| 0480717 | SPC/GB98/025 | United Kingdom | ⤷ Try a Trial | PRODUCT NAME: MONTELUKAST, OR A PHARMACEUTICALLY ACCEPTABLE SALT THEREOF, PREFERABLY MONTELUKAST SODIUM; REGISTERED: FI 12766 19970825; FI 12767 19970825; UK 00025/0357 19980115; UK 00025/0358 19980115 |
| >Patent Number | >Supplementary Protection Certificate | >SPC Country | >SPC Expiration | >SPC Description |
Similar Applicant Names
Here is a list of applicants with similar names.