OXYBUTYNIN Drug Patent Profile
✉ Email this page to a colleague
When do Oxybutynin patents expire, and when can generic versions of Oxybutynin launch?
Oxybutynin is a drug marketed by Barr Labs Div Teva, Chartwell Rx, Lannett Co Inc, Pharm Assoc, Pharmobedient Cnsltg, Accord Hlthcare, Ajanta Pharma Ltd, Amneal Pharms, Bionpharma, Impax Pharms, Osmotica Pharm Us, Pharmobedient, Rubicon Research, Unique, Zydus Pharms, Abhai Llc, Aiping Pharm Inc, Avet Lifesciences, Hibrow Hlthcare, Leading, Novast Labs, Novitium Pharma, Quantum Pharmics, Regcon Holdings, Rising, Strides Pharma, Strides Pharma Intl, Teva Pharms Usa, Tp Anda Holdings, Usl Pharma, and Watson Labs. and is included in thirty-five NDAs.
The generic ingredient in OXYBUTYNIN is oxybutynin chloride. There are sixteen drug master file entries for this compound. Thirty-nine suppliers are listed for this compound. Additional details are available on the oxybutynin chloride profile page.
DrugPatentWatch® Litigation and Generic Entry Outlook for Oxybutynin
A generic version of OXYBUTYNIN was approved as oxybutynin chloride by TEVA PHARMS USA on November 14th, 1988.
AI Deep Research
Questions you can ask:
- What is the 5 year forecast for OXYBUTYNIN?
- What are the global sales for OXYBUTYNIN?
- What is Average Wholesale Price for OXYBUTYNIN?
Summary for OXYBUTYNIN
| US Patents: | 0 |
| Applicants: | 31 |
| NDAs: | 35 |
| Raw Ingredient (Bulk) Api Vendors: | 72 |
| Clinical Trials: | 109 |
| Patent Applications: | 4,313 |
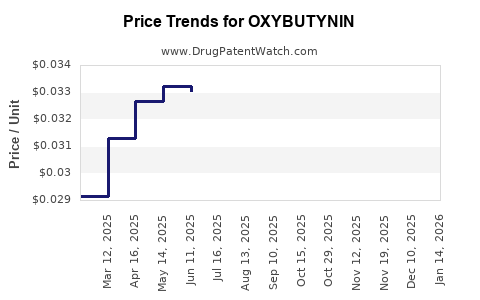
| Drug Prices: | Drug price information for OXYBUTYNIN |
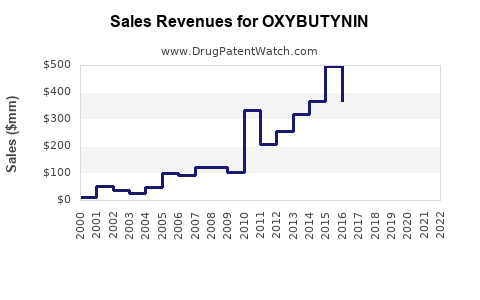
| Drug Sales Revenues: | Drug sales revenues for OXYBUTYNIN |
| DailyMed Link: | OXYBUTYNIN at DailyMed |
Recent Clinical Trials for OXYBUTYNIN
Identify potential brand extensions & 505(b)(2) entrants
| Sponsor | Phase |
|---|---|
| University of Arizona | PHASE4 |
| Patient-Centered Outcomes Research Institute | PHASE4 |
| Synmosa Biopharma Corp. | NA |
Anatomical Therapeutic Chemical (ATC) Classes for OXYBUTYNIN
Paragraph IV (Patent) Challenges for OXYBUTYNIN
| Tradename | Dosage | Ingredient | Strength | NDA | ANDAs Submitted | Submissiondate |
|---|---|---|---|---|---|---|
| OXYTROL | Transdermal System Extended-release | oxybutynin | 3.9 mg/24 hrs | 021351 | 1 | 2008-08-19 |
US Patents and Regulatory Information for OXYBUTYNIN
| Applicant | Tradename | Generic Name | Dosage | NDA | Approval Date | TE | Type | RLD | RS | Patent No. | Patent Expiration | Product | Substance | Delist Req. | Exclusivity Expiration |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Avet Lifesciences | OXYBUTYNIN CHLORIDE | oxybutynin chloride | TABLET;ORAL | 211682-001 | May 10, 2019 | DISCN | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | ||||
| Rubicon Research | OXYBUTYNIN CHLORIDE | oxybutynin chloride | TABLET, EXTENDED RELEASE;ORAL | 214415-003 | Oct 27, 2020 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| Rising | OXYBUTYNIN CHLORIDE | oxybutynin chloride | TABLET;ORAL | 209025-001 | Dec 21, 2017 | AB | RX | No | No | ⤷ Start Trial | ⤷ Start Trial | ⤷ Start Trial | |||
| >Applicant | >Tradename | >Generic Name | >Dosage | >NDA | >Approval Date | >TE | >Type | >RLD | >RS | >Patent No. | >Patent Expiration | >Product | >Substance | >Delist Req. | >Exclusivity Expiration |
EU/EMA Drug Approvals for OXYBUTYNIN
| Company | Drugname | Inn | Product Number / Indication | Status | Generic | Biosimilar | Orphan | Marketing Authorisation | Marketing Refusal |
|---|---|---|---|---|---|---|---|---|---|
| Teva B.V. | Kentera (previously Oxybutynin Nicobrand) | oxybutynin | EMEA/H/C/000532Symptomatic treatment of urge incontinence and/or increased urinary frequency and urgency as may occur in adult patients with unstable bladder. | Authorised | no | no | no | 2004-06-15 | |
| >Company | >Drugname | >Inn | >Product Number / Indication | >Status | >Generic | >Biosimilar | >Orphan | >Marketing Authorisation | >Marketing Refusal |
OXYBUTYNIN Market Analysis and Financial Projection
More… ↓