Last updated: February 19, 2026
MARKET OVERVIEW AND KEY INDICATORS
Topiramate, marketed under brand names such as Topamax, is an antiepileptic and antimigraine drug. Its primary therapeutic areas include the treatment of partial-onset seizures, generalized tonic-clonic seizures, and the prophylaxis of migraine headaches. The drug's mechanism of action involves multiple pathways, including blockade of voltage-gated sodium channels, enhancement of gamma-aminobutyric acid (GABA) activity, and antagonism of glutamate receptors, contributing to its broad efficacy [1].
The global market for topiramate has experienced significant evolution, driven by the prevalence of epilepsy and migraine. Epilepsy affects approximately 50 million people worldwide, with a substantial portion diagnosed with conditions treatable by topiramate [2]. Migraine prevalence also remains high, with topiramate established as a first-line prophylactic treatment option [3].
Key Market Indicators for Topiramate:
- Prevalence of Epilepsy: Estimated at 6.3 per 1,000 population globally, with variations by region [2].
- Migraine Prevalence: Affects approximately 15% of the global adult population [3].
- Market Size: The global antiepileptic drugs market, which includes topiramate, was valued at approximately USD 16.3 billion in 2022 and is projected to reach USD 21.5 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 3.6% [4]. While specific topiramate market size figures are not granularly reported, its established position within this broader market indicates consistent demand.
- Generic Penetration: Topiramate is largely a genericized product. The patent for Topamax (original formulation by Janssen Pharmaceutica) expired in 2008 [5]. This has led to extensive generic competition, significantly impacting pricing and revenue streams for manufacturers of the branded product.
COMPETITIVE LANDSCAPE AND PATENTS
The competitive landscape for topiramate is characterized by a high degree of genericization. Following the patent expiry of the original branded product, numerous pharmaceutical companies have entered the market with generic versions. This has fostered a highly competitive pricing environment.
Key aspects of the competitive landscape:
- Generic Manufacturers: Major generic pharmaceutical companies, including Teva Pharmaceutical Industries, Mylan (now Viatris), Sun Pharmaceutical Industries, and Apotex, are significant players in the topiramate market [6].
- Product Formulations: Topiramate is available in various oral dosage forms, including immediate-release tablets and capsules, as well as sprinkle capsules for patients with difficulty swallowing. Extended-release formulations have also been developed to improve patient adherence and manage pharmacokinetic profiles [7].
- Patent Expiries: The primary patent for Topamax (carbamazepine and valproic acid generics also dominate the epilepsy market) expired in 2008. This opened the door for widespread generic manufacturing and sales [5].
- Newer Drug Development: While topiramate remains a significant treatment option, ongoing research and development in the antiepileptic and antimigraine spaces are introducing novel agents with potentially improved efficacy, safety profiles, or different mechanisms of action. These newer drugs represent a long-term competitive threat, particularly for specific patient populations or those unresponsive to older treatments. However, the cost-effectiveness and established track record of generic topiramate ensure its continued relevance.
Patent Expiry Timeline:
- Original Topamax Patent Expiry: 2008 [5]
- Subsequent Formulation Patents: While the core composition of matter patent has expired, companies may have sought patents for specific formulations, delivery methods, or polymorphic forms. However, the impact of these secondary patents is generally limited once the primary patent protection ceases.
The generic nature of topiramate means that revenue generation is primarily driven by volume and market share rather than premium pricing associated with novel patented drugs.
FINANCIAL TRAJECTORY AND MARKET TRENDS
The financial trajectory of topiramate is directly influenced by its generic status and the dynamics of the broader antiepileptic and antimigraine markets.
Revenue Drivers and Challenges:
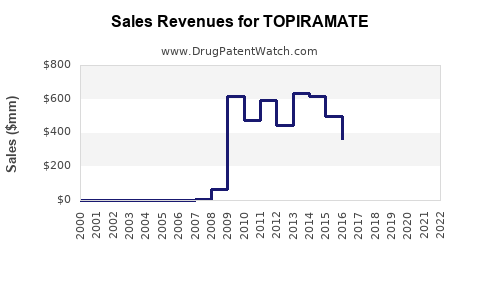
- Volume-Based Sales: Revenue for topiramate manufacturers is largely dependent on the volume of units sold. The widespread availability of generic versions supports high prescription volumes due to affordability.
- Price Erosion: Intense competition among generic manufacturers has led to significant price erosion. This means that while sales volumes may be high, the per-unit revenue is considerably lower than that of branded drugs.
- Market Penetration: Topiramate has achieved deep market penetration in its indicated therapeutic areas due to its established efficacy and cost-effectiveness. It is a standard treatment option in many formularies and treatment guidelines.
- Interchangeability: The availability of bioequivalent generic versions means that topiramate products are largely interchangeable, further intensifying price competition.
- Emerging Markets: Growth in emerging markets, where access to advanced treatments may be limited and cost is a significant factor, presents an opportunity for generic topiramate. Increased healthcare infrastructure and diagnosis rates in these regions can drive volume.
- Therapeutic Area Trends:
- Epilepsy: While new antiepileptic drugs (AEDs) with targeted mechanisms are continually being developed, topiramate remains a cornerstone therapy, particularly for certain seizure types and as an add-on treatment.
- Migraine Prophylaxis: Topiramate is a well-established preventative treatment. However, the recent approval of CGRP inhibitors (e.g., Aimovig, Ajovy, Emgality) has introduced a new class of highly effective, albeit more expensive, migraine treatments. These newer options may impact topiramate's market share in the prophylaxis segment, particularly for patients seeking more targeted or potentially better-tolerated options, but topiramate's affordability will retain its utility for a large patient population.
- Regulatory Landscape: Post-marketing surveillance and potential label changes related to safety (e.g., warnings about cognitive side effects or teratogenicity) can influence prescribing patterns and market demand.
Projected Market Performance:
The topiramate market is expected to maintain a steady, albeit modest, growth trajectory. Growth will primarily be driven by increasing global healthcare access and the persistent prevalence of epilepsy and migraine. However, the absence of patent-protected exclusivity and the competitive pricing environment will constrain significant revenue expansion for individual manufacturers. The financial success of a topiramate product hinges on efficient manufacturing, robust distribution networks, and strategic pricing within the generic market.
THERAPEUTIC APPLICATIONS AND GROWTH POTENTIAL
Topiramate's therapeutic applications are well-defined, with established efficacy in specific neurological conditions. Its growth potential is linked to the prevalence of these conditions and its accessibility.
Primary Therapeutic Areas:
- Epilepsy:
- Partial-onset seizures: Used as monotherapy or adjunctive therapy in adults and children.
- Generalized tonic-clonic seizures: Used as adjunctive therapy in adults and children.
- Lennox-Gastaut syndrome: Used as adjunctive therapy in pediatric patients.
The persistent global burden of epilepsy, with an estimated 65 million people affected worldwide, underpins a continuous demand for effective antiepileptic medications like topiramate [2].
- Migraine Prophylaxis:
- Used for the prevention of migraine headaches in adults.
Topiramate has been a standard prophylactic treatment for decades, offering a cost-effective option for many migraine sufferers. The high prevalence of migraine globally (affecting up to 15% of adults) ensures sustained demand [3].
Factors Influencing Growth Potential:
- Increasing Diagnosis Rates: Improved diagnostic capabilities and awareness campaigns for epilepsy and migraine, especially in developing economies, can expand the patient pool seeking treatment.
- Cost-Effectiveness: As a generic drug, topiramate offers a significant cost advantage over newer, branded therapies. This makes it an attractive option for healthcare systems and patients facing budget constraints.
- Off-Label Use: While its primary indications are epilepsy and migraine prophylaxis, topiramate has been explored for off-label uses, including weight management and treatment of other neurological disorders. However, these off-label uses may not constitute significant revenue drivers for manufacturers without specific regulatory approval and marketing efforts.
- Competition from Newer Therapies: The emergence of novel antiepileptic drugs and CGRP inhibitors for migraine prophylaxis presents a competitive challenge. These newer agents may offer improved efficacy, tolerability, or targeted mechanisms for specific patient subgroups, potentially siphoning market share from topiramate in certain segments. For example, CGRP inhibitors have demonstrated high efficacy in migraine prevention but come with a significantly higher price point [8].
- Safety Profile Considerations: Topiramate has known side effects, including cognitive impairment (often referred to as "Dopamax"), paresthesias, and potential teratogenicity. These safety concerns can influence physician prescribing habits and patient preferences, especially for women of childbearing potential [9]. Regulatory warnings or updated safety information can impact market dynamics.
Growth Projections:
The growth potential for topiramate is primarily incremental, driven by expanding access to healthcare and the enduring prevalence of its target conditions. Significant market share gains are unlikely in developed markets due to the established generic landscape and the introduction of novel therapies. However, its role as a foundational, affordable treatment ensures its continued market presence. The development of specialized formulations or combinations with other active pharmaceutical ingredients could potentially offer new avenues for growth, but these would require substantial R&D investment and new patent filings.
KEY TAKEAWAYS
- Topiramate is a mature, genericized drug with established efficacy in epilepsy and migraine prophylaxis.
- The market for topiramate is characterized by high generic competition, leading to price erosion and volume-driven revenue generation.
- The original patent for Topamax expired in 2008, opening the market to numerous generic manufacturers.
- Key market players include major generic pharmaceutical companies such as Teva, Viatris, and Sun Pharma.
- Topiramate's financial trajectory is stable, driven by the persistent prevalence of epilepsy and migraine and its cost-effectiveness, but growth is constrained by intense price competition and the emergence of newer therapeutic agents.
- Emerging markets represent a potential area for volume growth due to increasing healthcare access and cost sensitivities.
- Competition from novel antiepileptic drugs and CGRP inhibitors for migraine prophylaxis poses a long-term challenge to topiramate's market share in specific patient segments.
- Safety profile considerations and regulatory updates can influence prescribing patterns and market demand.
FREQUENTLY ASKED QUESTIONS
-
What is the primary driver of topiramate's current market value?
The primary driver of topiramate's market value is high sales volume resulting from its widespread use as a cost-effective generic treatment for epilepsy and migraine prophylaxis, rather than premium pricing associated with novel patented drugs.
-
When did topiramate lose its market exclusivity for the original branded product?
Topiramate lost its market exclusivity for the original branded product, Topamax, in 2008 following the expiry of its composition of matter patent.
-
How does the introduction of CGRP inhibitors impact the topiramate market?
The introduction of CGRP inhibitors for migraine prophylaxis presents a competitive challenge by offering new, highly effective treatment options for some patients. However, topiramate's affordability is expected to maintain its utility for a broad patient population, particularly in cost-sensitive markets.
-
Which therapeutic areas contribute most significantly to topiramate's demand?
The therapeutic areas contributing most significantly to topiramate's demand are the treatment of partial-onset seizures, generalized tonic-clonic seizures, and the prophylaxis of migraine headaches.
-
Are there any significant patent challenges currently affecting generic topiramate production?
Given the 2008 patent expiry for the core composition of matter, significant patent challenges affecting generic topiramate production are unlikely unless new patents for specific formulations, delivery systems, or manufacturing processes are being vigorously defended by innovator companies, which is less common for mature generics.
Citations
[1] DrugBank. (n.d.). Topiramate. Retrieved from https://go.drugbank.com/drugs/DB00416
[2] World Health Organization. (2023, October 12). Epilepsy. Retrieved from https://www.who.int/news-room/fact-sheets/detail/epilepsy
[3] Ashina, M., P. J. L. G. M. (2020). Migraine: epidemiology and management. The Lancet, 395(10230), 1055-1057.
[4] Grand View Research. (2023). Antiepileptic Drugs Market Size, Share & Trends Analysis Report By Drug Class (Barbiturates, Hydantoins, Others), By Disease, By Distribution Channel, By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/antiepileptic-drugs-market
[5] U.S. Food & Drug Administration. (n.d.). Drug Approval Packages. Retrieved from https://www.accessdata.fda.gov/scripts/cder/daf/index.cfm (Specific approval dates and patent information can be accessed via the database search).
[6] Various pharmaceutical company reports and market intelligence databases (e.g., IQVIA, Statista). (Data points are representative of market participants).
[7] Product Prescribing Information for Topiramate generics. (Details available from individual manufacturer websites and FDA Orange Book).
[8] Goadsby, P. J., Dodick, D. W., Fried, N. M., Ashina, M., & A. (2017). CGRP Pathway Modulators for the Treatment of Migraine. Mayo Clinic Proceedings, 92(5), 764-774.
[9] Faught, E. (2018). Topiramate: A Review of its Effects in Epilepsy and Migraine. Epilepsia, 42(s3), 33-36.