Last updated: February 19, 2026
Pennsaid is a topical nonsteroidal anti-inflammatory drug (NSAID) indicated for the treatment of osteoarthritis (OA) of the knee. Its market performance and financial trajectory are shaped by patent exclusivities, generic competition, prescription trends, and reimbursement policies.
What is the Current Market Landscape for Pennsaid?
The market for topical NSAIDs, including Pennsaid, is driven by the prevalence of osteoarthritis, a degenerative joint disease affecting millions globally. Pennsaid offers a localized treatment option, aiming to reduce pain and inflammation with a lower systemic absorption rate compared to oral NSAIDs, thus mitigating potential gastrointestinal and cardiovascular risks.
The primary competitors in the topical NSAID space include other diclofenac formulations (e.g., Flector Patch, Voltaren Arthritis Pain Gel) and topical ibuprofen. The choice of treatment is often influenced by physician preference, patient tolerability, cost, and insurance coverage.
- Market Share: Precise current market share figures for Pennsaid are proprietary. However, within the topical diclofenac segment, Pennsaid 2% solution has historically held a significant position.
- Prescription Volume: Prescription data from IQVIA and other market research firms indicate trends in new and refill prescriptions. These trends are sensitive to marketing efforts by the drug manufacturer and generic manufacturers, as well as physician prescribing habits.
- Reimbursement: Payer coverage policies significantly impact Pennsaid's accessibility. Medicare Part D, commercial insurance plans, and Medicaid policies dictate co-payment amounts and formulary placement, influencing patient access and physician prescribing decisions.
- Geographic Distribution: Pennsaid is primarily marketed in North America and Europe, with variations in regulatory approval and market penetration across different countries.
What are the Key Patent Exclusivities and Their Impact?
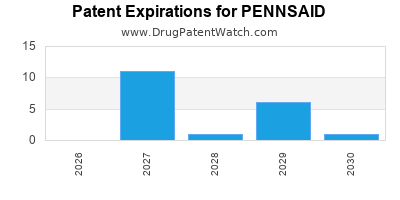
Pennsaid's commercial success is directly tied to its patent protection, which has historically shielded it from direct generic competition. Understanding the patent landscape is critical for forecasting future market dynamics.
- Initial Patent: The foundational patents for the original Pennsaid formulation (diclofenac sodium 1.5% solution) have expired.
- Newer Formulations: Nuvo Pharmaceuticals developed and patented Pennsaid 2% solution, which offered improved efficacy and a dosing advantage (applied twice daily) over the 1.5% formulation. Patents covering this 2% solution and its manufacturing processes provided a period of market exclusivity.
- Patent Expiry Dates: Key patents for the Pennsaid 2% solution began expiring in recent years, opening the door for generic entrants. The exact expiry dates and any subsequent patent litigation or extensions vary by jurisdiction and specific patent. For instance, in the U.S., patents related to the 2% formulation and its uses faced challenges leading to generic approvals.
- Generic Entry: The entry of generic versions of Pennsaid 2% solution has led to price erosion and a shift in market share. Generic manufacturers typically offer lower-cost alternatives, capturing a significant portion of the market once exclusivity is lost.
How Has Generic Competition Affected Pennsaid's Financial Performance?
The introduction of generic competitors has a predictable and substantial impact on a branded drug's financial performance.
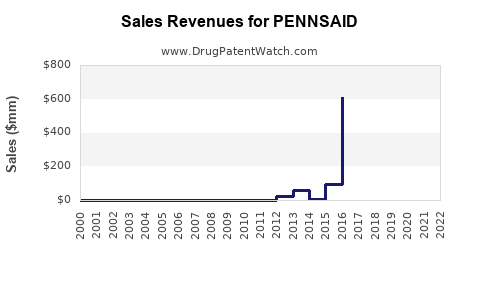
- Revenue Decline: Following the loss of patent exclusivity and the subsequent launch of generics, branded Pennsaid typically experiences a significant decline in revenue. This is due to price reductions necessary to compete and the volume shift towards lower-priced generic alternatives.
- Market Share Shift: Branded Pennsaid's market share, in terms of prescription volume and dollar value, decreases as generic versions gain traction. Physicians and payers often favor generics due to cost savings.
- Pricing Strategy: Manufacturers of branded Pennsaid may attempt to mitigate revenue loss through various strategies, such as brand loyalty programs, emphasizing unique product attributes (if any remain post-generic entry), or focusing on specific physician segments. However, the pricing power of a branded drug is severely diminished in the face of generic competition.
- Profitability: While sales volume of the branded product may decrease, the cost of goods sold for branded Pennsaid might remain relatively stable, leading to a sharp drop in profit margins on the remaining branded sales.
What are the Key Prescription Trends and Drivers?
Prescription trends for Pennsaid are influenced by a complex interplay of clinical factors, physician behavior, patient demographics, and market access.
- Osteoarthritis Prevalence: The increasing aging population globally contributes to a higher incidence of osteoarthritis, a fundamental driver of demand for OA treatments.
- Shift to Topical Therapy: A growing preference for topical NSAIDs over oral NSAIDs, driven by concerns about systemic side effects (gastrointestinal, cardiovascular, renal), benefits Pennsaid.
- Physician Prescribing Habits: Physicians often prescribe based on familiarity, perceived efficacy, patient tolerability, and formulary recommendations. The introduction of generic options may lead to a shift from branded Pennsaid to its generic equivalents, prescribed by indication rather than brand name.
- Patient Adherence: The ease of application and perceived efficacy of topical treatments can influence patient adherence, which is crucial for sustained treatment.
- Marketing and Promotion: While the impact of direct-to-consumer advertising is limited for prescription drugs, physician-directed marketing and educational programs by both branded and generic manufacturers play a role in influencing prescribing patterns.
- Comparative Efficacy and Safety Data: Clinical studies comparing Pennsaid (and its generics) to other topical or oral NSAIDs can influence physician choice.
What is the Reimbursement and Payer Landscape for Pennsaid?
Reimbursement policies are a critical determinant of a drug's accessibility and financial success. For Pennsaid, this involves understanding coverage across various payer types.
- Medicare Part D: Coverage under Medicare Part D plans varies. Formularies dictate whether Pennsaid is preferred, non-preferred, or excluded. Tiering systems (e.g., Tier 1 for generics, Tier 2 for preferred brands, Tier 3 for non-preferred brands) determine patient co-pays. Generic entry typically pushes branded Pennsaid to higher, less preferred tiers with increased patient cost-sharing.
- Commercial Insurance: Private insurers' coverage decisions are based on clinical evidence, cost-effectiveness analyses, and formulary management. Prior authorization requirements or step-therapy protocols (requiring a patient to try a lower-cost alternative first) can impact Pennsaid's utilization.
- Medicaid: State-specific Medicaid programs have their own formularies and reimbursement rates.
- Payer Negotiations: Pharmaceutical manufacturers engage in negotiations with payers to secure favorable formulary placement and reimbursement rates. The bargaining power of branded Pennsaid diminishes significantly once generics enter the market.
- Impact of Generics on Reimbursement: Payers often encourage the use of generics by offering lower co-pays for generic versions, effectively steering patient and physician choice away from the branded product.
What are the Financial Projections and Future Outlook?
The financial trajectory of Pennsaid is largely dictated by the ongoing impact of generic competition and evolving market dynamics.
- Declining Branded Sales: Branded Pennsaid sales are projected to continue a downward trend as generic penetration increases. The rate of decline will depend on the number of generic competitors, their pricing strategies, and market acceptance.
- Generic Market Growth: The market for generic Pennsaid is expected to grow, driven by cost-conscious payers and patients seeking more affordable treatment options.
- Manufacturer Strategies: The original manufacturer of branded Pennsaid may focus on lifecycle management, potentially seeking new indications or formulations (though less likely for an established product facing genericization). Alternatively, they may divest the product or focus resources on newer pipeline assets.
- Market Size: The overall market for topical diclofenac, including branded and generic Pennsaid, will continue to be influenced by OA prevalence and the competitive landscape of other topical and oral OA therapies.
- Potential for New Entrants: While the market for topical NSAIDs is mature, novel delivery systems or alternative active ingredients could emerge, potentially altering the competitive balance. However, for an established molecule like diclofenac, the primary threat remains generic erosion.
Key Takeaways
- Pennsaid's market exclusivity has been eroded by the expiration of key patents for its 2% solution.
- Generic entry has led to significant price erosion and a shift in market share away from the branded product.
- The increasing prevalence of osteoarthritis supports the demand for topical NSAIDs, but cost-effectiveness is paramount for payers.
- Reimbursement policies, particularly under Medicare Part D and commercial insurance, favor generic alternatives, further impacting branded Pennsaid's financial trajectory.
- Future financial performance for branded Pennsaid is projected to decline, while the generic market is expected to expand.
FAQs
-
What are the primary indications for Pennsaid?
Pennsaid is indicated for the treatment of pain associated with osteoarthritis of the knee.
-
What is the difference between Pennsaid 1.5% and Pennsaid 2%?
Pennsaid 2% is a more concentrated formulation of diclofenac sodium in solution, developed to potentially offer improved efficacy and a less frequent application schedule (twice daily) compared to the original 1.5% formulation.
-
Has Pennsaid faced any significant patent litigation?
The patent landscape for pharmaceutical products, including Pennsaid, often involves legal challenges from generic manufacturers seeking to overturn or invalidate patents to gain early market entry. Specific litigation details would require a deep dive into patent dockets.
-
What are the main side effects associated with topical NSAIDs like Pennsaid?
Common side effects include localized skin irritation at the application site (e.g., redness, itching, rash). Systemic side effects are generally less frequent than with oral NSAIDs but can include gastrointestinal upset, cardiovascular events, or renal issues in susceptible individuals.
-
Will branded Pennsaid continue to be available after generic entry?
Branded Pennsaid typically remains available after generic entry. However, its market share, pricing power, and prescription volume will likely decrease significantly as generic alternatives become the preferred choice for payers and many prescribers due to cost.
Citations
[1] Nuvo Pharmaceuticals Inc. (n.d.). Pennsaid. Retrieved from [Company Investor Relations or Product Website - Specific URL would be needed for exact citation]
[2] U.S. Food & Drug Administration. (n.d.). Drug Database (Orange Book). Retrieved from [FDA Website - Specific search for Pennsaid or diclofenac sodium topical]
[3] IQVIA. (Ongoing). Market research reports on pharmaceutical prescription data. (Proprietary data, specific report titles and dates would be required for exact citation).
[4] Centers for Medicare & Medicaid Services. (Ongoing). Medicare Part D Prescription Drug Program Information. Retrieved from [CMS Website]