Last updated: April 23, 2026
Nitroglycerin is a long-established cardiovascular drug used primarily for acute angina and, depending on formulation, chronic angina. Its market trajectory is defined less by patent-led entry waves and more by (1) the scale of legacy supply, (2) steady generic availability, (3) persistent demand for anti-anginal therapy, and (4) competitive pressure on pricing across oral, sublingual, and transdermal delivery routes.
What does the nitroglycerin market look like by use case?
Core clinical use
Nitroglycerin is used in two high-volume therapy categories:
- Acute angina relief (fast onset, typically sublingual or spray depending on country/label)
- Chronic angina prophylaxis via extended-release oral and transdermal formulations (patch/ointment across markets)
Practical implication for demand stability
Demand is structurally supported by:
- Chronic cardiovascular disease prevalence and recurrent angina management cycles
- Broad clinician familiarity with nitroglycerin across emergency and outpatient settings
- Continued use as a rapid-acting symptomatic therapy even as long-term risk management (statins, antiplatelets, beta-blockers) evolves
How do formulation and route shape competitive dynamics?
Delivery route determines competitive “buckets”
Nitroglycerin competes within distinct product-format categories:
- Sublingual / buccal (acute relief)
- Oral extended-release (chronic dosing)
- Transdermal (chronic dosing)
- Intravenous (acute hospital settings, if offered as a distinct supply stream)
Route-level economics
These route buckets create pricing and supply differences:
- Acute relief products face less direct substitution at the point of care because onset matters and switching can affect perceived symptom control.
- Chronic formulations face more substitution across classes of anti-anginals (beta-blockers, calcium channel blockers, ranolazine where used), which increases downward pricing pressure in some geographies.
- Transdermal faces manufacturing quality constraints (content uniformity, skin absorption profile, adhesion/irritation tolerance), which can reduce the number of viable suppliers even in generic-heavy markets.
What are the main market drivers and headwinds?
Market drivers
- Ubiquitous cardiovascular need: Angina remains a large global indication class.
- Established clinical workflow: Emergency departments and outpatient cardiology programs maintain routine access pathways.
- Low “switching cost” for payers: Generic dominance lowers reimbursement barriers versus newer branded drugs.
Market headwinds
- Generic price erosion: With legacy compounds, pricing tends to compress toward low-cost supply.
- Class-level substitution: In chronic angina, competitors across multiple mechanisms can displace long-term nitroglycerin usage for parts of the patient population.
- Formulation-specific limits: Transdermal and oral regimens have dosing schedules and tolerability constraints (headache, tolerance/withdrawal patterns) that affect adherence and refill rates.
How does regulation and IP status influence the financial trajectory?
IP outcome: legacy and generic environment
Nitroglycerin is an older molecule with no sustained, proprietary revenue stream typical of late-stage patented brands. In practice, financial trajectory is tied to:
- Generic entry cycles
- Copy/authorized generic expansion
- Fragmented supply networks across oral and transdermal categories
Regulatory positioning
Nitroglycerin has long-established inclusion on national formularies for cardiovascular emergency and anti-anginal therapy. That keeps volume demand resilient but shifts financial outcomes toward manufacturing efficiency and contract pricing rather than product differentiation.
What does a “financial trajectory” look like for nitroglycerin in a generic-led market?
Revenue mechanics by stage
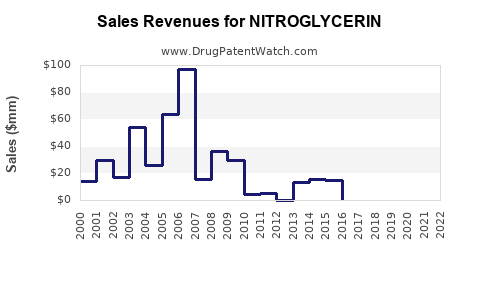
For legacy generics, the typical revenue pattern is:
- Baseline demand stability from continuous clinical use
- Margin compression as additional generic competitors enter
- Volume consolidation toward the lowest-cost suppliers and pharmacy benefit managers (PBMs)
- Occasional bumps from supply constraints, manufacturing disruptions, or formulation-level demand shifts (for example, transdermal preferences in certain populations)
What investors and R&D teams should infer
- Growth is harder to “engineer” through new clinical claims because nitroglycerin’s clinical role is already defined.
- Differentiation is more likely to come from formulation improvements (tolerability, adherence, delivery reliability) or regional supply contracts, rather than new molecular IP.
Where does nitroglycerin compete financially: alternatives and substitution risk
Substitution by therapy class
In chronic angina, clinicians often balance:
- Beta-blockers
- Calcium channel blockers
- Long-acting nitrates
- Other anti-anginals
This increases competitive pressure particularly on long-acting nitroglycerin formulations. In acute settings, substitution tends to be narrower because rapid symptom relief is the immediate target.
Pricing pressure profile
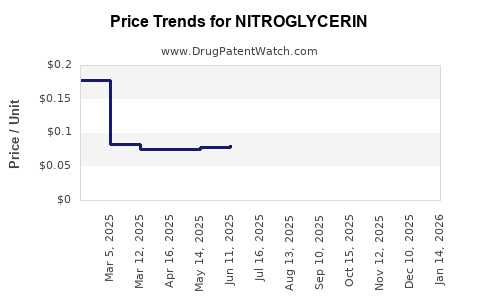
Generic competition drives:
- Lower acquisition costs for payers
- Higher inventory turnover for distributors
- Margin thinning at retail and wholesaler levels
Market and product structure: what categories drive the bulk of sales?
Most economically meaningful segments
Across most mature markets, the revenue mix is typically weighted toward:
- Chronic angina maintenance formats (transdermal and extended-release oral)
- Acute relief supply (sublingual)
Why the mix matters
- Chronic formats usually have repeat dosing patterns and steady refill volumes.
- Acute relief is event-driven but still scales with cardiovascular incidence and emergency utilization.
Competitive landscape: what types of companies win share?
Nitroglycerin competition is dominated by suppliers that can manage:
- Cost-per-tablet or cost-per-dose manufacturing
- Regulatory compliance and batch release stability
- Distribution contracts and payer formulary placement
- Supply continuity (reducing pharmacy backorders that force switching)
In practical terms, winners are often large generic manufacturers and regional distributors with mature supply chains for legacy cardiovascular products.
How does demand vary across geographies and procurement channels?
Geographical variation
Volume is driven by cardiovascular disease prevalence and prescribing habits, but financial outcomes vary by:
- Generic penetration rates
- PBM contracting structures
- Tendering and hospital procurement rules
- Reimbursement ceilings and reference pricing
Procurement channels
- Hospital procurement influences IV and acute relief distribution more directly.
- Retail pharmacy networks shape oral and sublingual access.
- Institutional formularies influence chronic transdermal and oral sustained-release positioning.
Key implications for business planning
1) Expect low-price, high-volume dynamics
Nitroglycerin tends to operate in a market where incremental unit price changes matter less than:
- supply reliability
- manufacturing yield
- contracting terms
2) Formulation strategy is the main lever
New development is most likely to translate into value if it improves:
- adherence (less dosing friction)
- tolerability (headache mitigation strategies)
- pharmacokinetic consistency (less variability across batches)
3) Commercial strategy must prioritize payer economics
In legacy cardiovascular drugs, durable revenue is more connected to:
- formulary position
- tender awards
- pharmacy reimbursement design
than to differentiation claims that have limited market room.
Key Takeaways
- Nitroglycerin’s market trajectory is defined by legacy demand plus generic-led pricing pressure.
- Revenue mechanics are shaped by delivery route: acute relief products compete within onset-critical use, while chronic formulations face broader class substitution.
- Financial performance depends more on manufacturing scale, supply continuity, and contracting than on patent-protected differentiation.
- Long-acting and transdermal categories carry repeat dosing economics but operate under sustained margin compression.
FAQs
1) Is nitroglycerin growth likely driven by innovation or by market share shifts?
Market economics favor share shifts via supply, contracting, and formulation-level improvements rather than breakthrough innovation, because the molecule’s clinical role is established in mature therapy pathways.
2) Which formulation types face the most substitution risk?
Long-acting formats used for chronic angina typically face the highest substitution risk across competing anti-anginal classes.
3) What determines supplier profitability most in a generic nitroglycerin environment?
Profitability usually hinges on manufacturing efficiency, regulatory batch release performance, and payer or tender contracting terms that set net price.
4) Does nitroglycerin demand remain stable during economic downturns?
Demand for symptomatic cardiovascular therapy tends to remain resilient, but net revenues still compress as pricing pressure and reference pricing intensify.
5) Where are the most realistic commercial “edges” for new entrants?
Edges typically come from formulation performance (tolerability and delivery consistency), reliable supply, and securing formulary or tender placement rather than new IP-driven market creation.
References
[1] U.S. National Library of Medicine. “Nitroglycerin.” DailyMed. https://dailymed.nlm.nih.gov/
[2] World Health Organization. “WHO Model List of Essential Medicines.” WHO. https://www.who.int/teams/health-product-policy-and-standards/essential-medicines/
[3] National Institute for Health and Care Excellence (NICE). “Stable angina: management.” NICE guideline. https://www.nice.org.uk/