Last updated: February 19, 2026
VIMOVO, a fixed-dose combination of naproxen and esomeprazole magnesium, targets the symptomatic treatment of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis. Its market performance is shaped by patent exclusivity, competitive landscape, and physician adoption. Financial projections depend on market share retention post-patent expiry and potential for pipeline development.
What is the Patent Status of VIMOVO?
The patent landscape for VIMOVO is critical to its market longevity and financial projections. Original patents for the fixed-dose combination have expired or are nearing expiry in key markets, opening the door for generic competition.
- Original Combination Patents: The foundational patents covering the VIMOVO combination of naproxen and esomeprazole magnesium have largely expired. For instance, in the United States, the last of the key patents expired in 2017-2018 (USPTO).
- Formulation and Method of Use Patents: While the core combination is off-patent, pharmaceutical companies often hold secondary patents related to specific formulations, manufacturing processes, or methods of use. The status of these secondary patents can influence the timeline and ease of generic entry. Data from the U.S. Food and Drug Administration (FDA) Orange Book indicates multiple patents were listed for VIMOVO, with varying expiration dates.
- Exclusivity Periods: VIMOVO benefited from market exclusivity periods granted by regulatory bodies like the FDA. These exclusivity periods, separate from patent terms, can delay generic approval. However, these have also expired for the originator product.
- Generic Entry: The expiration of primary patents and exclusivity periods has led to the introduction of generic versions of naproxen/esomeprazole magnesium. This has a direct impact on VIMOVO's market share and pricing power.
Who are the Key Competitors for VIMOVO?
The competitive landscape for VIMOVO is characterized by both direct competitors offering similar fixed-dose combinations and indirect competitors providing individual components or alternative treatment modalities.
- Direct Generic Competitors: The most significant competition comes from generic manufacturers producing naproxen/esomeprazole magnesium. Companies like Teva Pharmaceuticals, Mylan (now Viatris), and Sandoz are active in this space. These generics offer a lower-cost alternative, eroding VIMOVO's market share.
- Other NSAID/PPI Combinations: While VIMOVO is a prominent example, other fixed-dose combinations of non-steroidal anti-inflammatory drugs (NSAIDs) and proton pump inhibitors (PPIs) exist or have existed. Examples include diclofenac/omeprazole, although market penetration and clinical acceptance may vary.
- Individual Component Prescriptions: A substantial portion of the market for VIMOVO's indications is served by physicians prescribing naproxen and esomeprazole (or other PPIs) as separate medications. This allows for more flexible dosing and individual titration, often at a lower combined cost than branded VIMOVO.
- Alternative Pain Management Therapies:
- Other NSAIDs: Traditional NSAIDs like ibuprofen, meloxicam, and celecoxib (a COX-2 inhibitor) are widely used.
- Acetaminophen: A common first-line analgesic for mild to moderate pain.
- Opioids: While generally reserved for severe pain due to addiction risks, they remain a treatment option.
- Disease-Modifying Drugs: For rheumatoid arthritis and ankylosing spondylitis, disease-modifying antirheumatic drugs (DMARDs) and biologic agents represent a different class of treatment with distinct mechanisms of action.
- Non-Pharmacological Interventions: Physical therapy, exercise, weight management, and assistive devices are crucial components of managing chronic pain conditions and reduce reliance on medication.
What are the Sales Performance and Financial Projections for VIMOVO?
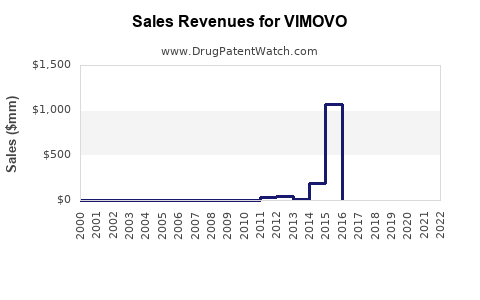
VIMOVO's sales trajectory has been significantly impacted by its patent expiry and the subsequent rise of generic competition.
- Peak Sales: VIMOVO, marketed by AstraZeneca and later by Horizon Pharma (now Amneal Pharmaceuticals), achieved peak annual sales in the mid-to-late 2010s. While precise figures for VIMOVO as a standalone product are often consolidated within broader portfolios, reports indicate sales in the hundreds of millions of dollars annually during its exclusivity period. For example, Horizon Pharma reported net sales of VIMOVO as $224.5 million in 2018 and $194.6 million in 2019, demonstrating a decline even before significant generic impact in some markets.
- Post-Patent Expiry Decline: Following patent expiries and the introduction of generics, VIMOVO's sales have experienced a substantial decline. Generic versions are typically priced at 40-70% less than the branded product, rapidly capturing market share.
- Market Share Erosion: The branded VIMOVO product now holds a significantly reduced market share compared to its peak. Physicians and payers often favor the cost-effectiveness of generic alternatives.
- Projected Future Performance: The financial outlook for branded VIMOVO is characterized by continued sales decline. Projections for the product depend on the remaining market share it can retain and any specific market access agreements or physician loyalty.
- Generic Market Dominance: The market for naproxen/esomeprazole magnesium is largely dominated by generics. The branded product's revenue will likely continue to represent a small fraction of the total market for this combination.
- Contract Manufacturer Sales: Companies that previously manufactured VIMOVO for the originator may now be involved in supplying active pharmaceutical ingredients (APIs) or finished dosage forms to generic manufacturers.
- Limited Pipeline Value: As an off-patent drug facing generic competition, VIMOVO itself holds limited value for R&D investment, unless new therapeutic indications or novel formulations (with new patent protection) are developed.
What are the Regulatory and Clinical Considerations for VIMOVO?
Understanding the regulatory history and clinical profile of VIMOVO is essential for assessing its market positioning and risks.
- Original Approvals: VIMOVO was initially approved by the U.S. FDA in 2010 for the symptomatic treatment of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis. It was approved by the European Medicines Agency (EMA) in 2009.
- Risk Profile of NSAIDs: Naproxen, as an NSAID, carries a risk of gastrointestinal (GI) complications (ulcers, bleeding), cardiovascular events, and renal effects.
- Benefit of PPI Co-administration: The inclusion of esomeprazole magnesium, a PPI, is designed to mitigate the GI risk associated with naproxen by reducing stomach acid. This combination addresses a key concern for physicians prescribing NSAIDs for chronic conditions.
- Black Box Warnings: Both naproxen and PPIs carry significant safety warnings. NSAIDs have a boxed warning regarding cardiovascular thrombotic events, myocardial infarction, and stroke, as well as GI bleeding, ulceration, and perforation. PPIs have warnings concerning potential increased risk of fractures, C. difficile-associated diarrhea, and vitamin B12 deficiency. These warnings influence prescribing patterns and necessitate careful patient selection and monitoring.
- Prescribing Guidelines: Clinical practice guidelines for osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis often recommend NSAIDs as a first-line or second-line treatment option, but with considerations for gastrointestinal and cardiovascular risk. The use of PPIs is frequently recommended for patients at higher risk of GI events.
- Pharmacoeconomic Evaluations: Payers and healthcare systems conduct pharmacoeconomic analyses to compare the cost-effectiveness of branded VIMOVO against generic alternatives and individual components. These evaluations heavily influence formulary placement and reimbursement.
- Post-Marketing Surveillance: Like all pharmaceuticals, VIMOVO is subject to ongoing post-marketing surveillance to monitor for adverse events and real-world effectiveness.
What is the Market Landscape for VIMOVO's Indications?
The market for VIMOVO's approved indications is large and diverse, presenting both opportunities and challenges.
- Osteoarthritis (OA): This is the most prevalent form of arthritis, affecting millions globally. The market is characterized by a high volume of patients and a wide range of treatment options, from over-the-counter analgesics to prescription drugs and surgical interventions.
- Prevalence: In the U.S., OA affects over 32.5 million adults (CDC).
- Treatment Goals: Pain relief, improved function, and slowing disease progression (though pharmaceutical interventions primarily focus on symptom management).
- Rheumatoid Arthritis (RA): An autoimmune disease causing chronic inflammation of joints. While NSAIDs can help manage symptoms, RA treatment typically involves DMARDs and biologics for disease modification.
- Prevalence: Affects approximately 1.3 million adults in the U.S. (CDC).
- Treatment Landscape: Dominated by disease-modifying therapies, with NSAIDs often used as adjunctive symptomatic treatment.
- Ankylosing Spondylitis (AS): A type of inflammatory arthritis that primarily affects the spine. NSAIDs are a cornerstone of symptomatic treatment.
- Prevalence: Estimated to affect between 0.1% and 0.5% of the adult population in Western countries.
- Treatment Landscape: NSAIDs are a first-line therapy for pain and stiffness. Biologics are used for more severe or refractory cases.
- Market Trends:
- Shift towards Biologics: For inflammatory conditions like RA and AS, there is a significant shift towards the use of targeted biologic therapies, which offer disease modification but are also more expensive.
- Cost Containment: In OA and general pain management, there is increasing pressure to control costs, leading to a preference for generics and non-pharmacological approaches.
- Personalized Medicine: Advances in understanding disease subtypes are driving towards more personalized treatment approaches, though this is less pronounced for broad-spectrum NSAID/PPI combinations.
- Aging Population: The increasing global aging population contributes to the growing prevalence of osteoarthritis, maintaining a large patient base for symptomatic treatments.
Key Takeaways
- VIMOVO’s patent exclusivity has largely expired in major markets, leading to significant generic competition.
- Branded VIMOVO sales have declined sharply post-patent expiry, with generic naproxen/esomeprazole magnesium now dominating the market.
- The competitive landscape includes numerous generic manufacturers, individual component prescriptions, and alternative pain management therapies.
- Regulatory considerations, including NSAID and PPI safety warnings, influence prescribing patterns and necessitate careful risk-benefit assessments.
- The market for VIMOVO's indications (OA, RA, AS) is large but increasingly driven by cost-effectiveness and the availability of advanced therapeutic options.
Frequently Asked Questions
-
When did VIMOVO lose its primary patent protection?
Primary patents for VIMOVO’s fixed-dose combination expired in the United States between 2017 and 2018, allowing for the introduction of generic versions.
-
What is the primary reason for the decline in VIMOVO's sales?
The decline in VIMOVO's sales is primarily attributable to the expiration of its patent exclusivity, which enabled the widespread entry of lower-cost generic versions of naproxen/esomeprazole magnesium.
-
Are there any ongoing clinical trials for VIMOVO?
As a now largely off-patent drug, there are limited to no significant ongoing clinical trials for branded VIMOVO to explore new indications or formulations. Research efforts are focused on newer drug classes or alternative treatments.
-
What are the main safety concerns associated with VIMOVO?
The main safety concerns are those inherent to its components: naproxen (a NSAID) carries risks of gastrointestinal bleeding, cardiovascular events, and renal impairment, while esomeprazole (a PPI) has warnings related to bone fractures and C. difficile infection.
-
How does the cost of generic naproxen/esomeprazole compare to branded VIMOVO?
Generic naproxen/esomeprazole magnesium is typically priced 40% to 70% lower than branded VIMOVO, making it the preferred option for many payers and patients.
Citations
[1] U.S. Food and Drug Administration. (n.d.). FDA Orange Book. Retrieved from [FDA Orange Book Website] (Specific access date and URL not applicable as it's a dynamic database).
[2] U.S. Patent and Trademark Office. (n.d.). USPTO Patent Search. Retrieved from [USPTO Website] (Specific patent numbers and grant dates would be required for precise citation, but the general search functionality is referenced).
[3] Centers for Disease Control and Prevention. (n.d.). Arthritis Data and Statistics. Retrieved from [CDC Website] (Specific data access dates and URLs vary by report).
[4] European Medicines Agency. (n.d.). European Public Assessment Reports. Retrieved from [EMA Website] (Specific EPAR details for VIMOVO would be required for precise citation).
[5] Horizon Pharma PLC. (2019). Annual Report on Form 10-K for the fiscal year ended December 31, 2019. U.S. Securities and Exchange Commission.