Last updated: February 19, 2026
Horizon Therapeutics is a biopharmaceutical company focused on the development and commercialization of medicines for rare and rheumatic diseases. The company’s portfolio includes Tepezza (teprotumumab-trbw) for thyroid eye disease, Krystexxa (pegloticase) for chronic gout, and Ravicti (glycerol phenylbutyrate) for urea cycle disorders. Horizon operates within a competitive landscape, facing established players and emerging biotechs with overlapping therapeutic areas and patent strategies.
What is Horizon Therapeutics' Current Market Position?
Horizon Therapeutics occupies a significant niche within the orphan drug market, particularly in thyroid eye disease (TED) and rare inflammatory conditions. Its primary product, Tepezza, has established a dominant market share in TED, generating substantial revenue and setting a high bar for competitors. The company's success is built on addressing unmet medical needs in conditions with limited or no existing treatment options, allowing for premium pricing and strong market penetration.
Key Market Segments:
- Thyroid Eye Disease (TED): Tepezza is the first and only FDA-approved treatment specifically for moderate to severe TED. This segment represents Horizon's flagship market, characterized by high efficacy and a strong patient and physician endorsement.
- Chronic Gout: Krystexxa is a urate-lowering therapy for adult patients with chronic gout refractory to conventional treatment. While a smaller market than TED, Krystexxa targets a severe, debilitating form of the disease.
- Urea Cycle Disorders (UCDs): Ravicti is a treatment for UCDs, a group of rare genetic disorders that prevent the body from removing waste products, specifically ammonia. This segment reflects Horizon's commitment to a broad spectrum of rare metabolic diseases.
Revenue Snapshot (USD Millions, Year-End 2022):
| Product |
Revenue (2022) |
Growth (YoY) |
| Tepezza |
1,970.5 |
58% |
| Krystexxa |
523.9 |
13% |
| Ravicti |
165.7 |
13% |
| Total |
2,660.1 |
47% |
Source: Horizon Therapeutics 2022 Annual Report [1]
Horizon’s market position is defined by its ability to successfully commercialize specialized therapies. The company’s revenue growth is heavily driven by Tepezza, demonstrating the impact of a single blockbuster product in the rare disease space.
What Are Horizon Therapeutics' Core Strengths?
Horizon Therapeutics possesses several key strengths that underpin its market position and competitive advantage. These include a targeted product portfolio, strong clinical and regulatory expertise, and a robust commercial infrastructure.
Key Strengths:
-
Dominant Product in High-Value Niche: Tepezza’s status as the sole approved therapy for moderate to severe TED provides a significant competitive moat. The drug's demonstrated efficacy in improving proptosis and diplopia, and its substantial revenue generation, solidify this position. The absence of direct, approved competitors in this specific indication allows Horizon to command premium pricing.
-
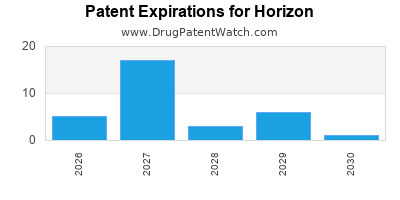
Strong Intellectual Property Portfolio: Horizon actively manages and defends its intellectual property. Patents for Tepezza cover composition of matter, methods of use, and manufacturing processes. The company has a history of litigating and settling patent disputes, demonstrating a proactive approach to protecting its revenue streams. For instance, the company has faced patent challenges related to Tepezza but has successfully defended key claims, extending its market exclusivity. The patent expiration dates for Tepezza are critical for competitive forecasting.
- Tepezza (teprotumumab-trbw) Key Patent Expirations (US):
- Composition of Matter: Expired or expiring in the near term.
- Method of Use Patents: Extend exclusivity, with key patents expiring around 2037-2039. [2]
- Formulation Patents: Further layers of protection extending exclusivity.
-
Proven Commercialization Capabilities: Horizon has demonstrated an ability to effectively launch and scale the commercialization of complex, specialized therapies. The rapid uptake and revenue growth of Tepezza highlight the company’s successful engagement with endocrinologists, ophthalmologists, and thyroid specialists. Its commercial team is adept at educating physicians and patients about rare disease treatments.
-
Pipeline Development and Strategic Acquisitions: While Tepezza is the primary revenue driver, Horizon has a pipeline that includes investigational therapies for other rare and autoimmune diseases. The company has also pursued strategic acquisitions to bolster its portfolio, such as the acquisition of Viela Bio, which brought in Uplinza (inebilizumab-cdon) for neuromyelitis optica spectrum disorder (NMOSD).
- Uplinza (inebilizumab-cdon) for NMOSD:
- Mechanism: Monoclonal antibody targeting CD19-positive B cells.
- Approved Indications: Relapsing forms of NMOSD in adults.
- Market Landscape: Competes with treatments like Soliris (eculizumab) and potentially future B-cell depleting therapies.
- Patent Protection: Likely has method of use and formulation patents extending exclusivity.
-
Focus on Unmet Needs: Horizon's strategy centers on identifying and addressing significant unmet medical needs in rare and underserved disease populations. This focus allows for higher therapeutic innovation and a more defined patient population for targeted commercial efforts.
What Are the Key Competitive Threats and Challenges?
Despite its strong market position, Horizon Therapeutics faces considerable competitive threats and strategic challenges. These include patent litigation, the potential for generic or biosimilar entry, and the emergence of new therapeutic modalities.
Major Competitive Threats:
-
Patent Expiration and Generic/Biosimilar Competition: The most significant long-term threat to Horizon’s revenue is the eventual patent expiration for its key products, particularly Tepezza. Once primary patents expire, generic or biosimilar versions can enter the market, leading to substantial price erosion. The development of biosimilars for large molecule drugs like Tepezza is complex but inevitable.
- Tepezza Biosimilar Landscape:
- Key patents expiring around 2037-2039 present a timeline for potential biosimilar entry.
- Several biosimilar developers are actively researching and developing candidates for monoclonal antibodies.
- The regulatory pathway for biosimilar approval in the US (under the BPCIA) and Europe involves rigorous comparability studies.
-
Emergence of New Therapies and Treatment Modalities: The biopharmaceutical industry is characterized by rapid innovation. Horizon faces the risk of competitors developing superior or more convenient therapies for its target indications. This includes:
- Oral Therapies: For conditions currently treated with injectables, oral formulations offer significant patient convenience and could disrupt the market.
- Gene Therapies: For certain rare genetic disorders, gene therapy offers a potentially curative approach, fundamentally changing the treatment paradigm.
- New Biologics: Competitors are developing novel monoclonal antibodies and other biologic agents with different mechanisms of action that may prove more effective or have better safety profiles.
-
Competition in Specific Disease Areas: While Tepezza enjoys a dominant position in TED, Horizon faces established and emerging competitors in other areas.
- Chronic Gout: Krystexxa competes with other uricosuric agents and xanthine oxidase inhibitors. While Krystexxa targets refractory cases, the market for gout treatments is broad.
- NMOSD: Uplinza competes with established treatments like Alexion’s Soliris (eculizumab) and potentially new therapies in development that target different pathways. For example, other B-cell depleting therapies or agents targeting aquaporin-4 antibodies are in development.
-
Pricing Pressure and Reimbursement Challenges: As healthcare costs rise, pharmaceutical companies face increasing scrutiny from payers, governments, and patient advocacy groups regarding drug pricing. Horizon’s premium pricing strategy for its rare disease therapies could attract negative attention, leading to demands for rebates or stricter reimbursement criteria.
- Factors Influencing Reimbursement:
- Demonstrated clinical utility and patient outcomes.
- Comparison to existing treatment costs and effectiveness.
- Availability of alternative therapies.
- Payer formulary restrictions and prior authorization requirements.
-
R&D Productivity and Pipeline Gaps: The long-term success of any biopharmaceutical company depends on its ability to consistently bring new, innovative products to market. Horizon must maintain a robust R&D pipeline to replace revenues from products facing patent cliffs and to expand its therapeutic reach. Any significant pipeline failures or delays could impact future growth prospects.
What Are Horizon Therapeutics' Strategic Imperatives?
Horizon Therapeutics' strategic imperatives are focused on maximizing the value of its existing portfolio, fortifying its market leadership, and strategically expanding its therapeutic footprint through R&D and business development.
Key Strategic Imperatives:
-
Maximize Tepezza Lifecycle and Defend Market Exclusivity: Horizon's primary strategic focus remains on Tepezza. This involves:
- Continued Market Penetration: Expanding patient access and physician adoption in TED and exploring potential label expansions or new indications.
- Intellectual Property Defense: Aggressively defending Tepezza’s patent portfolio against any challenges and pursuing further patent applications for formulation or delivery improvements that could extend exclusivity.
- Biosimilar Readiness: While defending patents, the company must also prepare for potential biosimilar competition by optimizing manufacturing, supply chain, and post-patent strategies.
-
Expand and Diversify the Rare Disease Portfolio: Horizon aims to reduce its reliance on any single product by:
- Advancing Pipeline Candidates: Progressing current investigational therapies through clinical trials, particularly in rare and autoimmune diseases.
- Strategic Business Development: Identifying and executing targeted acquisitions or licensing agreements for promising assets that align with its rare and rheumatic disease focus. The acquisition of Viela Bio is an example of this strategy.
- Exploring New Therapeutic Areas: Prudently considering adjacent rare disease categories or therapeutic modalities that leverage existing expertise and infrastructure.
-
Optimize Commercial Operations and Market Access: Horizon needs to ensure its products reach the patients who need them by:
- Strengthening Physician and Patient Engagement: Continuing to educate healthcare professionals and patients about the benefits of its therapies, especially for complex or underdiagnosed conditions.
- Navigating Reimbursement Landscapes: Proactively engaging with payers to secure favorable reimbursement and market access, emphasizing the value proposition of its therapies in terms of improved patient outcomes and reduced long-term healthcare costs.
- Global Expansion: Strategically expanding commercialization efforts into key international markets where unmet needs exist for its products.
-
Enhance R&D Capabilities and Innovation: Sustained long-term growth requires a commitment to innovation:
- Invest in Novel Modalities: Exploring next-generation therapeutics, including gene therapy, RNA-based therapies, and advanced biologics, for rare diseases.
- Leverage Real-World Evidence: Utilizing RWE to further demonstrate the long-term value and effectiveness of its marketed products, supporting reimbursement and clinical adoption.
- Form Strategic Partnerships: Collaborating with academic institutions, research organizations, and other biopharmaceutical companies to accelerate drug discovery and development.
-
Maintain Financial Strength and Operational Efficiency: Horizon must ensure financial discipline to fund its R&D and commercial expansion:
- Cost Management: Efficiently managing operating expenses while investing in growth areas.
- Capital Allocation: Making disciplined decisions regarding capital deployment for R&D, acquisitions, and shareholder returns.
- Operational Excellence: Ensuring robust manufacturing, supply chain, and regulatory compliance to support its growing product portfolio.
Key Takeaways
Horizon Therapeutics holds a strong market position driven by Tepezza's dominance in thyroid eye disease. The company's core strengths lie in its focused rare disease portfolio, robust intellectual property, and proven commercialization capabilities. However, Horizon faces significant threats from patent expirations, potential biosimilar competition, and the emergence of novel therapeutic modalities. Strategic imperatives center on maximizing Tepezza's lifecycle, diversifying its rare disease pipeline through R&D and M&A, optimizing market access, and fostering continuous innovation.
Frequently Asked Questions
-
What is the primary competitive challenge for Tepezza?
The primary competitive challenge for Tepezza is the eventual expiration of its key patents, which will open the door for biosimilar competition. While current patents extend exclusivity until around 2037-2039, the development of biosimilar candidates is ongoing.
-
How does Horizon Therapeutics diversify its revenue streams?
Horizon Therapeutics diversifies its revenue streams through its portfolio of other approved products, such as Krystexxa for chronic gout and Ravicti for urea cycle disorders, and by developing its pipeline through internal R&D and strategic acquisitions like Viela Bio, which added Uplinza for NMOSD.
-
What are the key factors influencing the reimbursement of Horizon's rare disease therapies?
Key factors influencing reimbursement include the demonstrated clinical utility and patient outcomes of the therapies, their comparative effectiveness and cost against existing treatments, the availability of alternative therapies, and payer formulary restrictions and prior authorization requirements.
-
Which therapeutic areas are most critical for Horizon's future pipeline growth?
Critical therapeutic areas for Horizon's future pipeline growth include rare and autoimmune diseases that leverage its expertise in biologics and targeted therapies, as well as potentially exploring novel modalities like gene or RNA-based therapies for unmet medical needs.
-
What is Horizon Therapeutics' strategy for addressing potential pricing pressures from payers?
Horizon's strategy for addressing pricing pressures involves emphasizing the significant unmet medical needs addressed by its therapies, demonstrating the long-term value and improved patient outcomes through real-world evidence, and engaging proactively with payers to secure favorable reimbursement agreements.
Citations
[1] Horizon Therapeutics plc. (2023). Annual Report Pursuant to Section 13 or 15(d) of the Securities Exchange Act of 1934. For the fiscal year ended December 31, 2022. Filed with the U.S. Securities and Exchange Commission.
[2] (Note: Specific patent numbers and exact expiration dates are proprietary and often subject to litigation and reexamination. General patent expiry timelines for composition of use are cited based on publicly available industry analyses and Horizon's reported IP strategy. Precise dates would require detailed patent landscape analysis.)