Last updated: February 19, 2026
Tacrolimus, a calcineurin inhibitor, demonstrates a complex market dynamic shaped by expiring patents, generic competition, and ongoing therapeutic applications. Its primary use in preventing organ rejection post-transplant and managing autoimmune conditions has established a significant global market. However, the erosion of exclusivity for originator products is leading to increased market fragmentation and price pressures.
What is the current patent status of key tacrolimus formulations?
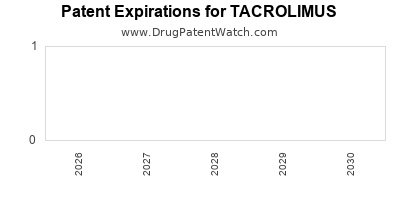
The patent landscape for tacrolimus is characterized by the expiration of core composition of matter patents for the originator molecule, largely impacting branded product exclusivity. While the original patents have expired in major markets, secondary patents related to specific formulations, delivery methods, and manufacturing processes continue to offer some level of protection.
- Original Composition of Matter Patents: The foundational patents covering the tacrolimus molecule itself have largely expired. For example, the initial patent protection for Astellas Pharma's Prograf in the United States expired in the early 2000s. Similar expirations have occurred in other key territories, including Europe and Japan, paving the way for generic entry.
- Formulation Patents: Pharmaceutical companies have pursued and obtained patents on improved formulations of tacrolimus. These include extended-release formulations designed to reduce dosing frequency and improve patient compliance, as well as topical formulations for dermatological applications. Examples include patents for once-daily oral formulations and specialized creams for eczema.
- Extended-Release Formulations: Patents in this category focus on drug delivery systems that control the rate at which tacrolimus is released into the bloodstream. These often involve specific polymer matrices or encapsulation technologies. For instance, patents describe granular compositions or microparticle formulations designed for sustained absorption.
- Topical Formulations: Patents for dermatological uses of tacrolimus, such as Protopic (Fujisawa Healthcare, now Astellas), address creams and ointments. These patents often cover specific excipient profiles, concentrations, and manufacturing methods to ensure stability and efficacy for skin application.
- Manufacturing Process Patents: Patents related to novel or more efficient manufacturing processes for tacrolimus can provide a competitive advantage. These may involve specific chemical synthesis routes, purification techniques, or polymorphic forms of the drug.
- Method of Use Patents: While less common for well-established drugs, method of use patents can cover novel therapeutic applications or specific dosing regimens. For tacrolimus, these might pertain to its efficacy in specific sub-populations of transplant recipients or its synergistic effects with other immunosuppressants in particular contexts.
The expiration of these patents has led to a significant influx of generic competitors, intensifying price competition and affecting the revenue streams of originator products. The remaining patent protection is predominantly focused on advanced delivery systems and novel applications, which are critical for differentiation and maintaining market share in the post-exclusivity era.
How has the market for tacrolimus performed financially?
The global market for tacrolimus has experienced substantial growth, driven by its critical role in immunosuppression and the increasing prevalence of organ transplantation and autoimmune diseases. However, recent performance is increasingly shaped by generic competition, leading to shifts in revenue distribution.
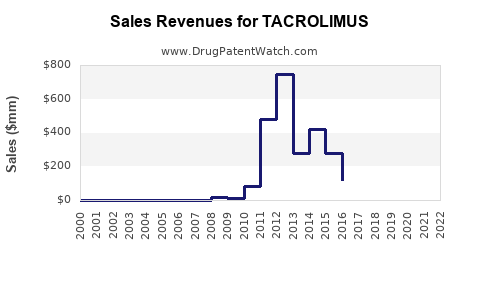
- Global Market Size: The global tacrolimus market was valued at approximately $2.1 billion in 2022 and is projected to reach $2.7 billion by 2030, exhibiting a compound annual growth rate (CAGR) of 3.0% from 2023 to 2030. (Source: Grand View Research)
- Originator Product Performance: Astellas Pharma's Prograf, the original branded formulation, historically dominated the market. Its peak annual sales were significant, contributing billions to the company's revenue. However, with patent expirations, Prograf's sales have declined considerably due to generic erosion. For example, in fiscal year 2023, Astellas reported lower sales for its immunosuppressant portfolio, largely attributed to generic competition for tacrolimus.
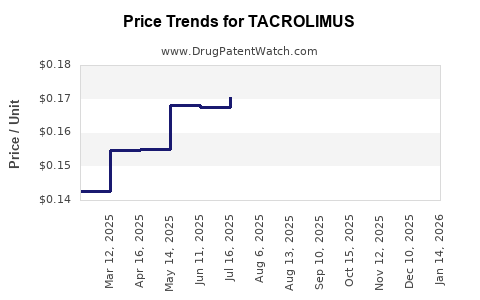
- Generic Market Growth: The generic tacrolimus segment has experienced rapid expansion following patent expiries. Generic manufacturers have introduced cost-effective alternatives, capturing a substantial share of the market. This has led to a significant decrease in the average selling price (ASP) of tacrolimus products globally.
- Regional Market Variations:
- North America: This region represents a significant market for tacrolimus, driven by advanced healthcare infrastructure and a high volume of organ transplants. Generic penetration is high, leading to price competition.
- Europe: Similar to North America, Europe has a mature tacrolimus market with strong generic presence. Regulatory approvals and reimbursement policies influence market dynamics.
- Asia-Pacific: This region is expected to witness the fastest growth, fueled by an increasing number of transplant procedures, rising disposable incomes, and expanding healthcare access. Generic manufacturers are prominent in this market.
- Key Drivers:
- Increasing incidence of organ transplantation procedures.
- Rising prevalence of autoimmune diseases like psoriasis and rheumatoid arthritis.
- Growing demand for generic immunosuppressants due to cost-effectiveness.
- Advancements in drug delivery systems offering improved patient compliance.
- Restraints:
- Intense price competition from generic products.
- Stringent regulatory requirements for drug approval and manufacturing.
- Potential for adverse effects associated with tacrolimus therapy.
The financial trajectory for tacrolimus is characterized by a mature, yet evolving, market. While the originator brands have seen their market dominance wane, the overall therapeutic demand ensures continued market value, now primarily driven by generic formulations and specialized, patented variants.
What are the primary therapeutic applications driving tacrolimus demand?
Tacrolimus's efficacy in modulating the immune system underpins its critical role in several therapeutic areas, with organ transplantation being its most prominent application.
- Organ Transplantation: Tacrolimus is a cornerstone of immunosuppressive therapy for recipients of solid organ transplants, including kidney, liver, heart, and lung transplants. Its primary function is to prevent the immune system from recognizing and rejecting the transplanted organ. By inhibiting T-cell activation, it significantly reduces the risk of acute and chronic rejection episodes. This application accounts for the largest segment of the tacrolimus market.
- Kidney Transplants: Tacrolimus is widely used as a maintenance immunosuppressant in kidney transplant recipients.
- Liver Transplants: It is a standard of care for preventing rejection in liver transplant patients.
- Heart and Lung Transplants: Tacrolimus is also crucial for managing immunosuppression in heart and lung transplant recipients, though often in combination with other agents.
- Autoimmune Diseases: Tacrolimus is also employed in the management of certain autoimmune conditions where aberrant immune responses target the body's own tissues.
- Atopic Dermatitis (Eczema): Topical formulations of tacrolimus (e.g., Protopic) are effective in treating moderate to severe atopic dermatitis in patients who have not responded to or cannot tolerate conventional therapies like corticosteroids. It works by suppressing skin inflammation.
- Inflammatory Bowel Disease (IBD): In some cases, oral tacrolimus is used off-label or in specific clinical scenarios to manage severe forms of ulcerative colitis and Crohn's disease, particularly when other treatments have failed.
- Ocular Inflammatory Conditions: Topical tacrolimus has been investigated and used for certain forms of non-infectious uveitis and dry eye disease.
- Graft-versus-Host Disease (GvHD) Prophylaxis: Following hematopoietic stem cell transplantation (HSCT), tacrolimus is used to prevent GvHD, a potentially life-threatening complication where the donor immune cells attack the recipient's tissues.
- Other Off-Label Uses: Tacrolimus is explored for various other inflammatory and autoimmune conditions, including psoriasis, Sjögren's syndrome, and certain types of nephrotic syndrome, although these applications are often less established and may be used in specific patient populations or research settings.
The breadth of these applications ensures a consistent and substantial demand for tacrolimus. The ongoing need for effective immunosuppression in transplantation and the management of chronic inflammatory conditions are primary drivers of its market presence.
Who are the key players in the tacrolimus market?
The tacrolimus market features a mix of originator pharmaceutical companies, generic manufacturers, and, to a lesser extent, companies developing specialized delivery systems. The competitive landscape is heavily influenced by patent expiry and the subsequent genericization of the market.
- Originator Companies:
- Astellas Pharma Inc.: As the developer of Prograf, the original branded tacrolimus, Astellas remains a significant player. While facing generic competition, they continue to market their branded product and may have interests in next-generation formulations or related products.
- Major Generic Manufacturers: The market is now dominated by numerous generic pharmaceutical companies that have entered after patent expiries. These companies focus on producing bioequivalent versions of tacrolimus capsules and injections. Key players include:
- Teva Pharmaceutical Industries Ltd.
- Dr. Reddy's Laboratories Ltd.
- Sun Pharmaceutical Industries Ltd.
- Mylan N.V. (now Viatris Inc.)
- Cipla Ltd.
- Lupin Ltd.
- Zydus Lifesciences (formerly Cadila Healthcare Ltd.)
- Specialty and Topical Formulations: Companies focusing on dermatological applications or novel delivery systems also hold a stake.
- Fujisawa Healthcare (now part of Astellas Pharma): Developed the topical formulation Protopic.
- Other companies: Various smaller and regional pharmaceutical companies manufacture and market generic topical tacrolimus products.
- Contract Manufacturing Organizations (CMOs): Many generic companies may leverage CMOs for the manufacturing of tacrolimus active pharmaceutical ingredient (API) or finished dosage forms, indicating a complex supply chain.
The competitive intensity is high, with price being a primary differentiating factor for generic products. Companies that can achieve efficient manufacturing and secure broad distribution networks are positioned to capture significant market share. Innovation in formulations, such as extended-release or combination therapies, remains an area where originator companies and some specialized firms can differentiate themselves.
What are the future trends and challenges for tacrolimus?
The future trajectory of the tacrolimus market will be shaped by several key trends, including advancements in drug delivery, evolving treatment paradigms, and persistent challenges related to market access and patient management.
- Trends:
- Advanced Drug Delivery Systems: Continued development of novel formulations aimed at improving patient compliance and reducing variability in drug absorption. This includes once-daily extended-release capsules and potentially even longer-acting injectable formulations. Research into microencapsulation and nanoparticle delivery is ongoing.
- Combination Therapies: Exploration and optimization of tacrolimus in combination with other immunosuppressants or novel agents to achieve enhanced efficacy, reduced toxicity, or overcome resistance mechanisms. This is particularly relevant in the context of personalized medicine.
- Targeted Therapeutic Approaches: A growing understanding of the genetic and molecular factors influencing immunosuppression may lead to more targeted use of tacrolimus in specific patient profiles, optimizing outcomes and minimizing side effects.
- Geographic Market Expansion: Increased demand for tacrolimus in emerging economies due to rising organ transplant rates, improving healthcare infrastructure, and greater access to generic medications.
- Focus on Topical Formulations: Continued research and market growth for topical tacrolimus in dermatological indications, with potential for new indications or improved formulations for managing chronic inflammatory skin conditions.
- Challenges:
- Intensifying Generic Competition and Price Erosion: The highly genericized nature of the market will continue to exert downward pressure on prices, impacting profitability for all market participants.
- Therapeutic Window Management: Tacrolimus has a narrow therapeutic window, meaning the difference between effective and toxic doses is small. This necessitates careful monitoring of drug levels and management of potential side effects, such as nephrotoxicity and neurotoxicity.
- Drug-Drug Interactions: Tacrolimus is subject to numerous drug-drug interactions, particularly with drugs that inhibit or induce its metabolism via cytochrome P450 enzymes. This complicates treatment regimens, especially in transplant patients on multiple medications.
- Regulatory Hurdles for New Formulations: Bringing novel tacrolimus formulations to market requires significant investment in clinical trials and navigating complex regulatory approval processes.
- Biosimilarity and Interchangeability Concerns: While not a biologic, the stringent requirements for demonstrating bioequivalence and interchangeability for generics, particularly for complex formulations, can be a challenge.
- Emergence of Alternative Immunosuppressants: The development of new classes of immunosuppressive drugs, such as mTOR inhibitors and JAK inhibitors, presents a competitive challenge, offering alternative mechanisms of action and potentially different side effect profiles.
The future market for tacrolimus will likely see a bifurcation: a highly competitive generic market for standard formulations, and a more specialized segment for innovative delivery systems and specific therapeutic niches. Companies will need to focus on operational efficiency, supply chain management, and strategic R&D to navigate these dynamics.
Key Takeaways
- The core patents for the tacrolimus molecule have expired, leading to widespread generic competition and significant price erosion for originator products.
- The global tacrolimus market was valued at approximately $2.1 billion in 2022 and is projected to grow at a CAGR of 3.0% through 2030, driven by transplant procedures and autoimmune disease management.
- Astellas Pharma, as the originator of Prograf, remains a key player, but the market is now largely dominated by generic manufacturers like Teva, Dr. Reddy's, and Sun Pharma.
- Key therapeutic applications include preventing organ transplant rejection, treating atopic dermatitis, and managing certain autoimmune conditions.
- Future trends include the development of advanced drug delivery systems, combination therapies, and expansion in emerging markets, while challenges include intensifying generic competition, therapeutic window management, and drug-drug interactions.
Frequently Asked Questions
- What is the primary mechanism of action for tacrolimus?
Tacrolimus is a calcineurin inhibitor that suppresses T-cell activation by blocking the production of interleukin-2 and other cytokines. This action inhibits the immune response that would otherwise lead to organ rejection or autoimmune damage.
- Are there any significant side effects associated with tacrolimus therapy?
Yes, common side effects include nephrotoxicity, neurotoxicity, tremor, hypertension, hyperglycemia, and increased susceptibility to infections. Close patient monitoring is essential.
- What is the difference between oral and topical tacrolimus?
Oral tacrolimus is used systemically for immunosuppression in transplant recipients and certain autoimmune diseases. Topical tacrolimus, typically in cream form, is applied to the skin to treat inflammatory skin conditions like atopic dermatitis.
- How does generic tacrolimus differ from branded tacrolimus?
Generic tacrolimus products are required to be bioequivalent to the branded product, meaning they deliver the same amount of active ingredient into the bloodstream over the same period. They contain the same active pharmaceutical ingredient (tacrolimus) and are therapeutically interchangeable. The primary difference is typically in price and inactive ingredients.
- What is the outlook for tacrolimus in the treatment of autoimmune diseases beyond atopic dermatitis?
While its use in atopic dermatitis is well-established, its role in other autoimmune diseases, such as inflammatory bowel disease or certain forms of nephrotic syndrome, is often considered for refractory cases or specific patient populations. Ongoing research may expand its therapeutic utility in these areas, though it faces competition from newer therapeutic classes.
Citations
[1] Grand View Research. (2023). Tacrolimus Market Size, Share & Trends Analysis Report By Type (Oral, Injectable, Topical), By Application (Organ Transplant, Autoimmune Diseases, Others), By Region, And Segment Forecasts, 2023 - 2030. Retrieved from https://www.grandviewresearch.com/industry-analysis/tacrolimus-market