Last updated: February 19, 2026
Executive Summary
Sumatriptan, a selective serotonin receptor agonist, remains a cornerstone in the acute treatment of migraine. The market is characterized by a mature landscape dominated by generic competition following patent expirations of originator products. Key market drivers include the prevalence of migraine disorders, increasing patient awareness, and the cost-effectiveness of generic sumatriptan formulations. Challenges stem from the availability of alternative migraine therapies, including newer classes of drugs and preventative treatments, as well as price erosion due to intense generic competition. The financial trajectory of sumatriptan products is largely dictated by volume sales of generics, with declining average selling prices. Future market growth will be modest, driven by demand in emerging economies and niche formulations.
What is the Global Market Size and Growth Outlook for Sumatriptan?
The global market for sumatriptan is substantial, primarily driven by the widespread use of generic versions. While precise, up-to-the-minute market size figures fluctuate due to reporting variances and the decentralized nature of generic markets, industry analyses from the past three to five years consistently place the market in the hundreds of millions to low billions of U.S. dollars annually.
For instance, reports from market research firms have estimated the sumatriptan market to be in the range of USD 600 million to USD 1.2 billion in recent years. These figures encompass all sumatriptan formulations, including oral tablets, nasal sprays, and injectable solutions.
The growth outlook for the sumatriptan market is projected to be modest. Compound Annual Growth Rates (CAGRs) are typically estimated to be between 2% and 4% for the next five to seven years. This growth is influenced by:
- Increasing Migraine Prevalence: Global estimates suggest that migraine affects approximately 15% of the adult population, leading to sustained demand for acute treatments.
- Cost-Effectiveness: Generic sumatriptan offers a significantly lower cost compared to branded migraine medications and newer therapeutic classes, making it a preferred choice for many patients and healthcare systems, particularly in price-sensitive markets.
- Emerging Markets: Growing access to healthcare and increased diagnosis rates in developing economies are contributing to market expansion.
However, growth is tempered by:
- Competition from Novel Therapies: The introduction of CGRP (calcitonin gene-related peptide) antagonists and other advanced migraine treatments, while often more expensive, offers differentiated efficacy and patient profiles, potentially drawing market share from triptans, especially for chronic or refractory migraine.
- Generic Price Erosion: The highly competitive generic landscape leads to continuous price declines, limiting overall revenue growth despite stable or increasing unit volumes.
What is the Patent Landscape and Generic Entry Timeline for Sumatriptan?
Sumatriptan was first patented by GlaxoWellcome (now GlaxoSmithKline) with the original patent for sumatriptan succinate expiring in the United States in 2008. The expiry of key composition of matter patents opened the door for widespread generic competition.
- Original Patent Expiry (US): 2008 (for sumatriptan succinate)
- Exclusivity Periods: Following initial patent expiry, various formulations and delivery methods may have had their own secondary patents or exclusivity periods. However, these have largely expired, allowing for comprehensive generic market entry.
- Generic Approvals: Regulatory bodies like the U.S. Food and Drug Administration (FDA) and the European Medicines Agency (EMA) have approved numerous generic sumatriptan products from various manufacturers. This has resulted in a highly fragmented generic market.
- Formulation Patents: While the core molecule is off-patent, there can still be patent activity around specific formulations, such as extended-release versions or novel delivery systems. However, these represent a smaller segment of the overall market compared to standard oral formulations.
The extensive genericization of sumatriptan means that the primary period of market exclusivity for the originator is long past. The market is now a competitive one driven by manufacturing efficiency, distribution networks, and pricing strategies among generic manufacturers.
How Do Different Sumatriptan Formulations Impact Market Position?
Sumatriptan is available in multiple dosage forms, each catering to different patient needs and preferences, influencing its market penetration and sales volume.
- Oral Tablets: This is the most common and widely prescribed formulation due to ease of use and patient acceptance. It represents the largest segment of the sumatriptan market by volume. Generic oral sumatriptan tablets are the benchmark for cost-effectiveness in acute migraine management.
- Nasal Sprays: Sumatriptan nasal sprays offer faster onset of action compared to oral tablets, making them suitable for patients experiencing nausea or vomiting with their migraines. This formulation commands a higher price point than oral tablets, reflecting its specialized delivery mechanism and faster relief. The market share is smaller but significant, particularly for patients who do not respond well to or cannot tolerate oral forms.
- Injectable Solutions: Subcutaneous injections of sumatriptan provide the most rapid relief among available sumatriptan formulations, typically within minutes. This is often reserved for severe migraine attacks or for patients who require very fast symptom resolution. Injectables are the least common formulation due to the invasive nature and higher cost, but they hold a critical niche for severe migraine sufferers.
- Other Formulations: Less common formulations may include suppositories or transdermal patches, though these have a more limited market presence compared to the primary three.
The market position of each formulation is dictated by its therapeutic advantage, cost, and convenience. Oral tablets dominate volume due to accessibility and price. Nasal sprays and injectables capture value by offering faster relief for specific patient needs, albeit at higher price points. The overall market for sumatriptan is a composite of these formulations, with oral generics forming the largest revenue and volume base.
What are the Key Drivers and Restraints for Sumatriptan Market Growth?
Key Market Drivers:
- High Global Prevalence of Migraine: Migraine disorders affect a significant percentage of the global population, creating a persistent demand for effective acute treatments. The World Health Organization (WHO) ranks migraine among the top 10 disabling diseases globally [1].
- Cost-Effectiveness of Generic Sumatriptan: As one of the earliest triptans to face generic competition, sumatriptan offers a well-established, reliable, and affordable treatment option. This is particularly important in healthcare systems with budget constraints and for uninsured or underinsured patients.
- Established Efficacy and Safety Profile: Sumatriptan has decades of clinical use, establishing a known efficacy and safety profile for acute migraine treatment. This familiarity provides a level of trust for both prescribers and patients.
- Accessibility of Over-the-Counter (OTC) Options: In some regions, lower-dose sumatriptan products are available OTC, further increasing accessibility and driving unit sales.
- Awareness and Diagnosis of Migraine: Increased public awareness campaigns and improved diagnostic capabilities lead to more individuals seeking treatment for their migraine episodes.
Key Market Restraints:
- Competition from Newer Migraine Therapies: The advent of CGRP antagonists (both injectable and oral forms) and ditans (e.g., lasmiditan) offers alternative mechanisms of action and may provide better efficacy or fewer side effects for some patient populations, particularly those with cardiovascular contraindications or who are refractory to triptans.
- Emergence of Preventative Therapies: Advances in migraine prevention, including CGRP monoclonal antibodies and oral preventative medications, can reduce the frequency of migraine attacks, thereby decreasing the demand for acute treatments like sumatriptan.
- Potential Side Effects and Contraindications: Sumatriptan is contraindicated in patients with uncontrolled hypertension, ischemic heart disease, or a history of stroke or transient ischemic attack (TIA). Side effects, such as chest tightness, flushing, and dizziness, can also limit its use.
- Price Erosion in the Generic Market: Intense competition among generic manufacturers drives down prices, capping revenue growth even as sales volumes may increase.
- Patient Preference for Novelty or Specific Symptom Relief: Some patients may actively seek newer treatments due to dissatisfaction with triptan efficacy or tolerability, or a preference for treatments addressing specific migraine symptoms beyond pain.
What is the Competitive Landscape for Sumatriptan Manufacturers?
The competitive landscape for sumatriptan is predominantly a generic market. The originator, GlaxoSmithKline (GSK), with its brand-name product Imitrex, has seen its market share diminish significantly following patent expiry. The market is now populated by a large number of generic pharmaceutical companies.
Key characteristics of the competitive landscape include:
- Fragmented Generic Market: Numerous companies manufacture and market generic sumatriptan products globally. This includes large multinational generic players and smaller regional manufacturers.
- Price-Based Competition: With multiple suppliers offering bioequivalent products, competition is primarily driven by price. Manufacturers with lower production costs and efficient supply chains gain a competitive advantage.
- Product Differentiation through Formulation and Delivery: While the active pharmaceutical ingredient (API) is identical, companies can differentiate through various formulations (tablets, nasal sprays, injections) and packaging. Offering a complete range of sumatriptan products can strengthen a manufacturer's position.
- Supply Chain and Distribution Prowess: Access to reliable API sources and robust distribution networks are critical for capturing market share, especially in large geographic regions.
- Regulatory Compliance and Quality Standards: Maintaining high manufacturing standards and navigating regulatory requirements in different markets are essential for market access and sustained sales.
- Strategic Partnerships: Some companies may engage in licensing agreements or partnerships for distribution or co-promotion, particularly in complex international markets.
Examples of companies involved in the sumatriptan market (as manufacturers or distributors of generic products) include, but are not limited to:
- Teva Pharmaceutical Industries
- Sandoz (a division of Novartis)
- Mylan N.V. (now part of Viatris)
- Aurobindo Pharma
- Dr. Reddy's Laboratories
- Sun Pharmaceutical Industries
- Perrigo Company
The absence of significant patent protection for the core sumatriptan molecule means that differentiation and success are achieved through operational excellence, cost management, and strategic market access rather than through novel product innovation of the API itself.
What is the Financial Trajectory and Profitability of Sumatriptan Products?
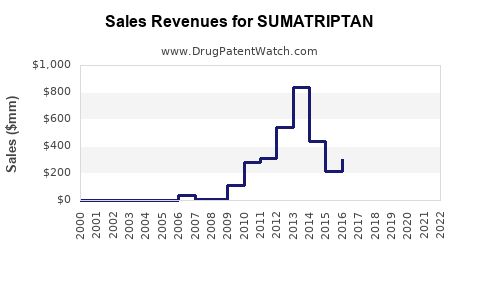
The financial trajectory of sumatriptan products is characterized by declining average selling prices (ASPs) and stable to growing volumes, driven by generic sales. Profitability for manufacturers is largely determined by economies of scale, efficient manufacturing processes, and control over supply chain costs.
- Revenue: The total revenue generated by sumatriptan products globally has plateaued or is experiencing modest growth. This is a consequence of price erosion caused by intense generic competition, which offsets gains from increased unit sales. For originator companies, revenue from branded sumatriptan has declined dramatically since patent expiry. For generic manufacturers, revenue is substantial in aggregate but individual company contributions can vary widely.
- Profit Margins: Profit margins on generic sumatriptan are typically lower than those for branded or patented drugs. However, for established generic manufacturers with optimized production and distribution, sumatriptan can still be a profitable product line due to high-volume sales and established manufacturing expertise. The gross margins are dependent on API sourcing costs, manufacturing efficiencies, and competitive pricing in specific markets.
- Cost Structure: The primary costs for generic sumatriptan manufacturers include API procurement, manufacturing, quality control, packaging, marketing (primarily to payers and healthcare providers in the generic space), and distribution. The ability to secure cost-effective API and optimize manufacturing yields is crucial for maintaining profitability.
- Investment: New investment in sumatriptan API production is generally limited, as the market is mature and competitive. Investment is more likely to be focused on optimizing existing facilities or developing more convenient or differentiated formulations (e.g., improved nasal sprays) if patentability for these specific aspects can be achieved. Research and development expenditure is significantly lower compared to novel drug development.
- Market Segmentation Impact: While oral tablets contribute the most volume, nasal sprays and injectables, being higher-priced formulations, can contribute disproportionately to revenue and potentially higher profit margins per unit, provided they can secure market access and demonstrate value.
In essence, the financial story of sumatriptan is one of volume-driven revenue in a highly competitive, price-sensitive generic market. Profitability relies on operational efficiency and scale rather than proprietary innovation.
Key Takeaways
- The global sumatriptan market is mature and dominated by generic competition, with annual market size in the hundreds of millions to low billions of U.S. dollars.
- Projected market growth is modest, between 2% and 4% CAGR, driven by migraine prevalence and cost-effectiveness but restrained by newer therapies.
- Original sumatriptan patents expired around 2008, leading to widespread generic entry and intense price competition.
- Oral tablets constitute the largest market segment by volume, while nasal sprays and injectables cater to specific patient needs at higher price points.
- Key market drivers include high migraine prevalence and sumatriptan's affordability, while restraints involve competition from novel CGRP inhibitors and preventative treatments.
- The competitive landscape is fragmented with numerous generic manufacturers competing primarily on price.
- The financial trajectory involves declining ASPs and stable to growing volumes, with profitability dependent on manufacturing efficiency and scale.
Frequently Asked Questions
1. What is the primary mechanism of action for sumatriptan?
Sumatriptan is a selective serotonin 5-HT1B/1D receptor agonist that causes vasoconstriction of cranial blood vessels and inhibits the release of pro-inflammatory neuropeptides, thereby alleviating migraine pain and associated symptoms.
2. How does sumatriptan compare in terms of speed of onset to newer migraine treatments?
Sumatriptan, particularly in injectable or nasal spray formulations, offers a relatively rapid onset of action. However, newer therapies like lasmiditan (a ditan) may offer comparable or faster relief for some patients without the vasoconstrictive effects of triptans, and CGRP antagonists provide a different treatment paradigm, often for prevention.
3. Can sumatriptan be used for migraine prevention?
No, sumatriptan is indicated for the acute treatment of migraine attacks and is not used for migraine prevention. Preventative therapies are a separate class of medications aimed at reducing the frequency and severity of migraine episodes.
4. What are the most common side effects associated with sumatriptan?
Common side effects include transient sensations of tingling, heat, or flushing, dizziness, drowsiness, and nausea. More serious side effects, such as chest pain or pressure, can occur and require immediate medical attention.
5. Are there any significant regulatory hurdles for generic sumatriptan manufacturers?
Generic sumatriptan manufacturers must meet stringent regulatory requirements, including demonstrating bioequivalence to the reference listed drug and adhering to Good Manufacturing Practices (GMP) for quality and safety. Navigating the approval processes of regulatory bodies like the FDA and EMA is a standard requirement.
Citations
[1] World Health Organization. (2023). Migraine disorders fact sheet. https://www.who.int/news-room/fact-sheets/detail/migraine-disorders